Aphelion Company Profile
✉ Email this page to a colleague
What is the competitive landscape for APHELION
APHELION has one approved drug.
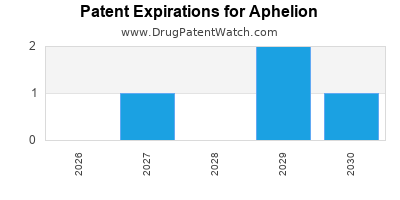
There are six US patents protecting APHELION drugs.
There are ninety-three patent family members on APHELION drugs in twenty-seven countries and fourteen supplementary protection certificates in thirteen countries.
Drugs and US Patents for Aphelion
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aphelion | PYLARIFY TRUVU | piflufolastat f-18 | SOLUTION;INTRAVENOUS | 220089-001 | Mar 6, 2026 | RX | Yes | Yes | 10,947,197 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Aphelion | PYLARIFY TRUVU | piflufolastat f-18 | SOLUTION;INTRAVENOUS | 220089-001 | Mar 6, 2026 | RX | Yes | Yes | 11,851,407 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Aphelion | PYLARIFY TRUVU | piflufolastat f-18 | SOLUTION;INTRAVENOUS | 220089-001 | Mar 6, 2026 | RX | Yes | Yes | 9,861,713 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Aphelion Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Lithuania | 2318366 | ⤷ Start Trial |
| Portugal | 3699162 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2017214470 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Aphelion Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2318366 | PA2023534 | Lithuania | ⤷ Start Trial | PRODUCT NAME: PIFLUFOLASTATAS (18F) ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/23/1746 20230724 |
| 2318366 | C20230027 00410 | Estonia | ⤷ Start Trial | PRODUCT NAME: PIFLUFOLASTAAT (18F);REG NO/DATE: EU/1/23/1746 25.07.2023 |
| 2318366 | CR 2023 00032 | Denmark | ⤷ Start Trial | PRODUCT NAME: PIFLUFOLASTAT (18F) ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/23/1746 20230725 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.