AMICUS THERAP US Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AMICUS THERAP US
AMICUS THERAP US has two approved drugs.
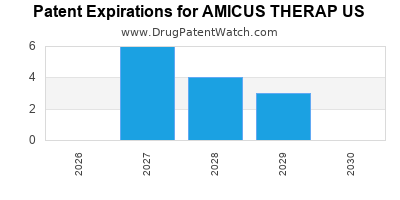
There are seventy-three US patents protecting AMICUS THERAP US drugs.
There are four hundred and forty-nine patent family members on AMICUS THERAP US drugs in forty-five countries and thirty-five supplementary protection certificates in eighteen countries.
Summary for AMICUS THERAP US
| International Patents: | 449 |
| US Patents: | 73 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for AMICUS THERAP US
International Patents for AMICUS THERAP US Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Taiwan | 201919623 | ⤷ Start Trial |
| Spain | 3013813 | ⤷ Start Trial |
| Australia | 2018220047 | ⤷ Start Trial |
| Japan | 2015509542 | ⤷ Start Trial |
| Japan | 2019510030 | ⤷ Start Trial |
| Lithuania | C2787345 | ⤷ Start Trial |
| South Korea | 102306577 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for AMICUS THERAP US Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2787345 | 53/2016 | Austria | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT ODER EIN SALZ DAVON, EINSCHLIESSLICH DES HYDROCHLORIDSALZES; REGISTRATION NO/DATE: EU/1/15/1082 (MITTEILUNG) 20160531 |
| 2787345 | 93312 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT OU UN SEL DE CELUI-CI , Y COMPRIS LE SEL DE CHLORHYDRATE; AUTHORISATION NUMBER AND DATE: EU/1/15/1082 - GALAFOLD - MIGALASTAT |
| 4273241 | LUC50007 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: CIPAGLUCOSIDASE ALFA; AUTHORISATION NUMBER AND DATE: EU/1/22/1714 20230324 |
| 4273241 | CA 2025 00017 | Denmark | ⤷ Start Trial | PRODUCT NAME: CIPAGLUCOSIDASE ALFA; REG. NO/DATE: EU/1/22/1714 20230324 |
| 2787345 | C02787345/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66108 28.10.2016 |
| 2787345 | 122016000090 | Germany | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT ODER EIN SALZ DAVON, EINSCHLIESSLICH DES HYDROCHLORIDSALZES; REGISTRATION NO/DATE: EU/1/15/1082/001 20160526 |
| 2787345 | 132016000116282 | Italy | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT O UN SUO SALE, COMPRESO IL SALE CLORIDRATO(GALAFOLD); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/15/1082, 20160531 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.