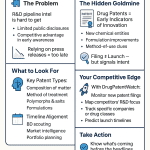
1. What ‘Patent Pending’ Actually Means in Pharma IP Law
‘Patent pending’ describes a single administrative fact: a patent application has been filed with a patent office, typically the USPTO, EPO, or via a PCT international route, and examination has not yet concluded. The designation begins at filing and ends when the application is either granted, abandoned, or rejected. In the U.S., that window averages roughly three years, though pharmaceutical applications with complex chemistry or biological sequences frequently push past five years in examination.
The legal substance here matters enormously for how you use the data. ‘Patent pending’ confers zero enforcement rights. The applicant cannot sue for infringement, obtain injunctive relief, or block a competitor’s commercial activity during pendency. What it does create is conditional liability: once a patent issues, the patentee can seek damages retroactively from the date the published application put competitors on constructive notice of the claims. That retroactivity provision in 35 U.S.C. Section 154(d) is what makes monitoring competitor pending applications commercially urgent, not just academically interesting.
1.1 First-to-File Mechanics and the Priority Date Race
The America Invents Act codified the first-to-file system in 2013, collapsing what had been a messy first-to-invent regime. The priority date, established on the day of filing, now governs who owns an invention in virtually every contested scenario. For pharmaceutical R&D teams running parallel programs on related targets, this creates a concrete operational imperative: file early and file often, even with incomplete data.
The provisional patent application (PPA) is the mechanism purpose-built for this environment. It costs a fraction of a full non-provisional, requires no formal claims, no oath or declaration, and no prior art statement. It establishes the priority date immediately and grants 12 months of ‘patent pending’ status. Within that 12-month window, the applicant must convert to a non-provisional or the provisional expires and the priority date is lost.

1.2 Rolling Provisional Applications: The Iterative Filing Architecture
Standard PPA strategy involves a single filing, conversion, and prosecution. Rolling provisional strategy, the dominant approach among large pharma IP shops, layers successive provisional filings on top of one another. Each new provisional incorporates additional subject matter, updated experimental data, or refined claims that have emerged from ongoing lab work. The non-provisional claims priority from the earliest provisional filing date for subject matter that appeared in that filing, while later-added subject matter claims priority from its own provisional filing date.
The practical result is a continuously expanding priority claim that tracks the drug’s scientific development in near real time. A compound patented in 2020 might have four or five layered provisionals covering the original molecule, then a new polymorph, then a specific formulation with improved bioavailability, then a method-of-use claim for a second indication. Each layer is dated, each layer is discoverable by competitors monitoring USPTO pending applications, and each layer reveals something about the development trajectory the company is pursuing.
For competitive intelligence purposes, tracking how a rival’s provisional filings evolve over time is more informative than reading a single granted patent. The rolling architecture is a breadcrumb trail.
Key Takeaways: Patent Pending Legal Fundamentals
- ‘Patent pending’ creates deterrence, not enforcement capability. Competitors who understand IP law will not be deterred by it alone.
- Retroactive damages from publication date (35 U.S.C. 154(d)) are the financial mechanism that makes ignoring a competitor’s pending application a genuine commercial risk.
- Rolling provisional applications are standard operating procedure at major pharma companies, and reading their sequence reveals R&D direction with surprising precision.
- The 12-month provisional window is a competitive intelligence gap: applications in that window are not yet published and are invisible to external monitoring.
2. The IP Valuation Problem: Why Pending Patents Are Mispriced Assets
Patent pending assets are systematically mispriced in pharmaceutical deal contexts because most valuation models apply binary logic: either a patent is granted and enforceable, or it is not. In practice, the probability-adjusted value of a well-constructed pending application can represent a substantial fraction of the granted patent’s value, particularly for applications with narrow, well-supported claims in crowded therapeutic areas where the grant is highly probable.
2.1 Grant Probability as a Valuation Input
USPTO grant rates for pharmaceutical patents run between 60% and 75% for non-provisional applications, depending on technology class, claim breadth, and examiner unit. Applications in Class 514 (Drug, Bio-Affecting and Body Treating Compositions) historically see first-office-action allowance rates below 20%, meaning most applications require multiple rounds of prosecution before either allowance or abandonment.
For valuation purposes, this creates a probability tree. A pending application claiming a novel active pharmaceutical ingredient (API) for a target with no current approved drugs, supported by strong Phase 1 safety data and mechanistic proof-of-concept, might carry a 70% conditional grant probability. If the eventual granted patent would support a 7-year period of market exclusivity on a drug with projected peak annual sales of $2 billion, the probability-adjusted NPV of that pending application is substantial before a single Phase 2 trial reads out.
Analysts who treat pending applications as zero-value assets until grant date are systematically undervaluing early-stage biotech pipelines and overpaying for late-stage assets where most of the patent risk has already resolved.
2.2 Claim Scope Risk: The Narrowing Problem
The second major mispricing factor is claim scope erosion during prosecution. A compound patent filed with broad genus claims covering a scaffold of potentially thousands of molecules often exits prosecution as a narrow species claim covering only the specific compound the applicant synthesized and tested. That narrowing can fundamentally alter the competitive moat. A species claim on a single enantiomer, for instance, may be designed around in 18 months by a competitor filing on the opposing enantiomer or a closely related analog.
IP valuation models for pending applications need to incorporate claim breadth scenarios, not just grant/no-grant binary outcomes. This requires reading the file wrapper, understanding what prior art the examiner has cited, and assessing whether the applicant has room to maneuver in claim amendments. Patent analytics platforms increasingly offer prosecution trajectory modeling using historical examiner data and claim language parsing, which is the right analytical tool for this problem.
2.3 Citation Forward Analysis: Predicting Strategic Value
Forward citation count, the number of subsequent patent applications that cite a given application as prior art, is a well-validated proxy for technical influence and strategic value. Applications that accumulate high forward citations during pendency are typically covering genuinely foundational territory that competitors must navigate around.
For a pending application still in prosecution, citation trajectories can be estimated using the citation patterns of closely related published applications in the same technology class. Applications covering novel kinase inhibitor scaffolds, for instance, or new bispecific antibody formats, tend to accumulate citations quickly because the field is active and applicants must distinguish their claims from the prior art. High forward citation density during pendency correlates with both higher grant probability (the examiner has evidence the art is foundational) and higher post-grant value (competitors are paying attention to it).
Key Takeaways: IP Valuation of Pending Applications
- Binary grant/no-grant valuation models systematically misprice pending pharmaceutical patents. Probability-weighted NPV models using grant rate data by technology class are more accurate.
- Claim scope erosion during prosecution is a major risk factor that requires scenario analysis, not a point estimate.
- Forward citation accumulation during pendency is a leading indicator of strategic value.
- The filing-to-publication gap (typically 18 months after filing) creates a window during which provisionals are invisible to external monitoring, concentrating risk in the pre-publication phase.
3. Competitive Intelligence: Reading the Filing Landscape Before Your Rivals Do
Patent pending data is the earliest externally observable signal of a competitor’s R&D investment decisions. Clinical trial registrations at ClinicalTrials.gov are earlier in some respects, but they capture trials that have already been designed and funded. Patent applications are filed years before IND submission, frequently at the stage where a compound has demonstrated acceptable in vitro potency and selectivity but before significant preclinical toxicology has been run. That makes filing patterns a 2-to-4-year leading indicator relative to what you will see in clinical registrations.
3.1 Filing Cluster Analysis: Identifying Strategic Pivots
The most actionable CI signal from patent pending data is not a single application, but a sudden increase in application density around a specific target class, mechanism, or indication. If a competitor files three to five applications in 18 months covering different aspects of a single biological pathway, GPR84 agonism or PCSK9 degradation, for instance, that cluster almost certainly reflects an internal decision to resource a full program. The filing cluster typically precedes IND submission by 18 to 36 months.
Tracking filing clusters requires continuous monitoring rather than periodic landscape snapshots. Automated alerting systems configured to flag new applications by competitor assignee, CPC classification, or keyword combination can surface these signals within days of publication. The 18-month publication delay from filing means you will always be working with data that is at least a year and a half old, which is why monthly monitoring cadences catch signals roughly on time, while annual patent reviews catch them far too late.
3.2 White Space Mapping: Where Are Competitors Not Filing?
Gap analysis on the pending application landscape, identifying therapeutic targets or mechanisms with minimal recent filing activity relative to their biological validation status, is among the highest-value outputs of systematic patent intelligence.
A target that appears in peer-reviewed literature with compelling genetic validation data but has generated fewer than 10 patent applications globally in the past three years is either genuinely difficult to drug, or it is an underexplored opportunity where first-mover advantage via early filing could establish a defensible IP position. Cross-referencing sparse patent filing density with high publication density in academic literature is a particularly reliable method for identifying these pockets, because it reveals cases where academic research has validated a target that industry has not yet aggressively pursued.
3.3 Assignee-Level Filing Trajectory as a Strategic Signal
Tracking total filing volume, IPC classification distribution, and PCT designation patterns for a competitor’s assignee account over five-year windows reveals strategic intent more reliably than any analyst report. A company historically concentrated in cardiovascular IP that begins filing aggressively in oncology classifications is signaling a business development or organic R&D pivot. A company with a historically broad discovery pipeline that suddenly concentrates filings in a single indication is signaling a focus shift, often driven by Phase 2 data that has not yet been publicly disclosed.
PCT application designations, the list of national phases into which an applicant files, also carry signal. A PCT application designating the U.S., EU, Japan, China, Canada, and Australia suggests commercial interest across major markets. A PCT application designating only the U.S. and Europe suggests either cost management or a deliberate decision not to pursue exclusivity in emerging markets, which has pricing and access strategy implications.
Key Takeaways: Competitive Intelligence from Patent Pending Data
- Filing clusters around a specific pathway are a 24-to-36-month leading indicator of a competitor’s clinical program.
- White space mapping using patent density vs. academic publication density identifies genuinely open opportunities.
- PCT designation patterns reveal geographic commercial intent years before a product launch.
- Monitoring must be continuous, not periodic. Annual patent reviews catch competitive signals too late to alter R&D investment decisions meaningfully.
Investment Strategy Note for Analysts Publicly traded biotech companies often present narrow pipeline disclosures in investor communications. Cross-referencing disclosed pipeline assets with pending application clusters under the same assignee can surface undisclosed programs. A company describing a ‘research program’ in investor materials accompanied by four recent patent filings in the same target class should be modeled as having a likely IND candidate in late preclinical, which materially affects probability-weighted pipeline valuation.
4. R&D Pipeline Management: Patent Data as a Capital Allocation Tool
The pharmaceutical R&D model has a well-documented attrition problem. Only roughly 10% of drug candidates entering Phase 1 reach approval. The aggregate cost of that attrition, accounting for the failed programs that subsidize eventual successes, pushes the true cost per approved drug above $2.5 billion by most rigorous estimates. In that context, using patent pending data to cull programs earlier, before substantial capital is committed, is not a theoretical best practice. It is a capital efficiency imperative.
4.1 Patent Overlap Analysis: The ‘Crowded Art’ Decision Gate
Before committing Phase 1 funding to a drug candidate, the IP team should run a comprehensive pending application overlap analysis for the relevant target, mechanism, and chemical scaffold. An analysis that reveals a competitor already has four issued patents and two pending applications covering the exact chemical scaffold your program is pursuing should trigger a go/no-go deliberation that questions whether the program can achieve commercially meaningful differentiation, even if it reaches approval.
This is distinct from a freedom-to-operate analysis, which asks whether your program can launch without infringing existing rights. Overlap analysis asks a prior and more strategic question: even if you can get around existing IP, can you build a defensible patent position of your own? A program entering a highly claimed chemical space may be able to operate freely and still produce a drug candidate that is extremely difficult to patent in ways that support sustainable market exclusivity.
4.2 Patent Cliff Anticipation: Using Competitor Pending Data to Time Pipeline Investment
The patent cliff, the revenue drop that follows loss of exclusivity on a high-revenue product, is not an unpredictable event. The expiration dates of existing patents are known years in advance. What is less predictable is whether lifecycle management strategies, including new formulation patents, new indication patents, or successor molecule filings, will extend effective exclusivity beyond the primary compound patent expiry.
Monitoring competitor pending applications for secondary patents on drugs approaching loss of exclusivity (LOE) gives a measurable probability estimate for effective exclusivity extension. A competitor drug with a primary compound patent expiring in 2027 that has generated six pending applications since 2022 covering extended-release formulations, new salt forms, and combination use patents is signaling an active lifecycle management effort. Those pending applications, if granted, could shift the effective LOE by two to four years relative to what the primary patent expiry date suggests.
For your own pipeline planning, this matters because generic entry timing into a competitor’s market directly affects the revenue opportunity size for differentiated entrants. A two-year LOE extension in a $5 billion market is a two-year shift in competitive dynamics that should be reflected in your revenue forecasts.
4.3 The ‘Fail Fast’ Discipline: Using Filing Density as a Red Flag
Programs operating in therapeutic areas where the top ten competitors are collectively filing more than 50 pending applications per year face a structural IP challenge: the cost of achieving and maintaining a differentiated, defensible patent position is high, and the probability that a competitor will file something that blocks a commercially meaningful claim is material. For small and mid-sized biotech companies without the resources for a sustained multi-front patent prosecution campaign, entering these high-density filing areas without a genuinely novel mechanism or structural scaffold is a capital allocation mistake.
Patent filing density analysis, run before committing to a new therapeutic area, is a direct input into that go/no-go capital allocation decision. Platforms like PatSnap and Derwent Innovation generate density heatmaps by CPC classification that make this analysis tractable in a matter of hours, not weeks.
Key Takeaways: R&D Pipeline Capital Allocation
- Patent overlap analysis should be a formal decision gate before Phase 1 funding commitments, not a post-hoc compliance check.
- Secondary filing activity around drugs approaching LOE is a quantifiable predictor of effective exclusivity extension and should feed directly into competitive market forecasting.
- Filing density analysis in a new therapeutic area is a capital allocation input, not just a legal risk assessment.
5. Evergreening Roadmaps: The Full Technology Lifecycle of a Blockbuster Patent Portfolio
Evergreening, the practice of filing successive patents on new aspects of an existing drug to extend effective market exclusivity, is structurally embedded in the economics of pharmaceutical R&D. A primary compound patent filed at the time of discovery and expiring 20 years later will, by the time of launch, have only 10 to 12 years of remaining patent life. That window is shorter than the time needed to recoup development costs for most therapeutics, which creates a systemic incentive to build out secondary patent estates.
5.1 The Patent Estate Architecture for a Typical Small Molecule Drug
A mature blockbuster small molecule patent portfolio typically has five distinct patent layers, each with its own filing timeline and contribution to effective market exclusivity.
The first layer is the compound patent, which claims the active ingredient itself, typically as a genus claim with a species focus on the lead candidate. This patent is filed earliest, frequently while the compound is in late lead optimization or early preclinical, and has the earliest expiration date. The compound patent is the primary target for Paragraph IV certification by generic filers under Hatch-Waxman.
The second layer is the polymorph and salt form patent estate. Most APIs can be crystallized in multiple forms with different physical properties. Polymorph patents claiming the crystalline form used in the commercial formulation are filed after characterization work confirms which polymorph offers optimal stability and manufacturability. These patents often expire four to six years after the compound patent.
The third layer is the formulation patent estate. Specific excipient combinations, coating technologies, drug delivery systems like extended-release matrices, and particle size specifications all produce patentable inventions if they provide measurable clinical benefit. Extended-release formulations that reduce dosing frequency, for example, have consistently survived validity challenges because the reduced dosing burden represents genuine clinical value, not purely incremental modification.
The fourth layer is the method-of-use patent estate, which claims specific indications, dosing regimens, or patient subpopulations. Method-of-use patents filed on new indications after initial approval are common. Dosing regimen patents, which claim specific dose amounts, frequency, or titration schedules optimized for efficacy or tolerability, are increasingly litigated because they appear frequently in Orange Book listings and generic filers must certify against them.
The fifth layer is the combination patent estate, which claims co-administration or fixed-dose combinations with other agents. These patents are most valuable when the combination reflects an emerging standard of care, as in checkpoint inhibitor combinations or HIV two-drug regimens.
5.2 Pending Application Timing Within the Evergreening Roadmap
Reading a competitor’s evergreening roadmap requires tracking not just granted patents but the sequence and timing of pending applications across all five layers. A company that files a compound patent in Year 0, polymorph patents in Years 2-3, formulation patents in Years 4-5, and begins filing method-of-use patents around Year 6-7 is executing a textbook lifecycle management strategy. The pending applications in Years 4-7 are the most actionable for generic manufacturers planning Paragraph IV strategies because they identify the secondary patent layers that will require certification challenges if generics want to avoid extended stay provisions.
For branded manufacturers, the pending applications in Years 8-10 of a drug’s commercial life represent the defensive perimeter. These late-stage filings on pediatric formulations, new salt forms with improved solubility, or next-generation delivery devices are designed to reset the exclusivity clock by 3-5 years. Tracking competitor pending applications in this phase tells you whether a competitor is actively defending their exclusivity position or allowing it to erode.
5.3 Orange Book Listing Strategy and Pending Patents
The FDA Orange Book lists patents that the NDA holder certifies as claiming the approved drug product or method of using it. Listing a patent in the Orange Book triggers a 30-month stay on generic approval upon a Paragraph IV certification, which is the core mechanism by which patent disputes delay generic entry.
Pending applications become Orange Book-relevant the moment they issue. Companies with pending applications on drugs approaching LOE are in an active race: if the pending application issues before the primary compound patent expires, it can be listed and will apply to any generic ANDA filers. If it issues after the primary patent expires but while there are still listed patents generating stays, it adds another layer of protection.
Monitoring competitor pending applications against the Orange Book timelines for their products is, therefore, a direct input into generic drug entry forecasting. An application published in 2024 with a likely issuance date in 2026, covering a new formulation of a drug whose primary compound patent expires in 2027, has the potential to delay generic entry by a full 30-month stay period, pushing the effective LOE to 2029 or beyond.
Key Takeaways: Evergreening and the Patent Estate Architecture
- A blockbuster small molecule drug typically has five distinct patent layers. Reading competitor pending applications requires tracking all five, not just the compound patent.
- The timing of secondary filings relative to the primary compound patent expiry is the most actionable intelligence for both generic challengers and branded lifecycle managers.
- Orange Book listing eligibility for pending applications creates a specific tactical race: issuance timing relative to primary LOE dates determines whether a secondary patent generates 30-month stay protection.
Investment Strategy Note for Analysts When modeling a branded drug’s revenue cliff, do not use the primary compound patent expiry as the effective LOE date without first auditing the secondary pending application portfolio. Failure to account for secondary patent filings and their likely Orange Book listing eligibility routinely produces LOE forecasts that are 2-4 years early, creating systematic mispricing opportunities in both long and short directions.
6. Biologics and Biosimilars: The Patent Thicket Architecture in Detail
The patent landscapes around biologic drugs differ from small molecules in both legal structure and strategic intent. The Biologics Price Competition and Innovation Act (BPCIA) created the biosimilar approval pathway and the ’12-year data exclusivity’ protection for reference products, but it did not create a simple analog to the Hatch-Waxman paragraph IV mechanism. Instead, the BPCIA established the ‘patent dance,’ a structured exchange between the biosimilar applicant and the reference product sponsor that produces litigation over selected patents from the originator’s patent list.
6.1 The Biologic Patent Thicket: Structure and Construction
A mature biologic patent estate is deliberately constructed to be dense. AbbVie’s adalimumab (Humira) estate is the most widely cited example: over 250 patents listed in the FDA Purple Book by 2023, covering the antibody sequence, formulation, manufacturing processes, methods of use, dosing devices, and combinations. That density was built over two decades through continuous pending application filings. Understanding how that estate was constructed, layer by layer, is more instructive than cataloguing the final count.
Antibody patents at the compound level typically claim the CDR sequences or the variable domain amino acid sequences. These are the core blocking patents that biosimilar developers must either design around or challenge. Pending applications at this level, typically filed early in development using provisional applications with broad sequence claims, are the most valuable and most carefully monitored by biosimilar developers.
Manufacturing process patents, which claim specific cell culture conditions, purification sequences, or analytical methods that achieve defined quality attributes, are frequently the most defensible patents in a biologic estate. Because biosimilar manufacturers must produce a product with a demonstrated high degree of similarity to the reference, they typically cannot materially deviate from the manufacturing process. This structural constraint means that manufacturing process patent claims have broader effective coverage for biologics than they would for small molecules.
Device and delivery system patents cover the autoinjector, prefilled syringe, or other delivery device used to administer the biologic. These patents have been effective in delaying biosimilar interchangeability designations because a biosimilar must be demonstrated substitutable for the reference product ‘without the intervention of the health care provider who prescribed the reference product,’ which typically requires using the same or equivalent delivery device.
6.2 Reading Pending Biologic Applications for Biosimilar Strategy
For a biosimilar developer, the pipeline of pending applications against the reference product is the primary strategic risk register. An application pending at the USPTO that, if granted, would cover the manufacturing cell line, the purification method, or the specific formulation of the commercial product can become a blocking patent between biosimilar application filing and launch.
The BPCIA patent dance generates litigation timelines that can be planned with considerable precision once the reference product sponsor’s full patent list is known. But pending applications that issue after the biosimilar application is filed can be added to subsequent litigation rounds. Monitoring the reference product sponsor’s pending applications continuously through the biosimilar development timeline is not optional for a developer with a serious commercial program.
Pending applications with sequence-based claims are assessed using degenerate sequence searching tools that map the claimed sequences to the biosimilar candidate’s known sequence. NLP-based claim parsing tools, now standard in platforms like PatSnap and Lumenci’s FTO suite, can automate preliminary assessments of sequence coverage risk and flag applications requiring manual review by a structural biologist and patent counsel. This combination of automated screening and expert review is the current state of the art in biologic patent risk management.
Key Takeaways: Biologic Patent Thickets
- Biologic patent estates are intentionally multi-layered, covering sequence, manufacturing process, formulation, delivery device, and indication. Biosimilar strategy requires mapping all layers, including pending applications.
- Manufacturing process patents have structurally broader effective coverage for biologics than for small molecules because biosimilar developers cannot arbitrarily redesign the production process.
- The BPCIA patent dance creates structured litigation timelines, but pending applications that issue post-filing can enter subsequent litigation rounds, requiring continuous monitoring.
7. Freedom-to-Operate Analysis: Pending Applications Are the Hidden Landmines
Freedom-to-operate (FTO) analysis answers a specific binary question: can a defined product or process be commercialized in a specific territory without infringing a valid, enforceable claim of an existing patent? It is distinct from patentability analysis, which asks whether the invention itself is novel and non-obvious. Both analyses are necessary, and both are systematically incomplete without coverage of pending applications.
7.1 Why Pending Applications Must Be Included in FTO Searches
The construction of an FTO opinion that excludes pending applications is legally defensible but practically negligent. Here is the mechanism for why.
A pending application is published 18 months after its earliest priority date. From the date of publication, it puts the world on constructive notice of the claimed subject matter. Under 35 U.S.C. 154(d), if the application later issues as a patent and the claims are substantially identical to those in the published application, the patentee can recover reasonable royalties from any infringer who had actual notice of the published claims, back to the publication date. This retroactivity provision means that a competitor who begins commercial activity after a relevant pending application is published is taking on infringement liability that accrues from the publication date, even if the patent has not yet been granted.
In practical terms: a biotech company that launches a product in 2025 without clearing a competitor’s published pending application from 2024, and that application issues in 2026, faces retroactive infringement liability going back to the 2024 publication date plus prospective liability. The failure to include the pending application in the 2025 FTO opinion created liability that a competent search would have identified.
7.2 Claim Construction Under Prosecution: The Moving Target Problem
The specific challenge of including pending applications in FTO analysis is that claims are not fixed during prosecution. Claims are amended, narrowed, broadened within prosecution history estoppel limits, and occasionally allowed in a form materially different from what was filed. An FTO opinion written against a pending application in 2024 may be inaccurate by 2026 if the claims have been substantially amended during examination.
Responsible FTO practice for high-value pending applications involves periodic re-review, at minimum at each major prosecution milestone: after first office action response, after final rejection response, and immediately prior to any notice of allowance. For applications covering foundational biologic targets or platform manufacturing technologies, quarterly monitoring of prosecution status through USPTO Patent Center is standard practice at major pharma companies.
7.3 Designing Around Pending Claims: Timing and Strategy
One structural advantage of pending application visibility is that design-around strategies can be initiated while the claims are still evolving. If a competitor’s pending application appears to cover a broad genus of compounds that includes your lead candidate, the question is not only whether you can license or challenge the patent, but whether you can make structural modifications to your candidate that would place it outside the claimed genus without compromising pharmacological activity.
This design-around analysis requires close collaboration between medicinal chemists, who can assess the structural modification options, and patent counsel, who can map those modifications against the pending claim language and prosecution history. The earlier this analysis begins, the more options are available. A design-around initiated against a pending application with claims still being examined costs a fraction of a design-around forced by an issued blocking patent combined with an infringement suit.
Key Takeaways: FTO and Pending Applications
- FTO opinions that exclude pending applications are incomplete. Retroactive liability under 35 U.S.C. 154(d) makes pending applications a real and quantifiable infringement risk from the date of publication.
- Claim construction evolves through prosecution. FTO opinions on high-value pending applications require periodic refresh at prosecution milestones.
- Design-around analysis is most cost-effective when initiated against pending claims, before issuance, when structural options are still available without forced product redesign.
8. M&A Due Diligence: How Pending Patents Move Deal Valuations
In biopharmaceutical M&A, the patent portfolio is the primary asset being acquired. Cash flows from currently marketed products will eventually be replicated by generics or biosimilars. The perpetual value in a pharma acquisition derives from the patent-protected pipeline. Pending applications, as the forward-looking portion of that portfolio, carry disproportionate strategic weight relative to their current legal status.
8.1 The Pending Application Audit: What the Standard Due Diligence Process Misses
Standard M&A IP due diligence focuses heavily on granted patents: ownership verification, assignment chain integrity, maintenance fee status, and litigation history. These are necessary but insufficient for accurate valuation.
Pending applications require a parallel audit covering six distinct dimensions. First, prosecution history review: has the applicant made any claim amendments or arguments that create prosecution history estoppel, limiting the doctrine of equivalents scope of any eventually issued claims? Second, prior art assessment: has the examiner cited prior art that, in a rigorous analysis, might anticipate or render obvious the pending claims? If so, the grant probability is lower than the base rate. Third, continuation strategy: has the applicant filed or indicated intent to file continuation or continuation-in-part applications? A robust continuation strategy means the pending application estate is actively being expanded even as earlier applications are pending, which increases the total value but also the complexity of the portfolio. Fourth, inter partes review (IPR) risk: are there any petitions filed or likely to be filed against pending applications’ parent cases that could affect the family’s validity? Fifth, terminal disclaimer issues: have terminal disclaimers been filed that link the pending application’s term to a parent case, potentially reducing effective exclusivity? Sixth, foreign counterpart status: do PCT or foreign national phase applications exist, and what is their prosecution status in key commercial territories?
8.2 Pending Pipeline Patents and Acquisition Premiums
Acquisition premiums in biopharmaceutical deals correlate more strongly with late-stage clinical pipeline quality than with currently marketed revenue, because the market is buying future exclusivity. Pending applications that protect Phase 2 and Phase 3 assets are directly priced into those premiums.
A target company with a Phase 3 asset in a large indication, clean patent ownership on the compound patent, and two well-constructed pending formulation and method-of-use applications provides meaningfully better exclusivity depth than the same Phase 3 asset with no secondary filings. The delta between those two scenarios is not speculative: it is the probability-weighted difference in years of post-launch market exclusivity, multiplied by the net revenue per year, discounted appropriately.
For acquirers, this means the pending application portfolio is not a footnote in the IP section of the data room review. It is a primary valuation input that requires the same analytical rigor as the clinical data package.
8.3 Red Flags in Pending Application Portfolios
Specific pending application characteristics should trigger valuation adjustments or deal structure modifications. Claims with extensive amendments that have substantially narrowed from original scope, combined with prior art of record that comes close to anticipating the remaining claims, suggest high invalidity risk and low commercial scope. Applications with pending interference or derivation proceedings indicate ownership disputes that must resolve before the acquiring company can rely on the patent. Applications that are national phase entries from PCT filings with missed response deadlines in key territories indicate either careless portfolio management or deliberate decisions not to pursue protection in specific markets.
The absence of secondary filings around a high-revenue marketed drug approaching LOE is itself a red flag. It suggests either that the IP team failed to execute lifecycle management filings at the appropriate time, or that the drug’s commercial profile did not support the investment, neither of which is a positive signal about portfolio management quality.
Key Takeaways: M&A Due Diligence
- Pending application due diligence requires six-dimensional analysis: prosecution history estoppel, prior art of record, continuation strategy, IPR risk, terminal disclaimers, and foreign counterpart status.
- Acquisition premiums on pipeline assets should be adjusted for pending application portfolio depth. Secondary filings around Phase 3 assets directly support a higher exclusivity-adjusted NPV.
- Specific prosecution history red flags, including heavy claim narrowing and close prior art of record, should trigger explicit valuation adjustments or representations and warranties in deal documentation.
Investment Strategy Note for Analysts Announced biopharmaceutical acquisitions can be evaluated for quality of IP coverage by running a pending application analysis within 48 hours of deal announcement using public USPTO and EPO databases. Deals where the acquirer is paying a large premium for an asset with thin or heavily amended pending application coverage deserve higher scrutiny.
9. Licensing Patent-Pending Inventions: Deal Structures and Risk Transfer
Licensing a patent-pending invention is commercially standard in pharmaceutical partnering, but the legal risk profile differs materially from licensing a granted patent. The licensor cannot represent that the licensed rights currently exist in enforceable form. The licensee acquires rights to something that may not exist in its current claimed form, or may not exist at all, by the time the product reaches market.
9.1 License Agreement Mechanics for Patent-Pending Assets
License agreements covering patent-pending inventions typically include several provisions that do not appear in standard granted patent licenses. The grant clause will include rights to ‘any patents issuing from’ the specified applications, plus continuation, divisional, and continuation-in-part applications claiming priority to the same specification. This broad coverage language is necessary because the licensed application may divide or continue before issuance.
Royalty rate structure for pending application licenses commonly uses a stepped rate: a reduced rate during pendency, a higher rate upon issuance, and a further adjustment upon claim scope finalization or upon regulatory approval of a product covered by the claims. Some agreements include a royalty refund or reduction clause that activates if the application is abandoned or issues with claims so narrow that they do not cover the licensed product.
Representations and warranties in pending application licenses are necessarily limited. The licensor can represent clean ownership of the application, the absence of known prior art or interference proceedings, and that it has not made prior assignments. It cannot represent validity or enforceability of claims that have not yet been examined. Sophisticated licensees negotiate for specific prosecution covenants: the licensor must keep the application in good standing, must consult with the licensee before making claim amendments that would affect coverage of the licensed product, and must not abandon the application without the licensee’s consent.
9.2 Field of Use Restrictions: Maximizing Value Across Applications
Biopharmaceutical compounds and biological platforms frequently have multiple potential applications across different disease areas. A licensing structure that grants exclusive rights to a pending application across all fields of use permanently transfers value in fields the licensor might later develop internally or license to a better-positioned partner.
Field of use restrictions in licenses on patent-pending assets are particularly valuable because the pending stage is often the point at which the full commercial potential of the invention is least well-defined. Limiting the license to the ‘treatment of Type 2 diabetes in human subjects’ rather than granting rights to ‘all therapeutic uses’ preserves oncology, cardiovascular, and other indications for separate licensing. This structure also gives the licensee a defined scope to validate commercially before the license is fully valued.
9.3 Sublicensing Rights and Change-of-Control Provisions
Sublicensing rights in pending application licenses carry specific risk. If the licensee sublicenses and then the underlying patent application is abandoned or issues with narrow claims that do not cover the sublicensed product, the licensee has created a sublicense to rights that do not exist. License agreements should address this scenario explicitly, either by conditioning sublicensing rights on patent issuance, or by requiring the licensee to include representations to sublicensees about the pending nature of the underlying IP.
Change-of-control provisions are particularly important for pending application licenses given the active M&A environment in biopharmaceuticals. An exclusive license on a pending application that passes to a large competitor through an acquisition of the licensee can create IP positions the original licensor did not intend. Anti-assignment provisions with a change-of-control trigger allow the licensor to negotiate the assignment to the acquirer, rather than having it occur automatically.
Key Takeaways: Pending Application Licensing
- License grant clauses must expressly cover continuations, divisionals, and CIPs. A grant limited to the filed application number will miss related applications that may have broader or differently structured claims.
- Prosecution covenants, including consultation rights on claim amendments and prohibition on abandonment without licensee consent, are essential deal terms that protect the licensee’s investment.
- Field of use restrictions are more valuable at the pending stage than at issuance, because the full application scope is unknown and multiple valuable fields can be preserved separately.
10. Market Forecasting with Patent Filing Data
Patent applications are filed 2-5 years before a drug candidate enters clinical trials and 8-12 years before potential approval. That gap makes them among the most temporally extended leading indicators available for pharmaceutical market forecasting.
10.1 Therapeutic Area Trajectory Forecasting
Annual filing counts by IPC class and CPC subclass can be used to construct innovation activity indices for specific therapeutic areas. A therapeutic area that generated 200 relevant pending applications per year from 2018-2021 and is now generating 500 per year from 2022-2025 is experiencing a doubling of R&D investment as measured by IP commitment. Historical analysis shows that filing rate increases in a therapeutic area precede clinical trial registration increases by roughly 24 months and approval rate increases by 8-10 years.
For market forecasters, this means that a 2025 filing surge in a specific modality, GLP-1 dual agonists targeting insulin resistance, for instance, or KRAS degraders beyond G12C, can be used to project that the competitive density in those spaces will be substantially higher by 2030-2032 than current clinical trial registrations suggest. This affects revenue concentration assumptions, pricing pressure forecasts, and market share modeling for assets in those spaces.
10.2 Generic Entry Timing Models Using Pending Secondary Patents
The standard generic entry timing model uses primary compound patent expiry, accounting for Patent Term Extensions (PTEs) and any existing regulatory exclusivity that runs concurrently. That model produces a base LOE date that is reliably wrong in cases where an active secondary filing program exists.
A more accurate model adds a probability-weighted assessment of pending secondary patents: for each pending application that could be listed in the Orange Book upon issuance, assign a grant probability based on prosecution history and examiner data, multiply by the expected 30-month stay period if a generic filer triggers litigation under Hatch-Waxman, and weight by the probability that the issuance date precedes primary LOE. The output is a probability distribution over effective LOE dates rather than a single point estimate. That distribution is substantially more useful for generic entry planning, market access strategy, and investment modeling.
Key Takeaways: Market Forecasting
- IPC/CPC filing count indices are 8-12 year leading indicators for approval-stage competitive density. Forecasters who ignore them will systematically underestimate competitive crowding in emerging high-activity spaces.
- Single-point LOE date estimates from primary compound patent expiry are reliably wrong for drugs with active secondary filing programs. Probability-weighted LOE distributions using pending application data are more accurate.
11. AI/ML: How It Changes Both Drug Discovery and Patent Intelligence
The integration of large language models, graph neural networks, and generative chemistry platforms into pharmaceutical R&D is not an incremental efficiency gain. It is compressing the timeline between target identification and IND candidate nomination from years to months for specific target classes. That compression will substantially increase the volume and velocity of new patent filings, and it is already beginning to do so.
11.1 The Inventorship Problem for AI-Generated Compounds
USPTO guidance as of 2024 requires that a human make a ‘significant contribution’ to each claim in a patent application for that person to qualify as an inventor. AI cannot be named as an inventor. For pharmaceutical companies using generative chemistry platforms that propose novel scaffolds or optimize lead compounds algorithmically, the inventorship question is not theoretical. It is a filing requirement that demands a documented answer for every application.
The most defensible inventorship position for AI-assisted drug discovery is one where the human scientists made specific, documented decisions about the AI model architecture used, the training data selected and excluded, the objective functions optimized, the outputs filtered and advanced, and the wet lab validation designed and interpreted. Under that framework, the AI is a sophisticated tool, analogous to a high-throughput screening robot, and the humans who directed its use and validated its outputs are the inventors.
Sloppy documentation of AI contributions in the drug discovery process is a patent validity risk. An examiner or IPR petitioner who can demonstrate that the claimed invention was generated entirely by an autonomous AI system with no documented significant human contribution can challenge inventorship, which in the U.S. is a basis for patent invalidity. Building documentation protocols that contemporaneously capture human scientific contributions at each decision point in AI-assisted discovery is IP hygiene that needs to be established now, not after the first challenge.
11.2 AI-Powered Patent Intelligence: Capabilities and Limitations
The current generation of AI patent analytics platforms provides capabilities that were not technically achievable five years ago. Semantic patent search using dense vector embeddings allows retrieval of conceptually similar patents regardless of whether they share keyword terms with your query, which substantially improves recall in FTO and landscape analyses. NLP-based claim parsing can extract specific structural elements, biological targets, or method steps from large claim sets and map them against query structures or processes. Prosecution history analysis tools can summarize claim amendments across entire patent families and flag arguments made to the examiner that may create estoppel.
These tools are genuinely useful and represent a meaningful productivity gain. They are not a substitute for the expert human analysis that translates a patent claim, as actually written and as interpreted by courts applying the Phillips claim construction standard, into an accurate infringement opinion. LLM-based patent analysis tools frequently miss structural claim limitations that are embedded in technical language the model misparses, and they cannot reliably assess whether a prior art reference anticipates or renders obvious a claim, because that analysis requires domain-specific scientific expertise. The correct deployment model uses AI tools for scale and initial screening, with expert human review on high-risk applications.
Key Takeaways: AI in Patent Intelligence
- AI-assisted drug discovery creates an inventorship documentation requirement that must be addressed contemporaneously, not after challenge. Every AI-assisted application needs a human contribution log.
- AI patent analytics platforms materially improve search recall and prosecution history analysis. They do not replace expert claim construction and infringement analysis for high-stakes FTO opinions.
- The AI-driven increase in compound generation velocity will significantly increase pending application volume in high-activity classes, making continuous automated monitoring more necessary and manual periodic searches less sufficient.
12. Regulatory Exclusivity vs. Patent Term: The Stacking Problem
A drug’s actual period of market protection derives from two overlapping but legally distinct systems: patent protection and regulatory exclusivity. Understanding how they interact, overlap, and sometimes conflict is essential for accurate LOE modeling and lifecycle management strategy.
12.1 The Regulatory Exclusivity Stack
The FDA grants exclusivity periods that run independently of patent protection, based on characteristics of the drug or the approval pathway. New Chemical Entity (NCE) exclusivity provides five years of data exclusivity for drugs containing a new active moiety, during which the FDA will not approve an ANDA that relies on the NDA holder’s safety and efficacy data. Orphan Drug Exclusivity (ODE) provides seven years of market exclusivity for drugs approved for rare diseases, defined as affecting fewer than 200,000 patients in the U.S. Pediatric Exclusivity adds six months to existing patent terms or exclusivity periods when the NDA holder conducts pediatric studies at FDA’s request. New Clinical Investigation exclusivity provides three years for new indications, formulations, or conditions of use that required new clinical studies.
Biologics Reference Product Exclusivity under the BPCIA provides 12 years of data exclusivity from the date of first approval, during which no biosimilar can be approved based on the reference product’s BLA data. This 12-year period is the dominant exclusivity driver for biologics and runs regardless of patent status.
12.2 Patent Term Extension and PTE Mechanics
Patent Term Extensions (PTEs) under 35 U.S.C. 156 restore some of the patent term consumed by FDA regulatory review. The extension equals half the clinical development time plus the full NDA review period, capped at five additional years and at a total extended term of 14 years from FDA approval. Only one patent per regulatory review is eligible for PTE, and the patentee must select which patent to extend within 60 days of FDA approval.
The PTE selection decision is itself a strategic IP question. Extending the compound patent protects the core active ingredient the longest. Extending a formulation or method-of-use patent may provide fewer additional years but may also have a later original expiry date, making the effective terminal date of protection further in the future. For drugs with complex secondary patent estates, PTE selection analysis should model all eligible patents against the expected competitive entry timeline to determine which extension produces the longest effective exclusivity.
12.3 Stacking Calculation for LOE Forecasting
Accurate LOE date calculation for a drug with a full protection stack requires modeling the expiry of each patent layer, each regulatory exclusivity type, any PTE, and any SPCs in European markets. The operative protection date is the last expiry in the stack that remains relevant to blocking the specific competitive threat being assessed.
For a hypothetical drug with a compound patent expiring in 2027, a PTE extending it to 2029, NCE exclusivity running through 2028, an extended-release formulation patent expiring in 2031 with a pending PTE application, and ODE running through 2032, the effective LOE date against a generic competitor seeking to use the NDA holder’s clinical data is 2032. Against a generic competitor with independent clinical data who is not relying on Hatch-Waxman, the operative date is the last blocking patent expiry, which may be the formulation patent at 2031.
Key Takeaways: Regulatory Exclusivity and Patent Term Stacking
- Patent protection and regulatory exclusivity are legally independent systems that must be modeled separately and then stacked to produce effective LOE dates.
- PTE selection is a strategic decision, not an administrative formality. Selecting which patent to extend requires modeling all eligible patents against the competitive entry timeline.
- European SPCs provide a separate exclusivity mechanism that frequently extends protection beyond U.S. patent expiry, creating territory-specific LOE date differences that global revenue models must capture.
13. Ethical and Legal Constraints: Patent Thickets, Transparency Acts, and Competitive Intelligence Boundaries
Patent thickets in pharmaceuticals produce commercially clear incentives and socially contested outcomes. The incentive structure is straightforward: every additional granted patent in the Orange Book is a potential 30-month stay trigger. Every additional Orange Book-listed patent requires a Paragraph IV certification from a generic filer, and each certification is a potential infringement suit. The combinatorial result of dense patent listing is that a generic entrant may face sequential 30-month stays from multiple patent litigations, not just one, which can extend the effective exclusivity far beyond what any single patent would provide.
13.1 The Orange Book Transparency Act and Its Practical Implications
The Orange Book Transparency Act of 2020 and subsequent FTC activity have increased scrutiny on inappropriate Orange Book listings. The FTC’s 2023 challenge to Orange Book listings by several pharmaceutical companies argued that patents on delivery devices, such as drug-device combination inhalers, are not properly listable because they do not claim the drug substance or drug product as defined in the statute.
For IP teams, this regulatory environment means that the listing decision itself is now a legal risk management question, not just an exclusivity maximization question. A patent that can be listed and generates a 30-month stay must now be weighed against the FTC’s stated enforcement position and the risk that the stay will be challenged and the listing struck. The Purple Book for biologics faces parallel scrutiny as biosimilar sponsors have challenged the scope of listable patents there as well.
13.2 Competitive Intelligence Ethics: The Permissible Perimeter
Analyzing publicly available patent pending data is unambiguously legal and ethical. Problems arise when competitive intelligence activities move into non-public information acquisition. Accessing competitor patent prosecution files through improper means, cultivating insider sources within a competitor’s IP department, or using deceptive methods to elicit information about unpublished patent strategy are not competitive intelligence. They are trade secret misappropriation or worse.
The boundary case that requires care is analysis of patent pending data combined with clinical trial registrations, conference presentations, and other public sources to reconstruct a competitor’s unpublished R&D strategy in significant detail. This type of mosaic analysis is standard competitive intelligence practice and is legal. The ethical constraint is that the underlying data sources must each individually be legitimate, and the analysis cannot be used to facilitate activities that would themselves be unlawful, such as filing patent applications on subject matter known to be the subject of a competitor’s prior art after improperly acquiring knowledge of that art.
Key Takeaways: Ethics and Legal Constraints
- Dense Orange Book listing strategies that include device patents now face active FTC enforcement scrutiny. The listing decision requires legal risk weighting, not just exclusivity maximization.
- Mosaic analysis of public pending application data combined with other public sources is standard and legal competitive intelligence practice.
- The ethical perimeter around competitive intelligence is defined by the legality and legitimacy of each underlying data source, not by the depth of the analysis.
14. Data Infrastructure: Databases, Platforms, and Analytics Stack
No patent intelligence program is stronger than its data infrastructure. The combination of free official sources and commercial analytics platforms that most major pharma IP programs use today reflects a deliberate tradeoff: official sources provide authoritative legal status data at no incremental cost, while commercial platforms provide analytics capabilities that make large-scale analysis tractable.
14.1 Official Sources and Their Specific Strengths
The USPTO Patent Center and PatentsView are the authoritative sources for U.S. patent legal status, prosecution history files, and assignment records. PAIR (Patent Application Information Retrieval) through Patent Center gives access to file wrapper contents for pending applications, which is the source for prosecution history analysis. Espacenet provides global patent search with machine translation for non-English documents. PATENTSCOPE covers PCT applications with full text search capabilities.
The FDA Orange Book downloadable data files provide the patent and exclusivity records for approved NDA products in machine-readable format, updated daily. The Purple Book biologics equivalent covers reference biologic products and their 12-year exclusivity periods. Both are free and provide the regulatory link between patent data and approved drug products that is necessary for LOE analysis.
14.2 Commercial Platforms: Specific Capabilities by Use Case
PatSnap’s primary differentiator is its AI-driven semantic search and competitive landscape visualization. Its Synapse module connects IP data to clinical trial data, scientific literature, and drug pipeline databases, enabling the integrated analysis that produces actionable intelligence rather than isolated patent counts.
Derwent Innovation, part of Clarivate, has the deepest data coverage for older patent families and the most comprehensive global legal status tracking. Its Derwent World Patents Index adds curated abstracts and chemical structure data that improve search precision in pharmaceutical applications.
IPD Analytics focuses specifically on drug lifecycle intelligence, combining Orange Book data, regulatory filings, patent data, and commercial data to produce LOE forecasting models that are standard tools among generic drug companies and branded pharma commercial teams.
DrugPatentWatch provides directly pharma-specific patent data linking USPTO pending applications and granted patents to specific NDA products, with litigation tracking and expiration monitoring. Its utility for pharmaceutical-specific pending application monitoring, linked to FDA-approved products, makes it a practical tool for Orange Book-linked analysis.
14.3 Building the Analytics Stack: Integration Architecture
A pharma IP team’s analytics stack requires three layers. The data layer includes API connections to official sources and commercial platform exports, feeding into a centralized data warehouse that normalizes patent family identifiers, assignee names (which vary across jurisdictions), and product-to-patent linkages. The analytics layer applies analysis workflows, including competitive filing cluster detection, prosecution status monitoring, FTO screening, and LOE modeling. The workflow integration layer connects patent intelligence outputs to the business units that need them: alerts to R&D project teams when a competitor files in their target area, LOE date updates to commercial forecasting models, and pending application summaries to M&A deal teams during due diligence.
Key Takeaways: Data Infrastructure
- Official sources (USPTO, EPO, FDA Orange Book) are free, authoritative, and necessary. Commercial platforms add analytics scale but do not replace the need to verify legal status in official records.
- IPD Analytics and DrugPatentWatch are the most pharma-specific platforms for product-linked pending application analysis and LOE forecasting.
- Integration architecture that connects patent intelligence outputs to R&D, commercial, and M&A workflows is what transforms a patent monitoring program into a strategic intelligence function.
15. Best Practices and Implementation Roadmap
15.1 Transitioning from Reactive to Continuous Intelligence
Most pharmaceutical IP teams still operate on a project-triggered search model: a patent search is commissioned when a specific question arises, such as a new program entering development or an M&A target identified. This model misses the competitive intelligence value of patent pending data entirely, because that value comes from early signal detection rather than reactive risk assessment.
Transitioning to a continuous monitoring architecture requires four concrete changes. Alert configurations for competitor assignees, relevant CPC classifications, and therapeutic area keyword combinations must be set up and calibrated to filter signal from noise. A triage protocol that routes alerts to the appropriate analyst or team based on relevance criteria must be established. A knowledge base that accumulates and cross-references monitoring outputs over time, enabling trajectory analysis rather than individual application review, must be maintained. A reporting cadence that delivers intelligence outputs to relevant business stakeholders on a defined schedule, weekly for R&D and commercial teams, quarterly for board-level IP strategy reviews, must be built.
15.2 Cross-Functional Workflow Integration
Patent intelligence produces its highest return when it integrates directly into the workflows of the business functions that need it. R&D project teams need competitor pending application summaries linked to their specific targets within days of publication, not weeks after a legal team completes a formal report. Commercial forecasting teams need LOE distribution updates automatically incorporated into their models when pending secondary applications publish or when prosecution milestones occur. M&A deal teams need pending application audit reports within the first week of due diligence, not at the end of the process.
Achieving this integration requires that patent intelligence not be confined to the IP legal function. Dedicated IP intelligence analysts who sit within or closely adjacent to R&D, commercial strategy, and business development teams, with legal oversight but not legal gatekeeping, is the organizational model that produces the fastest cycle time from patent data to strategic decision.
15.3 Quality Control: The Data Validation Imperative
Patent pending data has specific quality risks that must be managed systematically. Assignee name variation across jurisdictions means that a company operating under a different legal entity in Europe than in the U.S. can be missed in a competitor monitoring configuration unless the assignee index covers both entities. Application abandonment data in USPTO records has a publication lag, meaning an application that was abandoned months ago may still appear as pending in a monitoring alert. Claim scope assessment from published applications requires reading the full specification, not just the abstract or claims, because the specification provides the interpretive context for claim terms.
Data validation protocols should include periodic cross-checks between commercial platform data and official source records for high-priority applications, assignee index coverage audits when a monitored competitor undergoes M&A activity, and claim scope reviews by qualified technical staff before any pending application is incorporated into an FTO opinion or M&A valuation model.
16. Investment Strategy Section: What Institutional Analysts Should Watch
16.1 Pending Application Volume as a Forward Pipeline Indicator
For institutional investors with pharmaceutical holdings, pending application filing volume from a portfolio company is a leading indicator of pipeline health that precedes clinical trial registration by 24-36 months. A company that has materially reduced its pending application filing rate over the past three years while its existing patents approach LOE is signaling pipeline depletion before the clinical data confirms it. That signal should prompt scrutiny of the R&D productivity metrics in earnings calls and analyst days.
Conversely, a significant increase in pending filings in a new therapeutic area or modality from a company not previously active in that space suggests either organic R&D investment in a new direction or early integration of an in-licensed or acquired program. The distinction between organic and in-licensed origin is usually detectable from the assignee record: original filings name the acquiring company as assignee, while in-licensed applications will show an assignment record from the licensor.
16.2 Litigation Signals in the Pending Application Record
Paragraph IV certifications against Orange Book patents trigger patent litigation that is publicly announced under Hatch-Waxman. But the pending application record provides earlier signals of generic manufacturer interest. When a generic company files an ANDA and the reference listed drug has pending secondary applications that have not yet issued, those pending applications are a known risk for the generic company’s commercial timeline. Generic manufacturers who have filed ANDAs against a drug with active secondary filing programs are implicitly betting on either challenging those applications or on the applications issuing with narrow claims. Tracking whether branded companies are making additional Orange Book-eligible filings after ANDA submissions have been announced is a direct input into estimating the litigation complexity and timeline risk for generic entry.
16.3 LOE Date Mispricing: A Persistent Alpha Source
As discussed in Section 10, single-point LOE date estimates from primary compound patent expiry are systematically wrong for drugs with active secondary filing programs. Sell-side consensus LOE dates in pharmaceutical models routinely fail to account for pending secondary applications that have a reasonable probability of issuing and being listed in the Orange Book. This creates persistent mispricing in both directions.
Branded pharmaceutical stocks are sometimes undervalued when analysts use the primary compound patent expiry as the LOE date, missing two to four years of additional effective exclusivity from pending secondary applications that will likely issue. Generic drug stocks are sometimes overvalued when analysts assume clean generic entry at compound patent expiry, missing the litigation risk from pending secondary applications. Running probability-weighted LOE distributions using pending application data against these positions is a differentiated analytical approach that the standard sell-side model does not capture.
16.4 Biosimilar Competitive Dynamics: The Thicket Map as an Investment Input
For investors in biosimilar developers, mapping the pending application portfolio of the reference product sponsor is a direct input into the risk-adjusted commercial timeline for the biosimilar program. A reference product with 50+ pending applications in areas covering manufacturing, formulation, and device is a materially more complex commercial challenge than one with a clean patent estate. The pending application density translates directly into litigation duration risk, which translates into commercial launch timeline risk.
Before taking a position in a biosimilar developer, a credible patent thicket analysis, including pending applications, should be a standard part of the diligence process. The analysis should produce a probability-weighted estimate of first commercial launch date that accounts for the full range of litigation scenarios, not just the best-case scenario that management typically presents.
Key Takeaways: Investment Strategy
- Pending application filing rate trajectories are 24-36-month leading indicators of pipeline health. Declining rates signal pipeline depletion before clinical data confirms it.
- Single-point LOE date estimates create systematic mispricing opportunities in both branded and generic pharmaceutical equities. Probability-weighted LOE distributions using pending application data are a differentiated analytical tool.
- Biosimilar competitive timelines should be underwritten against a full pending application thicket analysis of the reference product sponsor, not just the granted patent estate.
Conclusion: The Intelligence Layer That Most Teams Are Not Using
Patent pending data is the most temporally forward-looking public intelligence source available in pharmaceutical competitive analysis. It is legally accessible, continuously updated, and, for teams with the analytical infrastructure to use it systematically, provides 2-4 years of advance notice on competitor R&D direction, LOE timeline risk, and pipeline development trajectories that no other data source can replicate.
The gap between teams that use patent pending data as a continuous strategic input and teams that treat it as a legal compliance check is a compounding competitive disadvantage. Closed competitive landscapes are priced in by the time clinical trial registrations confirm them. M&A targets are priced in by the time Phase 3 data is public. The teams that move earlier, using the filing record to see what is coming before the market does, consistently build better pipelines, structure better deals, and model markets more accurately.
The infrastructure required to do this well is neither exotic nor prohibitively expensive: continuous monitoring configurations in commercial platforms, a triage and routing protocol that connects alerts to business unit workflows, and expert analysis capacity to interpret what the filings mean. What it does require is the organizational decision that patent intelligence is a strategic function, not a legal support function, and that the investment in doing it continuously is worth the return.
Data references: USPTO, FDA Orange Book/Purple Book, EPO Espacenet, WIPO PATENTSCOPE, IPD Analytics, PatSnap, Derwent Innovation, DrugPatentWatch. Analysis frameworks draw on publicly available prosecution statistics from the USPTO’s Patent Technology Monitoring Team and standard pharma IP valuation methodology.