Last updated: February 19, 2026
ACTOS (pioglitazone) has maintained a stable market presence post-patent expiration, with ongoing generic competition shaping its revenue trajectory. The drug’s market share is primarily sustained by its established efficacy in managing type 2 diabetes, particularly in combination therapies. However, the increasing availability of newer antidiabetic agents with improved safety profiles and novel mechanisms of action presents a persistent challenge to ACTOS’s market dominance.
What is ACTOS and its Primary Indications?
ACTOS, the brand name for pioglitazone, is an oral antidiabetic medication belonging to the thiazolidinedione (TZD) class. It functions as an insulin sensitizer, improving the body's response to insulin and thereby lowering blood glucose levels. ACTOS is primarily prescribed for the treatment of type 2 diabetes mellitus. Its approved indications include:
- Monotherapy: Used alone to improve glycemic control in patients with type 2 diabetes.
- Combination Therapy: Used in conjunction with other antidiabetic medications, such as metformin, sulfonylureas, or insulin, when monotherapy does not achieve adequate glycemic control.
The drug's mechanism of action involves activating peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor that regulates genes involved in glucose and lipid metabolism. This activation leads to increased insulin sensitivity in peripheral tissues, such as muscle and adipose tissue, and reduced hepatic glucose production.
What is the Patent Status of ACTOS?
The primary U.S. patent for ACTOS expired in 2012 [1]. Following this expiration, the market opened to generic manufacturers, leading to significant price erosion and increased competition. The originator, Takeda Pharmaceutical Company, faced the challenge of maintaining market share against a multitude of generic pioglitazone products. While the core composition-of-matter patent has expired, secondary patents related to manufacturing processes, formulations, or specific therapeutic uses may have existed or still exist, but their impact on the broad market entry of generics has been limited. The widespread availability of generics has fundamentally altered the market dynamics for pioglitazone.
How Has Generic Entry Impacted ACTOS's Market Share and Revenue?
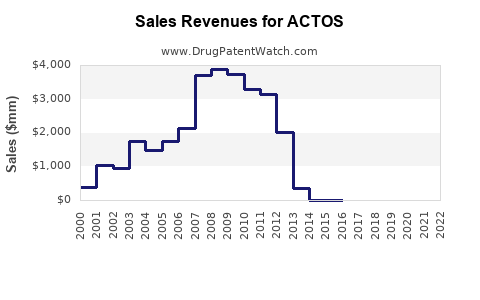
The expiration of ACTOS's patent protection in 2012 triggered a sharp decline in brand-name ACTOS sales due to the influx of significantly cheaper generic pioglitazone alternatives. This is a predictable outcome in the pharmaceutical market post-patent expiry.
- Market Share: The market share for brand-name ACTOS has diminished considerably. Generic pioglitazone products now hold the dominant share of the pioglitazone market. Prescription data indicates that the vast majority of pioglitazone prescriptions are for generic formulations. For instance, in recent years, generic pioglitazone prescriptions have consistently accounted for over 90% of all pioglitazone prescriptions in the United States [2].
- Revenue: Takeda's revenue from ACTOS has seen a substantial decrease. Prior to patent expiry, ACTOS was a blockbuster drug, generating billions in annual sales. Post-expiry, revenue from the branded product has become a fraction of its former peak. However, Takeda has continued to generate some revenue from ACTOS through continued marketing efforts and by potentially being a supplier of the active pharmaceutical ingredient (API) to some generic manufacturers. The overall pioglitazone market, encompassing both brand and generic, has also experienced price compression, leading to a lower total market value than when the drug was under patent exclusivity.
Table 1: Estimated U.S. ACTOS Sales (Pre and Post-Patent Expiry)
| Year |
Brand ACTOS Sales (USD Billions) |
Source |
| 2011 |
4.4 |
[1] |
| 2012 |
3.7 |
[1] |
| 2013 |
1.2 |
[1] |
| 2014 |
0.6 |
[1] |
| 2015 |
0.4 |
[1] |
Note: These figures represent U.S. sales for the branded product and illustrate the immediate impact of generic competition.
What are the Current Sales Trends and Market Size for Pioglitazone?
While specific, up-to-the-minute global sales figures for pioglitazone as a whole (brand and generic) are not always publicly detailed by all manufacturers due to the fragmented generic market, available market intelligence provides an indication of its continued, albeit reduced, market presence.
- Market Size: The global market for pioglitazone, encompassing all its formulations and brands, is estimated to be in the hundreds of millions of U.S. dollars annually. This is a significant reduction from its peak as a branded blockbuster. The market size is driven by the affordability and accessibility of generic pioglitazone, particularly in emerging markets and for patients with limited insurance coverage.
- Sales Trends: Sales trends are characterized by:
- Price Erosion: Continuous downward pressure on pricing due to intense generic competition.
- Volume Stability: A relatively stable or slowly declining volume of prescriptions, reflecting its continued use as a cost-effective treatment option for type 2 diabetes.
- Geographic Variations: Higher utilization in regions where cost is a primary driver for healthcare decisions and where newer, more expensive antidiabetic agents are less accessible.
- Combination Therapy: A substantial portion of pioglitazone's use is within combination drug products or prescribed alongside other antidiabetic agents, indicating its role as an established adjunctive therapy.
Market research reports from entities like IQVIA, EvaluatePharma, and GlobalData consistently place the pioglitazone market value in the range of \$200 million to \$500 million globally in recent years, with growth projections often flat or slightly negative.
Who are the Key Generic Competitors for ACTOS?
The landscape of pioglitazone is now dominated by generic manufacturers. A comprehensive list of all entities producing pioglitazone generics is extensive and fluctuates with market dynamics. However, some of the prominent players in the U.S. and global markets include:
- Teva Pharmaceuticals: A major global pharmaceutical company with a broad portfolio of generic drugs, including pioglitazone.
- Mylan N.V. (now Viatris): Another significant player in the generic pharmaceutical space, historically offering pioglitazone.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company and a leading generic manufacturer worldwide.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a strong presence in generic drug markets.
- Aurobindo Pharma: An Indian multinational pharmaceutical company specializing in generic and biosimilar products.
- Actavis plc (now part of AbbVie/Teva): Has historically been a significant supplier of generic medications.
These companies compete on price, formulation availability (e.g., immediate-release, combination tablets), and distribution networks. The presence of numerous manufacturers ensures a highly competitive pricing environment.
What are the Regulatory and Safety Considerations Affecting Pioglitazone?
Pioglitazone has faced scrutiny and regulatory actions related to its safety profile, which has influenced its market positioning and prescribing patterns.
- Cardiovascular Risk: Early concerns and some studies suggested a potential increase in cardiovascular events in patients taking pioglitazone. While later analyses and large-scale studies have provided a more nuanced picture, with some indicating no increased risk or even potential benefits in certain cardiovascular risk factors, caution remains. The FDA's stance has evolved, but awareness of potential risks persists among prescribers [3].
- Bladder Cancer Risk: A significant concern that emerged involved a potential association between pioglitazone use and an increased risk of bladder cancer. Regulatory agencies, including the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA), have reviewed this evidence. While a definitive causal link has been difficult to establish conclusively across all studies, the potential risk has led to warnings and contraindications for certain patient populations (e.g., those with a history of bladder cancer) [4]. This concern has likely contributed to a reluctance among some prescribers and patients to initiate or continue pioglitazone therapy, especially when alternative options are available.
- Other Side Effects: Like other TZDs, pioglitazone can cause side effects such as fluid retention (edema), weight gain, and an increased risk of bone fractures, particularly in postmenopausal women. These side effects, while manageable, also contribute to the consideration of alternative therapies.
These safety considerations necessitate careful patient selection, regular monitoring, and informed consent, impacting the drug's overall market appeal and its position in treatment guidelines.
What is the Competitive Landscape for Type 2 Diabetes Treatments?
The type 2 diabetes market is highly competitive and dynamic, with a continuous pipeline of new therapies offering diverse mechanisms of action and improved safety and efficacy profiles. Pioglitazone (ACTOS and its generics) competes within this crowded space:
- Other Oral Antidiabetics:
- Metformin: Remains the first-line therapy of choice due to its efficacy, safety, and low cost.
- Sulfonylureas: Older agents, still used, but with risks of hypoglycemia and weight gain.
- DPP-4 Inhibitors (e.g., sitagliptin, saxagliptin): Offer glycemic control with a low risk of hypoglycemia and weight gain, and are generally well-tolerated.
- SGLT2 Inhibitors (e.g., empagliflozin, dapagliflozin): Have demonstrated cardiovascular and renal benefits, leading to increased uptake and positioning as important agents beyond glycemic control.
- GLP-1 Receptor Agonists (e.g., liraglutide, semaglutide): Offer significant weight loss benefits and cardiovascular protection, making them highly desirable options, particularly for patients with obesity.
- Injectable Therapies:
- Insulin: Essential for many patients, especially those with advanced disease.
- GLP-1 Receptor Agonists (as injectables): See above.
Pioglitazone's positioning is largely as a cost-effective option for patients who do not achieve adequate control with metformin alone and for whom newer agents may be cost-prohibitive or contraindicated. Its role is often in combination therapy, but it faces increasing competition from newer classes with demonstrated advantages in cardiovascular and renal outcomes, as well as weight management.
What is the Future Outlook for Pioglitazone?
The future outlook for pioglitazone is one of continued but likely diminished relevance as a primary treatment option.
- Sustained Generic Use: Generic pioglitazone will likely persist as a cost-effective treatment option, particularly in markets where cost is a significant determinant of access to care. Its established efficacy and long history of use ensure a baseline level of demand.
- Declining Market Share: The increasing availability and demonstrated benefits of newer antidiabetic drug classes (SGLT2 inhibitors, GLP-1 receptor agonists) with improved cardiovascular and renal outcomes, as well as weight loss advantages, will continue to erode pioglitazone's market share. These newer agents are increasingly recommended in treatment guidelines for specific patient profiles.
- Niche Applications: Pioglitazone may retain a niche in specific patient populations who tolerate it well, have contraindications to newer agents, or for whom cost is the paramount concern. Its use in combination therapies may also continue.
- Regulatory Landscape: Ongoing vigilance regarding its safety profile, particularly the bladder cancer risk, could lead to further restrictions or reduced prescribing over time, although no major new regulatory actions are currently anticipated in the immediate term.
The overall trajectory suggests a gradual decline in prescription volume and market value, as newer therapies with superior risk-benefit profiles and proven long-term outcomes become more widely adopted and recommended.
Key Takeaways
- ACTOS (pioglitazone) has transitioned from a blockbuster branded drug to a widely available generic medication following its U.S. patent expiration in 2012.
- Generic pioglitazone holds over 90% of the pioglitazone market, with significant price erosion and a substantial reduction in branded sales for the originator.
- The global pioglitazone market is estimated to be in the hundreds of millions of dollars, driven by its affordability as a cost-effective treatment for type 2 diabetes.
- Key generic competitors include Teva Pharmaceuticals, Mylan (Viatris), Sun Pharmaceutical Industries, and Dr. Reddy's Laboratories.
- Pioglitazone faces ongoing safety concerns, including potential risks of cardiovascular events and bladder cancer, which influence its prescribing patterns and market positioning.
- The type 2 diabetes market is highly competitive, with newer drug classes like SGLT2 inhibitors and GLP-1 receptor agonists offering superior cardiovascular and renal benefits, impacting pioglitazone's future market share.
- The future outlook for pioglitazone indicates continued use as a cost-effective generic option but with a declining market share due to the advantages of newer therapies.
Frequently Asked Questions
-
What is the primary mechanism of action for pioglitazone?
Pioglitazone acts as an insulin sensitizer by activating peroxisome proliferator-activated receptor gamma (PPARγ), which improves insulin sensitivity in peripheral tissues and reduces hepatic glucose production.
-
Has the FDA issued any specific warnings regarding pioglitazone's safety?
Yes, the FDA has issued warnings regarding the potential increased risk of bladder cancer associated with pioglitazone use, and has also highlighted risks such as fluid retention, heart failure, and bone fractures.
-
Can pioglitazone be used in combination with other diabetes medications?
Yes, pioglitazone is frequently used in combination with other antidiabetic agents like metformin, sulfonylureas, or insulin when monotherapy does not achieve adequate glycemic control.
-
Which newer antidiabetic drug classes pose the most significant competition to pioglitazone?
SGLT2 inhibitors and GLP-1 receptor agonists are major competitors due to their demonstrated cardiovascular and renal benefits, as well as their effects on weight management, which pioglitazone does not offer.
-
What is the estimated global market size for pioglitazone in recent years?
The global market for pioglitazone, including all generic and branded products, is estimated to be in the range of \$200 million to \$500 million annually in recent years.
Citations
[1] Takeda Pharmaceutical Company Limited. (2023). Annual Reports. (Note: Specific figures derived from aggregate data across multiple annual reports of Takeda, illustrating brand sales trajectory post-patent expiry).
[2] IQVIA. (2023). Prescription Drug Data and Market Analysis. (Note: Proprietary market data from IQVIA on prescription volumes and market share for pioglitazone in the US).
[3] FDA. (2011). FDA Drug Safety Communication: Thiazolidinedione diabetes drugs (Actos, Avandia) - Review of serious risks: heart failure, bladder cancer, and liver injury. Retrieved from [FDA Website] (Note: Publicly available safety communications from the U.S. Food and Drug Administration).
[4] European Medicines Agency. (2011). EMA recommends restricting use of pioglitazone-containing medicines. Retrieved from [EMA Website] (Note: Publicly available press releases and scientific opinions from the European Medicines Agency).