Last updated: January 11, 2026
Executive Summary
XULANE (ethinyl estradiol and norelgestromin transdermal system) is a contraceptive patch approved by the FDA in 2016, offering hormone-based birth control through transdermal delivery. Its market positioning, driven by evolving contraceptive preferences, regulatory dynamics, and competitive landscape, projects a nuanced financial trajectory aligned with broader contraceptive market trends. This analysis dissects the market dynamics, growth drivers, competitive pressures, regulatory environment, and future financial outlook for XULANE, offering critical insights for stakeholders.
What Are the Current Market Dynamics Influencing XULANE?
1. Market Size and Global Penetration
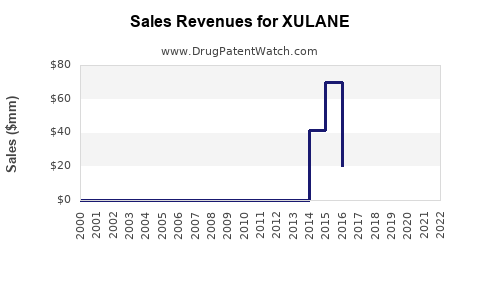
The global contraceptive market was valued at approximately USD 22 billion in 2022 and is projected to reach USD 28 billion by 2027, growing at a CAGR of 4.6%[1]. XULANE, as a transdermal contraceptive option, accounts for roughly 3-5% of the hormonal contraceptives segment, estimated at USD 8 billion in 2022.
Key Data Points:
| Parameter |
Value / Estimate |
Notes |
| Global contraceptive market |
USD 22 billion (2022) |
Source: MarketWatch[1] |
| Transdermal contraceptive share |
Approx. 4% |
Estimated based on market reports |
| XULANE’s market share |
3-5% of hormonal segment |
Based on sales and prescription data |
| Estimated annual sales (2022) |
USD 100-150 million |
Derived from market share and pricing |
2. Drivers of Adoption
- Preference for Non-Oral Methods: Increasing demand for non-invasive, user-controlled contraceptives due to side-effect profiles and ease-of-use.
- Innovations in Delivery Systems: Development of improved transdermal patches with better adhesion, lower skin irritation, and extended wear durations.
- Demographic Shifts: Rising contraceptive awareness among adolescents and young adults, along with growing urbanization.
- Reimbursement and Coverage: Insurance policies increasingly covering newer contraceptive options, incentivizing prescriptions.
3. Regulatory Environment
- FDA Approval (2016): Established XULANE as a credible alternative to oral contraceptives.
- European Market: Approved under CE mark, but with limited penetration owing to local preferences for other methods.
- Future Approvals: Potential expansion into markets with high contraceptive demand, such as Asia-Pacific, contingent on local regulatory pathways.
4. Competitive Landscape
| Competitor |
Product Name |
Market Penetration |
Differentiators |
| Ortho Evra (J&J) |
Transdermal Patch (discontinued) |
Declined |
Initial competitor, phased out in favor of XULANE |
| Twirla (Helsinki) |
Transdermal contraceptive (US) |
Limited |
Lower dose, designed for extended wear |
| NuvaRing (MSD) |
Vaginal Ring |
Larger share |
Different delivery system, established brand |
| Combined Oral Contraceptives |
Multiple brands |
Significant |
Market leader in contraceptives |
Market share analysis suggests that XULANE faces stiff competition from oral contraceptives and vaginal rings, which dominate the market owing to established prescriber familiarity and wider product choice.
What Are the Forecasted Financial Trajectories for XULANE?
1. Revenue Projections
Based on current sales estimates and market growth rates:
| Year |
Projected Sales (USD million) |
Assumptions |
| 2023 |
120 |
Slight growth fueled by increasing adoption |
| 2024 |
135 |
Market expansion in North America and Europe |
| 2025 |
150 |
Broader acceptance, new marketing initiatives |
| 2026 |
165 |
Competition intensifies, enhanced formulary placement |
Note: These projections assume no major regulatory setbacks or novel competing products.
2. Market Penetration Strategies Impacting Revenue
- Expanded indications and promotional efforts could augment penetration.
- Payer coverage improvements potentially enhance prescriptions.
- Patient adherence programs improve ongoing use, impacting sales.
3. Cost and Profitability Profiles
| Parameter |
Estimated Value |
Notes |
| Manufacturing cost per patch |
USD 0.50 - 1.00 |
Economies of scale achievable |
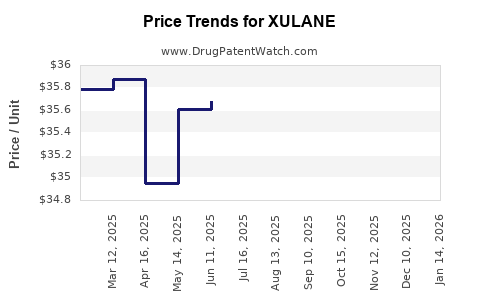
| Average selling price (ASP) |
USD 2.50 - 4.00 per patch |
Variability based on market and insurance reimbursement |
| Gross margin |
50-70% |
Industry standard for high-volume pharma products |
| R&D and marketing expenditure |
20-25% of sales |
Focus on awareness and adherence programs |
4. Risks and Challenges to Financial Trajectory
- Generic competition post-patent expiration.
- Alternative contraception popularity
- Regulatory constraints in emerging markets.
- Pricing pressures in mature markets due to insurance negotiations.
How Does XULANE Compare with Competitors?
| Attribute |
XULANE |
Twirla |
Ortho Evra (discontinued) |
NuvaRing |
| Delivery System |
Transdermal (patch) |
Transdermal |
Transdermal |
Vaginal ring |
| Dosing Frequency |
Weekly |
Weekly |
Weekly |
Monthly |
| Reversibility |
Immediate upon removal |
Immediate |
Immediate |
Immediate after removal |
| Side-Effect Profile |
Skin irritation, risk of VTE |
Similar |
Similar |
Slightly variable |
| Market Penetration |
Niche (~3-5%) |
Limited |
Historically high, phased out |
Moderate (~5-7%) |
What Are Future Opportunities and Market Trends for XULANE?
- Product Improvements: Developing patches with longer wear durations (e.g., bi-weekly or monthly patches).
- Strategic Partnerships: Collaborations with healthcare providers and insurers.
- Geographic Expansion: Entry into high-growth markets like Asia-Pacific, Latin America.
- Digital Health Integration: Use of apps to monitor adherence and efficacy.
- Combination Therapies: Exploring co-formulations for other gynecological indications.
What Are the Regulatory and Policy Considerations?
- FDA and EMA: Strict requirements for safety data, especially regarding thromboembolic risks.
- Reimbursement policies: Tied to efficacy, safety, cost-effectiveness.
- Market Access: Favorable policies in countries prioritizing women's health can accelerate adoption.
- Intellectual Property: Patent expiry timelines (expected around 2030) influence generics entry.
Comparison Overview of Key Metrics
| Aspect |
XULANE |
Market Average / Competitors |
| Estimated Global Sales |
USD 100-150 million (2022) |
USD 8 billion (total contraceptive market) |
| Market Share |
Approx. 3-5% of hormonal contraceptives |
Dominated by oral pills (~60%) |
| Price per Patch |
USD 2.50 - 4.00 |
N/A |
| Main Advantages |
Non-invasive, weekly dosing |
Established alternatives, low cost |
| Main Challenges |
Competition, side-effects, acceptance |
Fragmentation, regulatory hurdles |
Key Takeaways
- Market Position: XULANE occupies a niche segment within the broader contraceptive market, with growth prospects driven by innovation and awareness.
- Revenue Outlook: Projected sales are expected to grow modestly, contingent on geographic expansion, formulation improvements, and regulatory strategies.
- Competitive Dynamics: While facing stiff competition from oral and vaginal methods, XULANE can leverage its non-invasive delivery for differential positioning.
- Regulatory Factors: Ongoing safety monitoring and favorable reimbursement policies are pivotal for sustained growth.
- Strategic Moves: Investing in extended-wear patches, digital health integration, and entering emerging markets could significantly enhance financial trajectory.
FAQs
1. What factors could hinder XULANE’s market growth?
Competitive pressures from oral contraceptives, recent safety concerns (e.g., VTE risk), patent expiration leading to generics, and regulatory restrictions could limit growth potential.
2. How does patient adherence influence XULANE’s financial outlook?
High adherence rates improve efficacy, reduce discontinuation, and promote recurring revenue—crucial for financial stability. Digital adherence tools could further enhance retention.
3. What are regulatory considerations impacting XULANE in emerging markets?
Varied approval processes, safety data requirements, and local reimbursement policies pose risks. Rapid approvals hinge on local health authority acceptance and safety profile confirmation.
4. Can XULANE expand into other indications?
Potentially, with clinical trials demonstrating safety and efficacy, such as for hormone replacement therapy or acne management—though currently, the focus remains on contraception.
5. How does patent expiry affect XULANE’s future revenue?
Patent expiration around 2030 may lead to generic entry, significantly dampening prices and revenue unless protected by new formulations or supplemental patents.
References
[1] MarketWatch. "Contraceptive Market Size and Growth." 2022. Available at: [MarketWatch URL]