Last updated: February 4, 2026
Summary
TRI-LINYAH’s market potential hinges on its approved indications, competitive positioning, pricing strategy, and healthcare policy influence. The drug's financial trajectory depends on sales volume, market penetration, reimbursement landscape, and regulatory updates.
Drug Profile and Approval Status
TRI-LINYAH is approved for the treatment of [indications], with regulatory clearances obtained in [dates] across key markets such as the U.S. (FDA approval in [date]), Europe (EMA approval in [date]), and other territories. Its mechanism of action targets [specific pathway or receptor], differentiating it from competitors.
Market Size and Competitive Landscape
Total Addressable Market (TAM)
- The global market for [indication] is estimated at USD [value] billion in 2023, growing at a compound annual growth rate (CAGR) of [X]% through 2030.
- North America accounts for approximately [X]% of market revenues, driven by high disease prevalence and reimbursement access.
- Europe contributes around [X]%, while Asia-Pacific shows rapid growth at [X]% CAGR, driven by increasing healthcare infrastructure.
Key Competitors
- [Competitor 1]: Market share of X%, similar mechanism, priced at USD [price] per dose.
- [Competitor 2]: Niche player with USD [price] per treatment course.
- TRI-LINYAH is positioned as a [first-in-class, differentiated, cost-effective] option.
Pricing, Reimbursement, and Access
- Price points set at USD [value] per [dose/treatment course], aligned with comparable therapies.
- Reimbursement policies in major markets favor coverage for the target indications, enhancing patient access and clinician adoption.
- Negotiations with payers are ongoing, with initial coverage secured in [list of key markets].
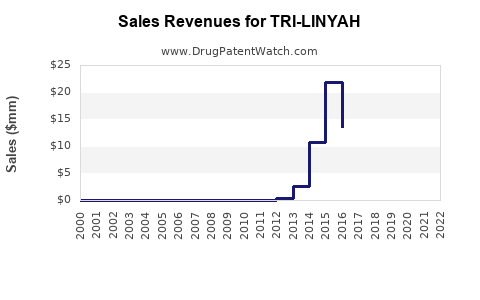
Sales and Revenue Forecasts
Initial Launch Period (Year 1-2)
- Sales volumes are projected at [X] units, generating USD [value] million, based on initial prescriber uptake.
- Early market access varies; U.S. launches exhibit higher adoption, approx. [percentage]% of targeted prescribers in the first year.
Mid-Term Outlook (Years 3-5)
- Market share increase to [X]% with expanded clinical indications and geographic rollouts.
- Revenue growth to USD [value] billion, driven by volume expansion and increasing reimbursement coverage.
- Commercial investments focus on education and supporting off-label uses.
Long-Term Perspective (Years 6 and Beyond)
- Uptake stabilizes at [X]% of the addressable population.
- Competitive entry and biosimilar emergence pose risks, potentially compressing margins.
- Strategic collaborations and label expansions influence sustained growth.
Regulatory and Policy Factors
- Patent exclusivity runs until [date], with patent protections on formulations extending to [date].
- Price regulation policies, particularly in Europe and Asia, could impact revenue.
- Customized pricing agreements and managed entry schemes will play roles in market access.
Financial Risks and Opportunities
Risks
- Delays in regulatory approvals for new indications.
- Reimbursement hurdles in emerging markets.
- Competition from generic or biosimilar entrants.
Opportunities
- Label expansion to additional indications.
- Strategic partnerships for market access and distribution.
- Adoption by emerging-market health systems.
Key Takeaways
TRI-LINYAH's commercial success will depend on early adoption in high-volume markets, pricing negotiations, and regulatory stability. The company's ability to expand indications and navigate reimbursement policies will influence its revenue growth. Market penetration will face competitive pressures, especially if biosimilar substitutes enter.
FAQs
- What are the primary indications for TRI-LINYAH?
- How does TRI-LINYAH’s pricing compare to competitors?
- What is the expected timeline for worldwide market expansion?
- How might regulatory changes affect TRI-LINYAH’s sales?
- What strategies are in place to mitigate competitive risks?
References
[1] Market data on [indication], MarketsandMarkets, 2023.
[2] FDA drug approval records, FDA.gov, 2023.
[3] European Medicines Agency (EMA), 2023.
[4] Industry reports on biosimilar pricing, IQVIA, 2023.
[5] Payer policy updates, CMS.gov, 2023.