TIVICAY PD Drug Patent Profile
✉ Email this page to a colleague
When do Tivicay Pd patents expire, and when can generic versions of Tivicay Pd launch?
Tivicay Pd is a drug marketed by Viiv Hlthcare and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and fifty-seven patent family members in thirty-five countries.
The generic ingredient in TIVICAY PD is dolutegravir sodium. There are seventeen drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the dolutegravir sodium profile page.
DrugPatentWatch® Generic Entry Outlook for Tivicay Pd
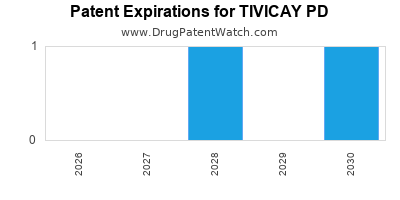
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 8, 2030. This may change due to patent challenges or generic licensing.
There have been twenty-four patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are eight tentative approvals for the generic drug (dolutegravir sodium), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TIVICAY PD?
- What are the global sales for TIVICAY PD?
- What is Average Wholesale Price for TIVICAY PD?
Summary for TIVICAY PD
| International Patents: | 157 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 45 |
| Clinical Trials: | 32 |
| Patent Applications: | 275 |
| Drug Prices: | Drug price information for TIVICAY PD |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TIVICAY PD |
| What excipients (inactive ingredients) are in TIVICAY PD? | TIVICAY PD excipients list |
| DailyMed Link: | TIVICAY PD at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TIVICAY PD
Generic Entry Date for TIVICAY PD*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET, FOR SUSPENSION;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TIVICAY PD
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Humanis Saglık Anonim Sirketi | PHASE1 |
| Chelsea and Westminster NHS Foundation Trust | Phase 1 |
| Chelsea and Westminster NHS Foundation Trust | Phase 3 |
Pharmacology for TIVICAY PD
Paragraph IV (Patent) Challenges for TIVICAY PD
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TIVICAY PD | Tablets for Suspension | dolutegravir sodium | 5 mg | 213983 | 1 | 2021-07-21 |
US Patents and Regulatory Information for TIVICAY PD
TIVICAY PD is protected by two US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TIVICAY PD is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | TIVICAY PD | dolutegravir sodium | TABLET, FOR SUSPENSION;ORAL | 213983-001 | Jun 12, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Viiv Hlthcare | TIVICAY PD | dolutegravir sodium | TABLET, FOR SUSPENSION;ORAL | 213983-001 | Jun 12, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for TIVICAY PD
When does loss-of-exclusivity occur for TIVICAY PD?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09325128
Estimated Expiration: ⤷ Start Trial
Patent: 14277831
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0923217
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 44019
Estimated Expiration: ⤷ Start Trial
Patent: 55957
Estimated Expiration: ⤷ Start Trial
China
Patent: 2245182
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 76080
Estimated Expiration: ⤷ Start Trial
Patent: 10603
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 43626
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 86478
Estimated Expiration: ⤷ Start Trial
Patent: 48595
Estimated Expiration: ⤷ Start Trial
Patent: 30891
Estimated Expiration: ⤷ Start Trial
Patent: 12131791
Patent: SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND THEIR INTERMEDIATES
Estimated Expiration: ⤷ Start Trial
Patent: 12511573
Estimated Expiration: ⤷ Start Trial
Patent: 16041727
Patent: カルバモイルピリドンHIVインテグラーゼ阻害剤及びそれらの中間体の合成 (SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND THEIR INTERMEDIATES)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 1942
Patent: SINTESIS DE INHIBIDORES DE INTEGRASA DE VIH DE CARBAMOIL-PIRIDONA E INTERMEDIARIOS. (SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES.)
Estimated Expiration: ⤷ Start Trial
Patent: 3683
Patent: SINTESIS DE INHIBIDORES DE INTEGRASA DE VIH DE CARBAMOIL-PIRIDONA E INTERMEDIARIOS. (SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES)
Estimated Expiration: ⤷ Start Trial
Patent: 11006241
Patent: SINTESIS DE INHIBIDORES DE INTEGRASA DE VIH DE CARBAMOIL-PIRIDONA E INTERMEDIARIOS. (SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES.)
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 27451
Patent: СИНТЕЗ КАРБАМОИЛПИРИДОНОВЫХ ИНГИБИТОРОВ ИНТЕГРАЗЫ ВИЧ И ПРОМЕЖУТОЧНЫХ СОЕДИНЕНИЙ (SYNTHESIS OF CARBAMOYL PYRIDONE INHIBITORS OF HIV INTEGRASE AND INTERMEDIATE COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 38923
Patent: Синтез карбамоилпиридоновых ингибиторов интегразы ВИЧ и промежуточных соединений (SYNTHESIS OF CARBAMOIL-PYRIDONE INHIBITORS OF HIV INTEGRASE AND INTERMEDIATE COMPOUNDS)
Estimated Expiration: ⤷ Start Trial
Patent: 11121785
Patent: СИНТЕЗ КАРБАМОИЛПИРИДОНОВЫХ ИНГИБИТОРОВ ИНТЕГРАЗЫ ВИЧ И ПРОМЕЖУТОЧНЫХ СОЕДИНЕНИЙ
Estimated Expiration: ⤷ Start Trial
Patent: 13153004
Patent: СИНТЕЗ КАРБАМОИЛПИРИДОНОВЫХ ИНГИБИТОРОВ ИНТЕГРАЗЫ ВИЧ И ПРОМЕЖУТОЧНЫХ СОЕДИНЕНИЙ
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 1308
Patent: SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1733625
Estimated Expiration: ⤷ Start Trial
Patent: 1847887
Estimated Expiration: ⤷ Start Trial
Patent: 110094336
Patent: SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES
Estimated Expiration: ⤷ Start Trial
Patent: 170038116
Patent: 카르바모일피리돈 HIV 인테그라제 억제제 및 중간체의 합성 (SYNTHESIS OF CARBAMOYLPYRIDONE HIV INTEGRASE INHIBITORS AND INTERMEDIATES)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 41765
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1030010
Patent: Synthesis of carbamoylpyridone HIV integrase inhibitors and intermediates
Estimated Expiration: ⤷ Start Trial
Patent: 83947
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TIVICAY PD around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Mexico | 2008005137 | DERIVADO DE CARBAMOILPIRIDONA POLICICLICA QUE TIENE ACTIVIDAD INHIBIDORA EN VIH INTEGRASA. (POLYCYCLIC CARBAMOYLPYRIDONE DERIVATIVE HAVING INHIBITORY ACTIVITY ON HIV INTEGRASE.) | ⤷ Start Trial |
| European Patent Office | 2527007 | Dérivé de carbamoylpyridone polycyclique doté d'une activité inhibitrice de l'intégrase du VIH (Polycyclic carbamoylpyridone derivative having HIV integrase inhibitory activity) | ⤷ Start Trial |
| Eurasian Patent Organization | 014162 | ПОЛИЦИКЛИЧЕСКОЕ КАРБАМОИЛПИРИДОНОВОЕ ПРОИЗВОДНОЕ, ОБЛАДАЮЩЕЕ ИНГИБИТОРНОЙ АКТИВНОСТЬЮ В ОТНОШЕНИИ ИНТЕГРАЗЫ ВИЧ (POLYCYCLIC CARBAMOYLPYRIDONE DERIVATIVE HAVING HIV INTEGRASE INHIBITORY ACTIVITY) | ⤷ Start Trial |
| Netherlands | 301109 | ⤷ Start Trial | |
| Slovenia | 3372281 | ⤷ Start Trial | |
| Lithuania | 3284520 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TIVICAY PD
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2465580 | C 2021 017 | Romania | ⤷ Start Trial | PRODUCT NAME: CABOTEGRAVIR; NATIONAL AUTHORISATION NUMBER: EU/1/20/1481; DATE OF NATIONAL AUTHORISATION: 20201217; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1481; DATE OF FIRST AUTHORISATION IN EEA: 20201217 |
| 2465580 | 122021000021 | Germany | ⤷ Start Trial | PRODUCT NAME: CABOTEGRAVIR ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ ODER SOLVAT DAVON; REGISTRATION NO/DATE: EU/1/20/1481 20201217 |
| 2465580 | 21C1023 | France | ⤷ Start Trial | PRODUCT NAME: CABOTEGRAVIR OU L'UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; NAT. REGISTRATION NO/DATE: EU/1/20/1481 20201221; FIRST REGISTRATION: - EU/1/20/1481 20201221 |
| 1874117 | PA2014021 | Lithuania | ⤷ Start Trial | PRODUCT NAME: DOLUTEGRAVIRUM NATRICUM; REGISTRATION NO/DATE: EU/1/13/892/001, 2014 01 16 EU/1/13/892/002 20140116 |
| 3494972 | PA2024516,C3494972 | Lithuania | ⤷ Start Trial | PRODUCT NAME: DOLUTEGRAVIRAS ARBA FARMACINIU POZIURIU PRIIMTINA DRUSKA, ISKAITANT DOLUTEGRAVIRO NATRIO DRUSKA IR LAMIVUDINA; REGISTRATION NO/DATE: EU/1/19/1370 20190701 |
| 2465580 | 2021C/519 | Belgium | ⤷ Start Trial | PRODUCT NAME: VOCABRIA-CABOTEGRAVIR; AUTHORISATION NUMBER AND DATE: EU/1/20/1481 20201221 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for TIVICAY PD
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.