Last updated: February 5, 2026
What Are the Market Dynamics for Sulindac?
Sulindac is a non-steroidal anti-inflammatory drug (NSAID) approved for the relief of pain, swelling, and inflammation associated with osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. It is also studied for cancer chemopreventive properties. Market dynamics for sulindac are shaped by several key factors:
Market Size and Growth Drivers
- Global NSAID Market: Valued at approximately $44 billion in 2022, with an expected compound annual growth rate (CAGR) of 4-5% through 2030 [1].
- Niche Positioning: Sulindac holds a small share of the NSAID sector, primarily used in specific indications such as colorectal cancer prevention and off-label uses.
- Indication Expansion: Research into additional uses, especially chemoprevention, could broaden its application scope.
- Generic Penetration: Sulindac was off-patent in many regions by the late 2000s, leading to increased generic competition and lower prices.
Competitive Landscape
- Major Alternatives: Other NSAIDs like ibuprofen, naproxen, diclofenac, and celecoxib dominate the market.
- Chemical Similarity: Share common mechanisms with other NSAIDs but differ in side effect profiles, influencing physician prescribing habits.
- Research Activity: Limited ongoing clinical trials, which constrains potential growth unless new indications are established.
Regulatory and Clinical Use Trends
- Regulatory Status: Approved by FDA, EMA, and other agencies. No recent major approvals or removals.
- Safety Profile: Concerns over cardiovascular risks typical of NSAIDs influence prescribing behaviors.
- Guideline Recommendations: Typically recommended for short-term, low-dose use; rarely used long-term due to safety.
Market Limitations and Opportunities
- Safety Concerns: Cardiovascular and gastrointestinal risks reduce long-term use.
- Potential for Repurposing: Chemopreventive studies in colorectal cancer show promise but are not yet reflected in significant market uptake.
- Patient Compliance: Side effect profile limits adherence, affecting market penetration.
What Is the Financial Trajectory for Sulindac?
Revenue and Sales Trends
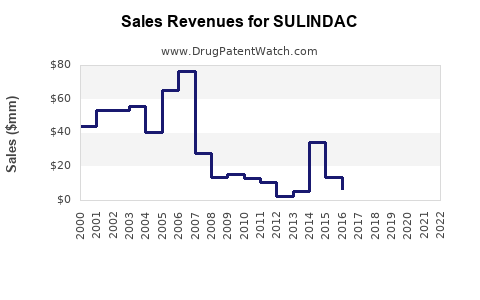
- Historical Revenues: As a generic drug, sales are mainly from established markets; precise figures are not publicly available but are estimated to be in the low hundreds of millions globally.
- Market Share Dynamics: Likely declining slightly due to generics and competition from newer NSAIDs with better safety profiles.
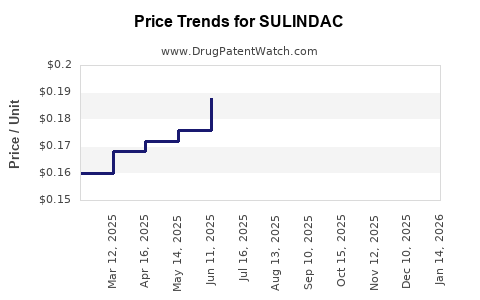
- Pricing: Lower than branded NSAIDs; price erosion driven by generic competition.
R&D and Clinical Investment
- Development Expenditure: Limited R&D activity; current investments primarily focus on assembling chemopreventive data.
- Pipeline Status: No significant ongoing clinical trials or future clinical development plans disclosed publicly.
Market Entry and Expansion Potential
- Obstacles: Safety concerns and existing competition limit growth prospects.
- Opportunities: Expanded indications through chemopreventive research could lead to niche market development.
- Regulatory Modifications: Potential approval for new indications could impact revenue positively, but no major filings are anticipated in the short term.
Financial Risks and Considerations
- Market Saturation: Extensive generic penetration reduces profit margins.
- Regulatory Risks: Changes in safety labeling or restrictions could diminish use.
- Competitor Innovation: Introduction of improved NSAIDs or alternative therapies may further erode market share.
Summary of Market and Financial Outlook
| Aspect |
Status |
Implication |
| Market Size |
USD 44 billion (2022), slow growth |
Limited growth driven by competition and safety concerns |
| Revenue Trends |
Low hundreds of millions globally, declining share due to generics |
Marginal revenue, constrained market expansion |
| Clinical Trials |
Limited recent activity; current focus on chemoprevention evidence |
Potential niche growth if evidence supports indication expansion |
| Regulatory Status |
Approved; no recent major regulatory changes |
Stability in approvals but limited prospects for new indications |
| Competition |
Dominated by other NSAIDs with improved safety profiles |
Challenges in expanding market share |
Key Takeaways
- Sulindac remains an established, off-patent NSAID with a stable, limited market.
- Its prospects for growth depend on successful validation for chemoprevention or new indications.
- Safety profiles impose constraints on long-term use, impacting sales.
- Extensive generic competition limits pricing power and revenue potential.
- Future market expansion is primarily tied to research outcomes rather than current clinical application.
FAQs
1. Is there any significant recent research that could change sulindac's market outlook?
No. Current research focuses mainly on chemopreventive potential, with no large-scale trials or regulatory filings anticipated in the near term.
2. How does the safety profile affect sulindac's commercial viability?
The associated cardiovascular and gastrointestinal risks limit long-term prescribing, reducing its appeal against newer NSAIDs with better safety profiles.
3. Can sulindac be repositioned for other indications?
Potential exists in chemopreventive applications, particularly colorectal cancer. However, regulatory approval and clinical validation are prerequisites.
4. What impact does generic competition have on sulindac's revenues?
It suppresses prices, reduces profit margins, and limits growth, similar to other off-patent NSAIDs.
5. Are there any regulatory hurdles to expanding sulindac’s approved uses?
Yes. Safety concerns and the need for clinical trial data pose significant barriers to label expansions.
Citations
[1] Grand View Research, "NSAID Market Size, Share & Trends," 2022.