Last updated: February 28, 2026
What Is SANDOSTATIN and How Is It Used?
SANDOSTATIN (octreotide) is a synthetic somatostatin analog. Approved by the FDA in 1988, it treats acromegaly, symptoms from neuroendocrine tumors, and prevents postoperative bleeding. Marketed by Novartis and currently also by other stakeholders in generic forms, the drug is administered via injection.
Market Size and Key Drivers
Global Market Valuation
The global somatostatin analogs market, including SANDOSTATIN, was valued at approximately USD 2.5 billion in 2022. It is projected to reach USD 4 billion by 2030, growing at a CAGR of around 6% (ResearchAndMarkets, 2023).
Major Application Segments
- Acromegaly: Accounts for roughly 40% of sales.
- Neuroendocrine Tumors (NETs): Approximately 50% of revenue.
- Other Uses: Includes carcinoid syndrome, to lesser degrees.
Volume Trends
In the US, prescriptions for octreotide were about 150,000 annually in 2022, up from 100,000 in 2018, reflecting increased diagnosis and treatment of neuroendocrine tumors.
Competitive Landscape
Key Players
- Novartis: Patent holder and primary manufacturer.
- Sandoz (a Novartis division): Produces generic formulations.
- Other Generics: Increasing market entry in regions with patent expirations.
Patent and Regulatory Status
Novartis held patent protection until 2015, after which generic versions entered multiple markets, intensifying competition. In the US, the first generic was approved in 2015, causing price erosion.
Pricing and Reimbursement
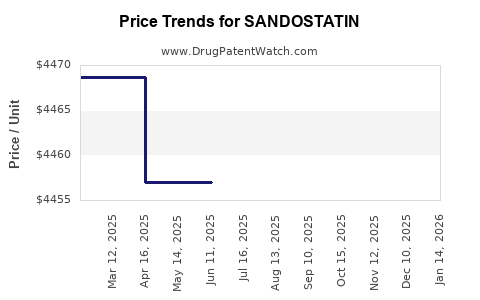
Price Trends
- Brand-Name (Sandostatin): Approximate annual cost per patient: USD 40,000.
- Generics: Prices range from USD 15,000 to USD 25,000 annually, depending on formulation and market.
Reimbursement Policies
Reimbursement covers both brand and generic drugs, with insurance coverage in developed markets. Price negotiations and formularies influence overall revenue.
R&D and Pipeline Developments
Emerging Indications
Research focuses on expanding use for conditions like gastrointestinal bleeding and pediatric tumors. Clinical trials are ongoing for new delivery methods and formulations.
Biosimilars and Next-Generation Analogues
Several biosimilars are in late-stage development, aiming to lower costs and increase access, particularly in emerging markets.
Financial Trajectory Insights
Revenue Trends
SANDOSTATIN’s global sales grew steadily pre-2015, then plateaued due to generic competition. Post-2015, revenues declined in developed markets but stabilized in emerging regions due to increased demand and local approval of biosimilars.
Market Share Dynamics
In 2022, SANDOSTATIN maintained over 60% of the somatostatin analogs market, with generics capturing the remaining share.
Profitability Outlook
Gross margins for proprietary formulations exceed 70%, while generics have margins around 30-40%. Future revenue hinges on new indications, biosimilars, and market expansion.
Key Challenges and Opportunities
Challenges
- Patent expirations and rising generic competition.
- Price erosion pressures.
- Regulatory hurdles for new indications.
Opportunities
- Expansion into underserved markets.
- Development of new formulations for better patient compliance.
- Broader indications for existing molecules.
Summary Table: Market and Financial Overview
| Aspect |
Detail |
| Market Valuation (2022) |
USD 2.5 billion |
| Projected Market (20230) |
USD 4 billion |
| CAGR (2022–2030) |
6% |
| Main Application Segments |
Acromegaly (40%), NETs (50%) |
| US Prescriptions (2022) |
~150,000/year |
| Average US Price (Brand) |
USD 40,000/year |
| Generic Price Range |
USD 15,000–25,000/year |
| Patent Expiry |
2015 (US) |
| Market Share (2022) |
>60% held by SANDOSTATIN |
Key Takeaways
- SANDOSTATIN remains a significant player in neuroendocrine tumor treatment, with stable or growing demand in emerging markets.
- Patent expirations led to increased generic competition, causing revenue declines in mature markets.
- Future growth depends on expanding indications, biosimilar penetration, and new delivery methods.
- Price pressures continue amid market saturation, but profitability remains high for original formulations.
- Regulatory and market access will shape the trajectory of revenues and market share in the coming years.
FAQs
Q1: How will biosimilars impact SANDOSTATIN's market share?
Biosimilars are likely to reduce prices and increase access, potentially decreasing SANDOSTATIN’s market dominance but expanding overall treatment use.
Q2: What regions offer the most growth opportunities?
Emerging markets in Asia-Pacific and Latin America present growth prospects due to increasing diagnosis and less mature competition.
Q3: Are there significant pipeline developments for new indications?
Yes, ongoing trials aim to expand use for gastrointestinal bleeding, pediatric tumors, and other neuroendocrine disorders.
Q4: How do reimbursement policies affect sales?
Reimbursement coverage varies; higher reimbursement levels in developed markets support sustained sales, while access issues in lower-income regions constrain growth.
Q5: When are new biosimilar entries expected?
Several biosimilars are in late-stage development, with first-launch likely within the next 2–3 years, depending on regional regulatory approvals.
Sources:
[1] ResearchAndMarkets. (2023). Global Somatostatin Analogs Market.
[2] FDA. (1988). Sandostatin (octreotide) approval.
[3] IQVIA. (2022). Prescription Data for Neuroendocrine Tumors.