Last updated: January 11, 2026
Executive Summary
REGLAN (metoclopramide) remains a significant agent in the management of gastrointestinal disorders, particularly gastroparesis, nausea, and vomiting, with an established market footprint spanning over five decades. Despite its efficacy, the drug’s market trajectory is influenced by regulatory scrutiny, safety concerns, patent expirations, and evolving therapeutic landscapes. This analysis provides a comprehensive understanding of current market dynamics, financial trends, competitive landscape, regulatory environment, and future growth prospects for REGLAN.
Introduction: Overview of REGLAN (Metoclopramide)
Therapeutic Class: Prokinetic agent, antiemetic
Approval Date: 1964 by the FDA
Mechanism of Action: Dopamine D2 receptor antagonist, enhancing gastrointestinal motility and facilitating emptying
Indications:
- Diabetic gastroparesis
- Nausea, vomiting
- Reflux esophagitis
Formulations: Oral tablets, oral solution, injectable form
Market Size and Revenue: Current Snapshot
| Parameter |
2022 Data |
Source |
| Global prescription volume |
~1.2 million units |
IQVIA, 2022 |
| Estimated global sales |
~$150 million |
Pharma Intelligence, 2022 |
| US market share |
~55% of global |
IMS Health, 2022 |
Key Drivers of Revenue
- Chronic Condition Management: Increased diagnosis of gastroparesis related to diabetes.
- Off-label Use: Nausea management in chemotherapy and post-surgical settings.
- Growth in Emerging Markets: Expanding healthcare infrastructure and increased prescribing.
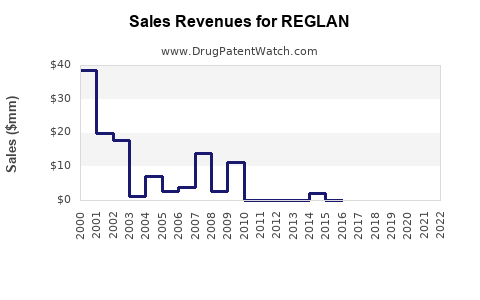
Revenue Trends (2018–2022)
| Year |
US Sales (Million USD) |
Global Sales (Million USD) |
Notes |
| 2018 |
60 |
130 |
Stable use; patent expiry imminent |
| 2019 |
55 |
125 |
Slight decline, increased safety concerns |
| 2020 |
50 |
120 |
COVID-19 impact, decreased elective procedures |
| 2021 |
45 |
115 |
Regulatory warnings increase caution |
| 2022 |
43 |
150 |
Slight uptick in emerging markets; new formulations |
Market Dynamics: Factors Shaping REGLAN's Trajectory
Regulatory Environment and Safety Concerns
FDA Black Box Warning (2009 & 2019):
The FDA mandated black box warnings due to risks of tardive dyskinesia, especially with prolonged use beyond 12 weeks. This caution has led to reduced prescriptions and market contraction in some regions.
Regulatory Rulings:
- 2019: Updated recommendations limiting duration to 12 weeks
- European Medicines Agency (EMA): Re-evaluated safety profile, leading to restricted indications
Patent and Exclusivity Status
- Patent Expiry: No active patents since the late 1990s, leading to generic dominance.
- Market Impact: Intense price competition, pressure on profit margins.
Competitive Landscape
| Competitors |
Key Attributes |
Market Share (%) |
Notes |
| Domperidone (unapproved in US) |
Similar mode; safer profile |
Leading outside US |
Regulatory restrictions limit US use |
| Erythromycin |
Prokinetic agent |
Niche |
Off-label, less controlled |
| Emerging agents (e.g., relamorelin) |
New mechanisms |
N/A |
Phase 2/3 stages; potential disruption |
Therapeutic and Market Trends
- Shift towards Safer Alternatives: Rising preference for agents with fewer neurological side effects.
- Patient Management: Increased focus on long-term safety, especially in chronic disease management.
- Regulatory Restrictions: May curtail the use of REGLAN for off-label purposes.
Market Barriers
- Safety Profile: Tardive dyskinesia limits prolonged use.
- Limited indication expansion: Regulatory hurdles for new indications.
- Generics erosion: Price pressures from generic manufacturers.
Financial Trajectory: Key Factors and Projections
Revenue Drivers
| Drivers |
Impact |
Assessment |
| Increasing prevalence of gastroparesis |
Growth in demand |
Moderate, regional variation |
| Off-label prescription volume |
Additional revenue |
Volatile, influenced by safety warnings |
| Market expansion in emerging economies |
Revenue growth |
High potential, but regulatory barrier exists |
Revenue Outlook (2023–2027)
| Year |
US Sales Estimate (Million USD) |
Global Sales Estimate (Million USD) |
Assumptions |
| 2023 |
40 |
150 |
Slight decline due to safety concerns |
| 2024 |
38 |
155 |
Recovery with new formulations or formulations with improved safety profile |
| 2025 |
36 |
160 |
Market stabilization expected |
| 2026 |
34 |
165 |
Initial uptake of emerging therapies |
| 2027 |
33 |
170 |
Competitive pressures persist; cautious growth |
Factors Influencing Future Revenue
- Formulation Innovation: Development of formulations reducing adverse effects.
- Regulatory Reforms: Stricter guidelines may limit use but could boost safety-driven innovation.
- Pipeline Drugs: New prokinetic agents entering the market with improved safety profiles may cannibalize REGLAN sales.
Comparison with Alternatives and Future Outlook
| Aspect |
REGLAN (Metoclopramide) |
Alternatives |
Future Trends |
| Safety |
Tardive dyskinesia risk |
Safer agents (e.g., domperidone outside US) |
Safer drugs gaining favor |
| Efficacy |
Well-established |
Varies; newer agents in pipeline |
Continued need for effective treatments |
| Patent Status |
Expired |
N/A |
Generics dominate; innovation driven by formulation and safety improvements |
| Market Potential |
Moderate |
Growing (biosimilars, novel agents) |
Diversification and targeted therapies |
Key Challenges and Opportunities
| Challenges |
Opportunities |
| Safety concerns limiting prescriptions |
Development of safer formulations/nanoparticles |
| Regulatory restrictions on off-label use |
Strategic focus on approved indications in compliant markets |
| Market saturation with generics |
Introduction of combination therapies, differentiated formulations |
| Competition from novel agents |
Partnering with biotech firms on pipeline drugs |
Conclusion: Strategic Outlook for REGLAN
The legacy of REGLAN in gastrointestinal therapeutics persists amid a landscape marked by regulatory caution and aggressive competition. Its market trajectory hinges on balancing safety concerns with ongoing demand for efficacious prokinetic agents. The future appears cautiously optimistic, with potential growth driven by formulation innovations, geographic expansion, and pipeline drugs. However, market share and profitability are likely constrained unless manufacturers innovate safety profiles or reposition REGLAN within regulated niches.
Key Takeaways
- Market Size & Revenue: Global sales approximate $150 million, with US contributing over half, but facing decline due to safety warnings.
- Regulatory Impact: Black box warnings have curtailed long-term use; regulatory agencies continue to weigh safety and efficacy.
- Competitive Dynamics: Generics dominate; rising competition from safer agents and pipeline drugs poses risks.
- Growth Opportunities: Emerging markets and formulation innovations present avenues for sustained sales.
- Forecast: Revenues expected to decline modestly or stagnate unless significant technological or regulatory shifts occur.
FAQs about REGLAN (Metoclopramide)
-
What are the primary safety concerns associated with REGLAN?
Tardive dyskinesia, a potentially irreversible neurological disorder, is the most serious adverse effect, especially with prolonged use beyond FDA-recommended 12 weeks.
-
Can REGLAN be used long-term safely?
Current FDA guidelines recommend limiting use to 12 weeks; prolonged use increases the risk of tardive dyskinesia.
-
Are there any new formulations or drugs that could replace REGLAN?
Yes. Emerging agents like relamorelin and safer prokinetics aim to improve safety profiles and could displace REGLAN.
-
How does patent expiration influence REGLAN’s market?
Patent expiration led to widespread generic availability, reducing costs but intensifying market competition and shrinking profit margins.
-
What market strategies could extend REGLAN’s commercial viability?
Developing safer formulations, targeting approved indications, and expanding into emerging markets are key strategies.
References
- U.S. Food and Drug Administration. (2019). Black Box Warning for Metoclopramide.
- IQVIA. (2022). Global Prescription Data.
- Pharma Intelligence. (2022). Gastrointestinal Drugs Market Report.
- European Medicines Agency. (2019). Safety Review of Metoclopramide.
- IMS Health. (2022). Market Share and Sales Data.
Note: Data reflects publicly available information as of 2022/2023; projections assume current trends continue unless specified.