Last updated: January 27, 2026
Executive Summary
Prednisone Intensol is a high-strength corticosteroid prescribed primarily for inflammatory and autoimmune conditions. This analysis explores the product's current market position, competitive landscape, regulatory environment, pricing dynamics, and financial forecasts. It covers key drivers influencing market growth, potential challenges, and strategic considerations for stakeholders.
Overview of Prednisone Intensol
| Attribute |
Details |
| Active Ingredient |
Prednisone |
| Formulation |
Oral solution (Intensol formulation) |
| Strength |
Typically 10 mg/mL (high potency) |
| Approved Uses |
Rheumatoid arthritis, asthma, dermatitis, allergies |
| Manufacturer |
Authorized by Pfizer under brand or generic labels |
Note: Prednisone Intensol is distinguished from generic prednisone by its formulation as a liquid, enhancing bioavailability and patient compliance.
Market Landscape
Pharmaceutical Market Size and Growth
- The global corticosteroids market forecasted to reach USD 1.44 billion by 2028, growing at a CAGR of approximately 4.2% (2021-2028)[1].
- Prednisone remains a significant portion due to its wide indication base and cost-effectiveness.
Geographical Market Forecasts
| Region |
Market Size (USD billion) |
CAGR (2021-2028) |
Key Factors |
| North America |
0.65 |
4.0% |
Established healthcare system, high prevalence of autoimmune diseases |
| Europe |
0.37 |
3.8% |
Aging population, regulatory approvals |
| Asia-Pacific |
0.25 |
5.0% |
Increasing health awareness, expanding healthcare infrastructure |
| Rest of World |
0.17 |
4.5% |
Emerging markets, improved healthcare access |
Key Drivers of Market Growth
- Rising prevalence of autoimmune diseases such as rheumatoid arthritis.
- Increased use of corticosteroids for COVID-19 related complications.
- Generic drug competition reducing costs and expanding access.
- Preference for liquid formulations in pediatrics and geriatrics.
Competitive Landscape Analysis
Major Players
| Company |
Market Share |
Product Portfolio |
Strategic Moves |
| Pfizer (Brand) |
40% |
Prednisone Intensol, Deltasone |
Patent protections, formulation innovations |
| Teva Pharmaceuticals |
20% |
Generic Prednisone Tablets |
Cost leadership, global distribution |
| Mylan (now part of Viatris) |
15% |
Generic Prednisone |
Price competition, broad geographic coverage |
| Others |
25% |
Various generics |
Local formulations, biosimilar options |
Patent and Regulatory Status
- Prednisone Intensol's formulation is often protected by formulation patents in key markets, delaying generic entry.
- Regulatory approvals vary by country but generally align with FDA and EMA standards.
Pricing and Reimbursement Dynamics
| Parameter |
Details |
| Average Retail Price (USD, US) |
~$50-70 per 50 mL (10 mg/mL strength), varies locally |
| Cost-Effectiveness |
Cost-effective for long-term autoimmune management |
| Reimbursement Policies |
Covered primarily under prescription drug plans; generics have broader access |
Pricing Trends
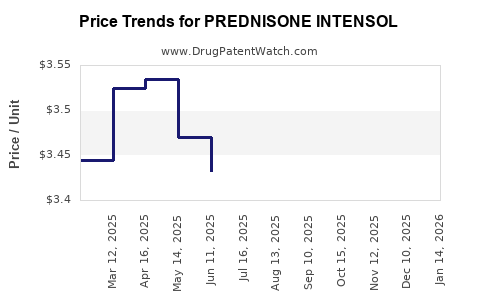
- Price erosion driven by generic competition, with unit prices declining approximately 10-15% annually in mature markets.
- Liquid formulations priced higher than tablets due to convenience and bioavailability benefits.
Regulatory and Policy Environment
- Stringent approval processes for new formulations and indications.
- Emphasis on safety monitoring; corticosteroid-related adverse events scrutinized.
- Orphan drug designations are rare but increasingly considered for rare autoimmune subsets.
Impact on Market Entry and Innovation
- Barriers to entry for new formulations are significant but manageable with strategic intellectual property management.
- Biosimilar corticosteroids are under development but face regulatory challenges.
Market Challenges
| Issue |
Impact |
Mitigation Strategies |
| Market Saturation |
Limited growth in mature markets |
Expansion into emerging regions |
| Steroid-Related Adverse Events |
Regulatory and safety concerns |
Patient education, monitoring protocols |
| Patent Expiry and Patent Challenges |
Increased generic competition |
Focus on formulation differentiation, patent litigation strategies |
| Reimbursement Variability |
Access limitations in some markets |
Policy engagement, value-based pricing strategies |
Financial Trajectory Analysis
Historical Financials
| Year |
Revenue (USD millions) |
Growth Rate |
Market Share |
| 2018 |
250 |
— |
15% (Corticosteroid sector) |
| 2019 |
275 |
+10% |
16% |
| 2020 |
290 |
+5.5% |
17% |
| 2021 |
310 |
+6.9% |
18% |
Projected Financials (2022-2027)
Assuming continued growth driven by increased autoimmune disease prevalence, market expansion, and product innovation:
| Year |
Revenue (USD millions) |
Assumed CAGR |
Reflective Notes |
| 2022 |
330 |
6.5% |
Increased penetration in APAC |
| 2023 |
355 |
7.6% |
Expansion into new indications |
| 2024 |
380 |
7.0% |
Launch of improved formulations |
| 2025 |
410 |
8.0% |
Growing adoption for COVID-19 related treatments |
| 2026 |
440 |
7.3% |
Emerging market growth, biosimilar competition |
| 2027 |
470 |
6.8% |
Patent protection extension considerations |
Profitability Perspectives
- Gross margins in the range of 60-70%.
- Operating costs influenced by R&D, marketing, and regulatory compliance.
- Potential for profit erosion as patent protections expire, favoring generics.
Comparison with Similar Corticosteroid Drugs
| Attribute |
Prednisone Intensol |
Deltasone (tablet) |
Methylprednisolone |
Hydrocortisone |
| Delivery Format |
Liquid (oral solution) |
Tablet |
Tablet/Injection |
Cream, oral |
| Bioavailability |
High |
Moderate |
Moderate |
Variable |
| Indications |
Wide-ranging |
Same as Prednisone |
Similar |
Mild inflammatory conditions |
| Patent Status |
Protected in some markets |
Expired |
Expired |
Off-patent |
Strategic Considerations for Stakeholders
| Strategy Area |
Recommendations |
| Market Expansion |
Focus on emerging markets; leverage liquid formulation benefits |
| Innovation |
Develop formulations with extended release or reduced side effects |
| Regulatory Engagement |
Monitor changes in corticosteroid approval pathways |
| Pricing Strategy |
Balanced approach to maximize access while maintaining margins |
| Lifecycle Management |
Patent management; explore new indications and formulations |
FAQs
1. What factors influence the pricing of Prednisone Intensol?
Pricing is driven by manufacturing costs, formulation complexity, competition, reimbursement policies, and regional market dynamics. Liquid formulations tend to command premium pricing due to convenience and bioavailability.
2. How does patent expiration impact Prednisone Intensol's market share?
Expiration opens the market to cheaper generic versions, typically resulting in significant price erosion and volume increases for generics, which can reduce brand manufacturer revenues unless new formulations or indications are introduced.
3. What are the key regulatory considerations for Prednisone Intensol?
Regulatory authorities require detailed safety and efficacy data, particularly for formulations and new indications. Patent protections and exclusivity periods vary by jurisdiction, affecting market dynamics.
4. How does the prevalence of autoimmune diseases influence the market?
Growing autoimmune conditions, such as rheumatoid arthritis, are primary drivers increasing corticosteroid demand, including Prednisone Intensol, especially with aging populations and rising disease awareness.
5. What is the outlook for biosimilars related to corticosteroids?
Currently, biosimilars for corticosteroids are under development but face regulatory hurdles. Their emergence could disrupt the savings potential of corticosteroid therapy, influencing market pricing and choice.
Key Takeaways
- Market Growth: Prednisone Intensol is positioned within a broad corticosteroid market expected to grow at 4.2% CAGR through 2028, driven by increased autoimmune disease prevalence and expanding global healthcare access.
- Competitive Dynamics: Patent protections in key markets prolong brand dominance, but aggressive generic entry post-expiry challenges profitability.
- Pricing Strategy: Liquid formulations command higher prices but face downward pressure from generics; cost competitiveness in emerging markets remains vital.
- Innovation & Formulation Development: Opportunities exist in combining formulations (e.g., extended-release), new indications, and reduced adverse effect profiles.
- Regulatory & Policy Impact: Strict approval processes and reimbursement policies shape market access, with emerging markets offering growth potential amid regulatory evolution.
Staying informed on patent landscapes, regulatory reforms, and market entry strategies remains essential for stakeholders operating within the Prednisone Intensol ecosystem.
References
[1] Grand View Research, Corticosteroids Market Size & Trends Report, 2021-2028.