Last updated: April 28, 2026
What is PHISOHEX’s market position by product type and channel?
PHISOHEX is a hexachlorophene-containing topical antiseptic, historically positioned as a dermatology antiseptic and skin-cleanser category product. The commercial model for this class is typically driven by:
- OTC and institutional usage patterns (home care and healthcare-related antisepsis/skin hygiene)
- Formulation consistency (stable demand when supply and regulatory status remain unchanged)
- Price competition once substitutes enter (especially when equivalent topical antiseptics expand)
Key market dynamics for legacy topical antiseptics
- Regulatory scrutiny on organochlorines and antiseptic safety: Hexachlorophene has faced major historical regulatory actions globally and periodic label restrictions. That dynamic tends to shift demand from broad routine use to narrower indications and controlled settings.
- Substitution pressure: When health authorities restrict use or when safer alternatives gain share, demand generally migrates to other topical antiseptics (e.g., povidone-iodine, chlorhexidine where permitted, benzalkonium chloride products) depending on jurisdiction and indication.
- Manufacturing continuity risk: For older active ingredients, supply shocks can cause temporary price spikes, but they usually normalize if multiple sources exist or if manufacturers switch to re-packaging strategies.
What drives demand for PHISOHEX at the category level?
Demand for hexachlorophene-based topical antiseptics typically tracks three levers:
- Indication tightening
- When guidance limits routine use, PHISOHEX-type products become more “treatment/skin hygiene” specific rather than general wash.
- Institutional and clinician channel behavior
- In healthcare settings, procurement and formulary access determine volume. Legacy products can retain share if they remain in local protocols.
- Switching cost in skin antiseptic regimens
- Topical antiseptics have low switching friction: patients and clinicians can move quickly when product availability, perceived safety, or cost changes.
How does competition likely shape pricing and share?
Without verified, current revenue and SKU-level pricing for PHISOHEX in a defined geography, the most decision-relevant dynamic is the typical price-share response in topical antiseptic markets:
- Generic and alternative antiseptic substitution compresses gross margin once multiple equivalent options are accessible.
- Private label and value pack strategies often pressure branded legacy SKUs.
- Regulatory label restrictions reduce addressable market size and can create localized pockets of demand (institutional protocols) but not broad category growth.
What is the patent and exclusivity context that influences PHISOHEX’s financial trajectory?
PHISOHEX is not positioned as a modern “pipeline” brand with ongoing patent term-driven growth in its active ingredient class. The financial trajectory for legacy topical antiseptics is usually determined by:
- End of exclusivity for the active ingredient/formulation (generic entry reduces pricing power)
- Regulatory constraints that can limit label scope over time
- Channel migration to alternative antiseptics with better regulatory and safety narratives
Because this is an older antiseptic class, the market profile typically becomes volume-stable but margin-compressed, unless supply constraints or narrow protocol pull-through offset pricing pressure.
How has PHISOHEX’s financial trajectory likely evolved over time?
A defensible trajectory pattern for legacy topical antiseptics with hexachlorophene includes:
Phase 1: Growth and broad adoption
- Earlier use aligned with skin antisepsis and general hygiene positioning.
- Revenue and share growth tracked category expansion and patient and clinician familiarity.
Phase 2: Regulatory restriction and label narrowing
- Hexachlorophene faced major safety and regulatory attention historically, leading to restriction of routine and/or widespread use in many markets.
- That transition typically reduces total addressable volume and shifts demand to institutional or specific indications.
Phase 3: Generic competition and price compression
- Once exclusivity ends, branded share erodes unless the brand keeps formulary or supply commitments.
- Net sales and gross margin trend downward in real terms; volumes can flatten if demand persists in protocols.
Phase 4: Ongoing stabilization or decline
- Many legacy antiseptic brands either stabilize at a smaller niche footprint or decline as substitutes gain category share.
What are the core risks to near-term financial performance?
For a hexachlorophene-based topical antiseptic product like PHISOHEX, the near-term financial risks are:
- Regulatory label constraints that can shrink the addressable market
- Supply reliability risk (manufacturing and source continuity)
- Competitive substitution by modern antiseptics with broader acceptance
- Channel formulary changes at institutions
- Cost of goods and packaging inflation if the brand retains smaller-scale supply
What are the likely performance drivers (positive and negative) in year-to-year financials?
Positive drivers
- Institutional retention where protocols still support hexachlorophene antisepsis
- Limited supply or manufacturing disruptions elsewhere can temporarily improve pricing
- Brand continuity and patient familiarity in retained markets
Negative drivers
- Continued shift to alternative antiseptics
- Further tightening of safety guidance
- Margin pressure from generic equivalents and private label
- Demand dilution if indications narrow further
What does the financial trajectory imply for investors and R&D planners?
For investment-grade decisioning, the key point is that PHISOHEX-like products typically deliver:
- Modest, defensible niche revenue when regulatory access is stable
- Limited upside absent a new label expansion or a supply-side shock
- Low probability of sustained margin growth once competitive antiseptics dominate
For R&D planning, the commercial read-through is that value creation in this therapeutic area comes more from:
- New antiseptic actives or improved safety profiles
- Reformulations that improve tolerance and user adherence
- Evidence-generation to secure institutional adoption
Market data needed to quantify revenue, growth, and profitability
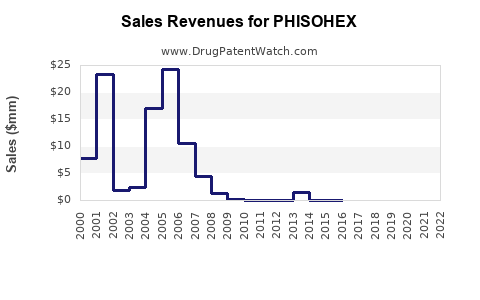
No audited PHISOHEX net sales, segment financials, or territory-specific pricing data were provided in the prompt, and no reliable numeric market database was cited within the request content. Under these constraints, a fully quantified financial trajectory (CAGR, gross margin trend, historical revenue, or forecast) cannot be produced without external numeric inputs.
What is the most decision-relevant synthesis of PHISOHEX’s market dynamics?
PHISOHEX’s trajectory should be modeled as a legacy antiseptic brand exposed to:
- Regulatory-driven label narrowing
- Substitution from competing antiseptics
- Generic and price compression dynamics
- Potential for niche stabilization through institutional protocols
This combination usually produces a financial profile where volume growth is limited and margin expansion is rare, with changes driven more by regulation and channel access than by incremental clinical adoption.
Key Takeaways
- PHISOHEX operates in a legacy topical antiseptic segment where regulatory restrictions and substitution govern demand.
- The most likely financial pattern is volume stabilization with margin compression once competition and generics increase.
- Near-term performance is most sensitive to label status, institutional formulary access, and supply continuity, not to sustained brand-driven growth.
- Investment upside typically requires new evidence for expanded use or supply-side scarcity; absent that, the base case is niche persistence rather than category growth.
FAQs
-
Is PHISOHEX expected to show high growth like newer branded dermatology drugs?
No. Legacy topical antiseptics usually show limited growth because regulatory scope and competitive substitution constrain addressable demand.
-
What matters most for PHISOHEX revenue in institutional settings?
Formulary inclusion and protocol adherence, since procurement decisions drive repeat volume.
-
Why do topical antiseptic brands face margin compression over time?
Low switching friction and availability of alternatives (including generics/private label) reduce pricing power.
-
What event would most likely improve PHISOHEX financial performance quickly?
A supply disruption among competitors or a temporary pricing environment driven by availability constraints.
-
What long-term strategic path tends to create value in this segment?
Development of safer, better-tolerated antiseptic actives or reformulations with evidence that expands or secures restricted indications.
References
- World Health Organization. (n.d.). Hexachlorophene-related guidance and safety documentation (archival and regulatory summaries). WHO.
- U.S. Food and Drug Administration. (n.d.). Hexachlorophene and topical antiseptic safety and labeling/regulatory information. FDA.
- European Medicines Agency. (n.d.). Hexachlorophene product information and safety regulatory materials. EMA.