OCALIVA Drug Patent Profile
✉ Email this page to a colleague
When do Ocaliva patents expire, and what generic alternatives are available?
Ocaliva is a drug marketed by Intercept Pharms Inc and is included in one NDA. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and twenty-three patent family members in thirty-seven countries.
The generic ingredient in OCALIVA is obeticholic acid. There is one drug master file entry for this compound. Additional details are available on the obeticholic acid profile page.
DrugPatentWatch® Generic Entry Outlook for Ocaliva
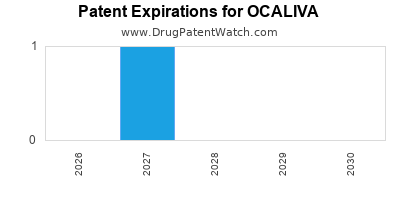
Ocaliva was eligible for patent challenges on May 27, 2020.
There have been eight patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (obeticholic acid), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for OCALIVA?
- What are the global sales for OCALIVA?
- What is Average Wholesale Price for OCALIVA?
Summary for OCALIVA
| International Patents: | 123 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 62 |
| Clinical Trials: | 5 |
| Patent Applications: | 1,808 |
| Drug Prices: | Drug price information for OCALIVA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OCALIVA |
| What excipients (inactive ingredients) are in OCALIVA? | OCALIVA excipients list |
| DailyMed Link: | OCALIVA at DailyMed |
Recent Clinical Trials for OCALIVA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Universitaire Ziekenhuizen KU Leuven | N/A |
| Intercept Pharmaceuticals | N/A |
| M.D. Anderson Cancer Center | Phase 1 |
Paragraph IV (Patent) Challenges for OCALIVA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OCALIVA | Tablets | obeticholic acid | 5 mg and 10 mg | 207999 | 5 | 2020-05-27 |
US Patents and Regulatory Information for OCALIVA
OCALIVA is protected by seven US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-001 | May 27, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-001 | May 27, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-001 | May 27, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-002 | May 27, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-002 | May 27, 2016 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OCALIVA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-001 | May 27, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-001 | May 27, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-002 | May 27, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-002 | May 27, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| Intercept Pharms Inc | OCALIVA | obeticholic acid | TABLET;ORAL | 207999-002 | May 27, 2016 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for OCALIVA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| ADVANZ PHARMA Limited | Ocaliva | obeticholic acid | EMEA/H/C/004093Ocaliva is indicated for the treatment of primary biliary cholangitis (also known as primary biliary cirrhosis) in combination with ursodeoxycholic acid (UDCA) in adults with an inadequate response to UDCA or as monotherapy in adults unable to tolerate UDCA. | Authorised | no | no | yes | 2016-12-12 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for OCALIVA
When does loss-of-exclusivity occur for OCALIVA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 4427
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 16255045
Estimated Expiration: ⤷ Start Trial
Patent: 20205315
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017023161
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 83609
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 17002727
Estimated Expiration: ⤷ Start Trial
China
Patent: 7531742
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 17011535
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 170492
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 17078433
Estimated Expiration: ⤷ Start Trial
El Salvador
Patent: 17005555
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 1792354
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 88958
Estimated Expiration: ⤷ Start Trial
Patent: 71199
Estimated Expiration: ⤷ Start Trial
Patent: 71616
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5269
Estimated Expiration: ⤷ Start Trial
Patent: 4575
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 41057
Estimated Expiration: ⤷ Start Trial
Patent: 18514534
Estimated Expiration: ⤷ Start Trial
Patent: 21183651
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 17013805
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 529
Estimated Expiration: ⤷ Start Trial
Patent: 999
Estimated Expiration: ⤷ Start Trial
Nicaragua
Patent: 1700128
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 180690
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 017501956
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 202003110P
Estimated Expiration: ⤷ Start Trial
Patent: 201708606V
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1707981
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 170140325
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1703773
Estimated Expiration: ⤷ Start Trial
Patent: 23017
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 17000452
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering OCALIVA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2018172438 | オベチコール酸の調製、使用および固体形態 (PREPARATION, USES AND SOLID FORMS OF OBETICHOLIC ACID) | ⤷ Start Trial |
| Taiwan | I673284 | ⤷ Start Trial | |
| Denmark | 3336097 | ⤷ Start Trial | |
| South Korea | 20180083966 | 오베티콜산의 제조법, 용도 및 고체 형태 (PREPARATION USES AND SOLID FORMS OF OBETICHOLIC ACID) | ⤷ Start Trial |
| Denmark | 1392714 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OCALIVA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1392714 | LUC00018 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: ACIDE OBETICHOLIQUE ET SES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES (OCALIVA); AUTHORISATION NUMBER AND DATE: EU/1/16/1139 20161215 |
| 1392714 | 598 | Finland | ⤷ Start Trial | |
| 1392714 | C01392714/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: ACIDUM OBETICHOLICUM; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66530 01.06.2018 |
| 1392714 | 300877 | Netherlands | ⤷ Start Trial | PRODUCT NAME: OBETICHOLZUUR; REGISTRATION NO/DATE: EU/1/16/1139 20161215 |
| 1392714 | 2017/019 | Ireland | ⤷ Start Trial | PRODUCT NAME: OBETICHOLIC ACID; REGISTRATION NO/DATE: EU/1/16/1139/001 EU/1/16/1139/002 20161212 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.