NUZOLVENCE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Nuzolvence, and when can generic versions of Nuzolvence launch?
Nuzolvence is a drug marketed by Entasis Therap and is included in one NDA. There are six patents protecting this drug.
This drug has sixty-two patent family members in thirty-seven countries.
The generic ingredient in NUZOLVENCE is zoliflodacin. One supplier is listed for this compound. Additional details are available on the zoliflodacin profile page.
DrugPatentWatch® Generic Entry Outlook for Nuzolvence
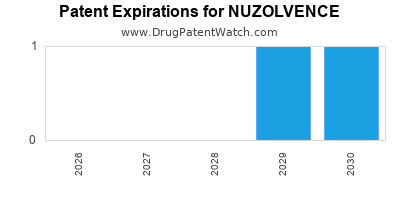
Nuzolvence will be eligible for patent challenges on December 12, 2029. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 12, 2035. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for NUZOLVENCE?
- What are the global sales for NUZOLVENCE?
- What is Average Wholesale Price for NUZOLVENCE?
Summary for NUZOLVENCE
| International Patents: | 62 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| What excipients (inactive ingredients) are in NUZOLVENCE? | NUZOLVENCE excipients list |
| DailyMed Link: | NUZOLVENCE at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for NUZOLVENCE
Generic Entry Date for NUZOLVENCE*:
Constraining patent/regulatory exclusivity:
GENERATING ANTIBIOTIC INCENTIVES NOW NDA:
Dosage:
FOR SUSPENSION;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
US Patents and Regulatory Information for NUZOLVENCE
NUZOLVENCE is protected by six US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of NUZOLVENCE is ⤷ Start Trial.
This potential generic entry date is based on GENERATING ANTIBIOTIC INCENTIVES NOW.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | 9,839,641 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | 9,040,528 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | 9,540,394 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Entasis Therap | NUZOLVENCE | zoliflodacin | FOR SUSPENSION;ORAL | 219491-001 | Dec 12, 2025 | RX | Yes | Yes | 9,187,495 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for NUZOLVENCE
When does loss-of-exclusivity occur for NUZOLVENCE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 3862
Patent: COMPUESTOS HETEROCICLICOS ESPIROFUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09305203
Patent: Fused, spirocyclic heteroaromatic compounds for the treatment of bacterial infections
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0920180
Patent: composto, uso de um composto, método para tratar uma infecção bacteriana em um animal de sangue quente, composição farmacêutica, e, processo para preparar um composto
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 36335
Patent: COMPOSES HETEROAROMATIQUES SPIROCYCLIQUES FUSIONNES POUR TRAITER LES INFECTIONS BACTERIENNES (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11000838
Patent: Compuestos heterociclicos derivados de tetraciclos ortocondensados con un grupo pirimidina 2,4,6-triona espirocondensado, inhibidores de la adn girasa bacteriana; proceso de preparacion; composicion farmaceutica; y su uso para el tratamiento de una infeccion bacteriana.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2245605
Patent: Fused, spirocyclic heteroaromatic compounds for the treatment of bacterial infections
Estimated Expiration: ⤷ Start Trial
Patent: 4086565
Patent: FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 82126
Patent: COMPUESTOS HETEROAROMATICOS FUSIONADOS Y ESPIROCICLICOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110195
Patent: COMPUESTOS HETEROAROMÁTICOS ESPIROCÍCLICOS FUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 110083
Patent: COMPUESTOS HETEROAROMÁTICOS ESPIROCÍCLICOS FUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000102
Patent: COMPUESTOS HETEROAROMATICOS ESPIROCICLICOS FUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010980
Patent: COMPUESTOS HETEROAROMÁTICOS ESPIROCÍCLICOS FUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 1100613
Patent: КОНДЕНСИРОВАННЫЕ, СПИРОЦИКЛИЧЕСКИЕ ГЕТЕРОАРОМАТИЧЕСКИЕ СОЕДИНЕНИЯ ДЛЯ ЛЕЧЕНИЯ БАКТЕРИАЛЬНЫХ ИНФЕКЦИЙ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 50077
Patent: COMPOSÉS HÉTÉROAROMATIQUES SPIROCYCLIQUES FUSIONNÉS POUR TRAITER LES INFECTIONS BACTÉRIENNES (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 60127
Patent: 用於治療細菌感染的稠合、螺環雜芳族化合物 (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 03483
Patent: 用於治療細菌感染的稠合、螺環雜芳族化合物 (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 49169
Estimated Expiration: ⤷ Start Trial
Patent: 12505261
Estimated Expiration: ⤷ Start Trial
Patent: 15178510
Patent: 細菌感染の処置用の縮合スピロ環式ヘテロ芳香族化合物 (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11003598
Patent: COMPUESTOS HETEROAROMATICOS ESPIROCICLICOS, FUSIONADOS, PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS. (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS.)
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1103533
Patent: FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1676886
Estimated Expiration: ⤷ Start Trial
Patent: 110090895
Patent: FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 65611
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1020258
Patent: Chemical compounds 542
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 174
Patent: NUEVOS HETEROCICLOS SUSTITUIDOS, COMPOSICIONES FARMACEUTICAS CONTENIENDOLOS, PROCEDIMIENTOS DE PREPARACION Y APLICACIONES
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering NUZOLVENCE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Costa Rica | 20110195 | COMPUESTOS HETEROAROMÁTICOS ESPIROCÍCLICOS FUSIONADOS PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS | ⤷ Start Trial |
| Taiwan | 201020258 | Chemical compounds 542 | ⤷ Start Trial |
| Canada | 2897887 | COMPOSES ET PROCEDES POUR LE TRAITEMENT D'INFECTIONS BACTERIENNES (COMPOUNDS AND METHODS FOR TREATING BACTERIAL INFECTIONS) | ⤷ Start Trial |
| San Marino | T202000261 | ⤷ Start Trial | |
| Eurasian Patent Organization | 026901 | СОЕДИНЕНИЯ И СПОСОБЫ ЛЕЧЕНИЯ БАКТЕРИАЛЬНЫХ ИНФЕКЦИЙ (COMPOUNDS AND METHODS FOR TREATING BACTERIAL INFECTIONS) | ⤷ Start Trial |
| Mexico | 2011003598 | COMPUESTOS HETEROAROMATICOS ESPIROCICLICOS, FUSIONADOS, PARA EL TRATAMIENTO DE INFECCIONES BACTERIANAS. (FUSED, SPIROCYCLIC HETEROAROMATIC COMPOUNDS FOR THE TREATMENT OF BACTERIAL INFECTIONS.) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Market Dynamics and Financial Trajectory for NUZOLVENCE
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.