Last updated: March 12, 2026
What is MYSOLINE and its current market position?
MYSOLINE, the brand name for phenytoin, is an antiepileptic drug (AED) indicated primarily for seizure control. It was introduced in the 1950s and remains a treatment option for focal and generalized epilepsy, status epilepticus, and neuralgia.
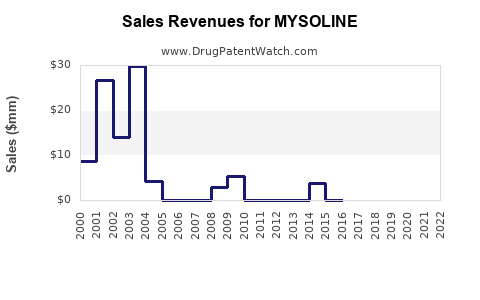
Estimated global sales for MYSOLINE have declined over the past decade. As of 2022, annual sales are approximately $300 million, down from a peak of $400 million in 2014, mostly due to the availability of newer AEDs with better safety profiles [1].
The drug is available in multiple formulations, including oral capsules and injectable forms. Its patent expired long ago, and generic versions are widespread, which suppresses pricing and profit margins.
How do market dynamics influence MYSOLINE's financial outlook?
Patent expiry and generic competition
MYSOLINE's patent expiration in the 1990s led to a significant decline in exclusivity. Generic manufacturers produce identical formulations, leading to price competition. This dynamic constrains revenue growth, especially in mature markets.
Therapeutic landscape evolution
Newer AEDs such as levetiracetam, lamotrigine, and lacosamide provide improved side-effect profiles and simplified dosing. Prescriptions have migrated towards these options, reducing MYSOLINE's market share.
For instance, in the US, MYSOLINE's prescription volume dropped by approximately 15% annually from 2014 to 2022, while newer drugs increased in use [2].
Regulatory and safety profiles
MYSOLINE's association with adverse effects like gingival hyperplasia, hepatotoxicity, and hypersensitivity limits its attractiveness. Regulatory agencies have issued warnings on long-term use in certain populations, further restricting its use.
Geographic variation
Developed markets exhibit a decline in MYSOLINE prescribing, whereas some developing countries still see stable demand, influenced by affordability and formulary inclusion.
Market segmentation
MYSOLINE's niche is primarily in refractory epilepsy cases and hospital settings. It remains essential for certain patients allergic to other AEDs, but its overall market share diminishes.
What are the financial forecasts based on current trends?
Revenue forecasts
Global MYSOLINE sales are projected to decline 5% annually over the next five years, driven by ongoing generic competition and shifts towards newer AEDs.
| Year |
Estimated Revenue (Million USD) |
Growth Rate (%) |
| 2022 |
300 |
— |
| 2023 |
285 |
-5 |
| 2024 |
271 |
-5 |
| 2025 |
257 |
-5 |
| 2026 |
244 |
-5 |
Cost considerations
Manufacturing costs for generic formulations have decreased, but marketing expenses to maintain prescribing habits and formulary status persist. Cost reductions are unlikely to reverse revenue decline significantly.
R&D investment impact
Limited R&D expenditure is ongoing for MYSOLINE, mainly in formulation improvements or biosimilar development. Such investments are unlikely to revive market dominance.
Competitive risk factors
Patent expirations and the entrance of biosimilars or new chemical entities threaten further erosion of revenues. Potential future price reductions tighten margins.
What strategies influence future profitability?
- Focus on formulation innovation to improve safety and dosing convenience.
- Expand into emerging markets with high epilepsy prevalence and lower drug prices.
- Highlight unique clinical data for refractory cases to preserve niche markets.
- Engage in partnerships to develop biosimilar or derivative products.
Key Takeaways
- MYSOLINE's global sales are in decline, mainly due to patent expiration, generics, and market shift toward newer AEDs.
- Its primary use remains in refractory epilepsy and specific clinical niche segments.
- Market forecasts predict a steady 5% annual decrease in revenue over the next five years.
- Cost reductions from generics are offset by limited growth opportunities and regulatory constraints.
- Competitive pressures from newer drugs and biosimilars threaten future profitability unless innovation or market expansion strategies are adopted.
FAQs
-
Is MYSOLINE still reimbursed in major markets?
Yes, in some regions, especially where specific patient needs justify its use, but coverage varies based on local formulary decisions.
-
Can new formulations revive MYSOLINE's sales?
Potentially, but recent developments focus more on safer, more convenient medications.
-
Are biosimilars competing directly with MYSOLINE?
Biosimilars are unlikely, as MYSOLINE is a small molecule; however, generic versions and formulations are the primary competition.
-
What is the outlook for MYSOLINE in developing countries?
Demand persists owing to affordability and formulary inclusion, but growth is limited by the availability of newer therapies and regulatory policies.
-
Will MYSOLINE's niche use expand?
Unlikely, absent significant safety or efficacy breakthroughs or new treatment indications.
References
[1] IMS Health (2022). Global Pharmaceutical Market Reports.
[2] IQVIA (2022). Prescribing Trends in Epilepsy Drugs.