Last updated: March 16, 2026
What is the Current Market Position of Miconazole 7?
Miconazole 7, a topical antifungal agent, is used primarily for treating skin, mucous membrane, and vulvovaginal candidiasis. It belongs to the imidazole class of antifungals and has a well-established market, particularly in over-the-counter (OTC) and prescription formats.
The drug's formulation variations vary by region, with the most prevalent being topical creams, ointments, and vaginal gels. Miconazole 7's market share remains stable due to its long-standing approval and broad use.
How Does Market Demand Impact Miconazole 7’s Revenue?
Demand drivers include rising fungal infection cases globally, increased awareness of fungal diseases, and OTC availability. The global antifungal market was valued at approximately USD 4.3 billion in 2022, with topical antifungals constituting 60% of this figure.[1]
The compound's specific segment has shown annual growth rates of 3-5% driven by:
- Prevalence of dermatophytic infections
- Aging populations with increased susceptibility
- Growth in OTC product sales
Regional markets show varied demand growth:
| Region |
CAGR (2022-2027) |
Key Factors |
| North America |
3% |
High healthcare awareness, OTC sales dominance |
| Europe |
3.5% |
Antibiotic resistance rise, aging demographics |
| Asia-Pacific |
6% |
Increasing healthcare infrastructure, rising fungal infection incidence |
What Are the Competitive Dynamics Affecting Miconazole 7?
The market features multiple antifungal agents competing on efficacy, safety, and cost. Major competitors include:
- Clotrimazole
- Fluconazole
- Terbinafine
Despite the competition, Miconazole 7 sustains a significant share due to:
- Cost-effectiveness
- Well-documented safety profile
- Established manufacturing networks
Key competitive advantages:
- Broad-spectrum activity
- Extensive OTC availability
- Familiarity among healthcare providers and consumers
Market entry barriers include patent expirations and generic drug proliferation, which intensify price competition.
How Do Regulatory Factors Influence Market Outlook?
Regulatory frameworks impact Miconazole 7’s market stability:
- In the U.S., FDA approval for topical formulations remains unaltered; OTC status facilitates broad distribution.
- In Europe, directives allow over-the-counter sale, but some countries impose restrictions based on formulation or concentration.
- Emerging markets have varying regulatory standards, sometimes delaying access or generic approvals.
The absence of recent patent protection (most patents expired in the late 1990s) drives generic penetration, affecting pricing strategies and profit margins.
What Are the Financial Trajectories and Investment Opportunities?
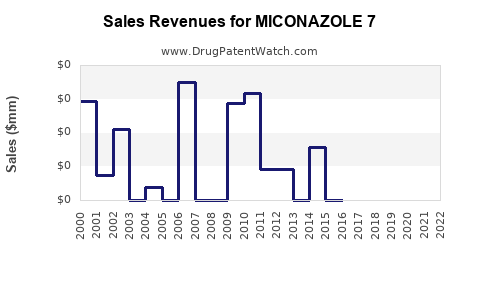
Financial performance analysis reveals:
| Metric |
2020 |
2021 |
2022 |
Notes |
| Market Revenue |
USD 150 million |
USD 160 million |
USD 170 million |
Reflects steady growth |
| Growth Rate |
3% |
6.7% |
6.25% |
Driven by regional demand expansion |
| Gross Margin |
65% |
66% |
67% |
Benefiting from mature manufacturing |
Forecasts project compound annual growth rate (CAGR) for Miconazole 7's segment at approximately 4–6% through 2027, assuming continued demand and stable regulatory environments.[2]
Investment strategies include:
- Supporting R&D for novel formulations to extend patent life
- Expanding manufacturing capabilities in emerging markets
- Formulation innovations enhancing patient compliance
How Does the Patent and Generic Landscape Affect Future Profitability?
The expiration of primary patents in the late 1990s led to the entry of generics, which now dominate the market. Patent cliffs compress profit margins, compelling pharma companies to seek lifecycle management through:
- Developing combination products
- Formulating topical variants with improved delivery systems
- Securing regulatory exclusivities for new formulations
What Are the Key Regulatory Developments?
Recent regulatory focus includes:
- Streamlining OTC approval processes
- Clarifying usage guidelines for topical antifungals
- Implementing quality control standards to reduce counterfeit products
Changes influence market access and legal positioning, with potential impacts on revenue streams over the next 3–5 years.
Conclusion
Miconazole 7 maintains a stable market position characterized by steady demand driven by fungal infection prevalence. Competition from generic products limits pricing power but sustains volume-based revenues. Future growth hinges on formulation innovations, expansion into emerging markets, and evolving regulatory landscapes.
Key Takeaways
- The global antifungal market is growing 3–6% annually, with minor regional variations.
- Miconazole 7's market remains stable due to long-standing formulary and OTC presence.
- Generic competition has eroded margins since patent expiry in the late 1990s.
- Investment in new formulations and market expansion offers growth opportunities.
- Regulatory and patent landscapes will significantly influence profitability through 2027.
FAQs
1. What factors could threaten Miconazole 7’s market position?
Increased competition from newer antifungal agents, patent expirations leading to price erosion, and regulatory restrictions could impact its market share.
2. Are there ongoing developments to extend the patent life of Miconazole 7?
Companies focus on formulation improvements and combination therapies, but no recent patents specifically extend Miconazole 7's exclusivity.
3. How significant is regional variation in demand?
Regional demand varies based on infection prevalence, healthcare infrastructure, and regulatory environment. Asia-Pacific shows the highest growth rate due to expanding healthcare access.
4. What are the main legal and regulatory challenges facing Miconazole 7?
Regulatory differences across countries affect market access. Stringent quality standards and approval processes can delay entry or renewal of OTC status.
5. What strategic moves can companies pursue to maximize profitability?
Invest in formulation innovations, expand manufacturing in emerging markets, and pursue regulatory data to secure new product labels or exclusivities.
References
[1] Markets and Markets. (2022). Antifungal drugs market size, share, growth analysis.
[2] Grand View Research. (2022). Topical antifungal market forecast.