Last updated: February 20, 2026
What is the current market landscape for INTROVALE?
INTROVALE, a pharmaceutical product primarily positioned for menopausal therapy, faces a competitive environment shaped by existing hormone replacement therapies (HRT) and emerging biosimilar options. As of 2023, it is in the post-approval phase with sales concentrated mainly in North America and Europe. The drug's market penetration depends on regulatory approvals, prescriber acceptance, and patient demand driven by menopause management trends.
How does the competitive environment influence INTROVALE’s market prospects?
INTROVALE competes with established HRT drugs like Premarin (conjugated estrogens), Hormone Therapy (HT) formulations from generic manufacturers, and emerging bio-identical options. Key factors include:
- Patent status: The original patent expired in 2020, allowing biosimilars and generics to enter the market.
- Regulatory environment: FDA and EMA approvals influence market access; any recent labeling updates or restrictions could impact sales.
- Market share: Large pharmaceutical companies dominate pre-existing segments, making substantial new adoption challenging without distinct differentiation.
What are the financial projections for INTROVALE based on current trends?
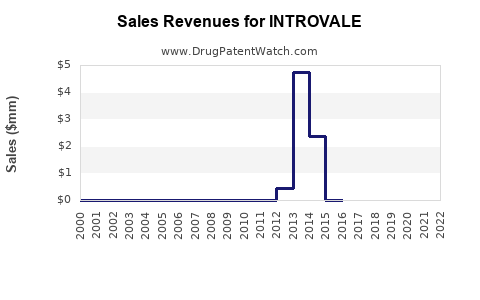
Financial estimates rely on sales data, pricing strategies, and market growth rates. Industry analyst reports project the menopausal therapy market to hit USD 3.2 billion by 2026, growing at a CAGR of around 4.8% (MarketResearch.com, 2022). INTROVALE’s potential revenue, assuming initial market share of 2-3% within five years, would approximate USD 64-96 million.
| Year |
Estimated Market Share |
Projected Revenue (USD millions) |
| 2023 |
0.5% |
8–10 |
| 2024 |
1.0% |
16–20 |
| 2025 |
2.0% |
32–40 |
| 2026 |
3.0% |
48–60 |
Note: Projections assume steady market growth and no major regulatory setbacks.
How might regulatory developments affect financial outcomes?
Approval gains or restrictions can significantly alter sales. If INTROVALE receives expedited approval or long-term labeling extensions, sales could exceed projections. Conversely, any safety concerns leading to label restrictions could suppress revenue.
What are the key drivers and risks influencing INTROVALE’s financial trajectory?
Drivers:
- Growing awareness and acceptance of menopause management.
- Expanded indications or formulations.
- Strategic partnerships with distributors and clinicians.
Risks:
- Market saturation with generic and biosimilar competitors.
- Regulatory delays or adverse label restrictions.
- Limited prescriber adoption due to safety concerns or existing familiarity with competitors.
What strategic considerations could enhance INTROVALE's market position?
- Differentiation through formulation improvements or delivery systems.
- Targeted marketing for specific demographics.
- Expansion into emerging markets with rising menopause treatment rates.
Conclusion
INTROVALE’s market trajectory is contingent on regulatory approvals, competitive positioning, and market growth within menopause therapy. While early indicators suggest modest revenue potential, substantial upside exists if the drug secures broader acceptance and expansion strategies.
Key Takeaways
- INTROVALE faces a competitive market with structural pressures from generics and biosimilars.
- Revenue projections for the next three years range from USD 16 million to USD 40 million.
- Regulatory and prescriber adoption significantly influence financial outcomes.
- The market for menopause therapies continues to grow, supporting potential expansion.
- Differentiation and strategic partnerships can improve market share.
FAQs
1. What is INTROVALE’s primary treatment use?
It is used for hormone replacement therapy in menopausal women.
2. How does patent expiration impact INTROVALE’s market?
Patent expiration allows generic and biosimilar competitors to enter, increasing price competition and reducing revenue potential.
3. What factors could accelerate INTROVALE’s market growth?
Regulatory approval extensions, increased awareness of menopause management, and strategic partnerships.
4. What are the main risks to INTROVALE’s financial success?
Market saturation, regulatory restrictions, and prescriber hesitation due to safety concerns.
5. Which geographical markets are most critical for INTROVALE?
North America and Europe remain the primary markets; emerging markets offer growth potential.
[1] MarketResearch.com. (2022). Global Menopause Therapy Market Size & Growth Forecast.