INBRIJA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Inbrija, and when can generic versions of Inbrija launch?
Inbrija is a drug marketed by Merz and is included in one NDA. There are six patents protecting this drug.
This drug has one hundred and twenty-nine patent family members in twenty-two countries.
The generic ingredient in INBRIJA is levodopa. There are eighteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the levodopa profile page.
DrugPatentWatch® Generic Entry Outlook for Inbrija
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 16, 2032. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for INBRIJA?
- What are the global sales for INBRIJA?
- What is Average Wholesale Price for INBRIJA?
Summary for INBRIJA
| International Patents: | 129 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 134 |
| Drug Prices: | Drug price information for INBRIJA |
| What excipients (inactive ingredients) are in INBRIJA? | INBRIJA excipients list |
| DailyMed Link: | INBRIJA at DailyMed |
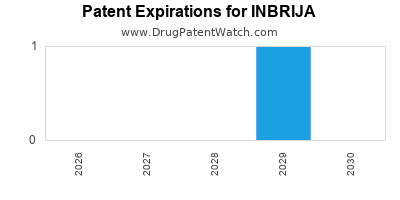

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for INBRIJA
Generic Entry Date for INBRIJA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INHALATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for INBRIJA
| Drug Class | Aromatic Amino Acid |
US Patents and Regulatory Information for INBRIJA
INBRIJA is protected by six US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of INBRIJA is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for INBRIJA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| Merz | INBRIJA | levodopa | POWDER;INHALATION | 209184-001 | Dec 21, 2018 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for INBRIJA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Acorda Therapeutics Ireland Limited | Inbrija | levodopa | EMEA/H/C/004786Inbrija is indicated for the intermittent treatment of episodic motor fluctuations (OFF episodes) in adult patients with Parkinson’s disease (PD) treated with a levodopa/dopa-decarboxylase inhibitor. | Authorised | no | no | no | 2019-09-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for INBRIJA
When does loss-of-exclusivity occur for INBRIJA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 13342246
Estimated Expiration: ⤷ Start Trial
Patent: 13342247
Estimated Expiration: ⤷ Start Trial
Patent: 13342248
Estimated Expiration: ⤷ Start Trial
Patent: 17279626
Estimated Expiration: ⤷ Start Trial
Patent: 18204674
Estimated Expiration: ⤷ Start Trial
Patent: 18222983
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2015010601
Estimated Expiration: ⤷ Start Trial
Patent: 2015010603
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 90451
Estimated Expiration: ⤷ Start Trial
Patent: 90454
Estimated Expiration: ⤷ Start Trial
Patent: 90459
Estimated Expiration: ⤷ Start Trial
China
Patent: 4918607
Estimated Expiration: ⤷ Start Trial
Patent: 5120843
Estimated Expiration: ⤷ Start Trial
Patent: 9106697
Estimated Expiration: ⤷ Start Trial
Patent: 0833539
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 16821
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 16821
Estimated Expiration: ⤷ Start Trial
Patent: 16826
Estimated Expiration: ⤷ Start Trial
Patent: 25611
Estimated Expiration: ⤷ Start Trial
Patent: 15679
Estimated Expiration: ⤷ Start Trial
Patent: 57301
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 12884
Estimated Expiration: ⤷ Start Trial
Patent: 13186
Estimated Expiration: ⤷ Start Trial
Patent: 13187
Estimated Expiration: ⤷ Start Trial
Patent: 13535
Estimated Expiration: ⤷ Start Trial
Patent: 14957
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 47786
Estimated Expiration: ⤷ Start Trial
Patent: 48501
Estimated Expiration: ⤷ Start Trial
Patent: 69808
Estimated Expiration: ⤷ Start Trial
Patent: 21629
Estimated Expiration: ⤷ Start Trial
Patent: 36834
Estimated Expiration: ⤷ Start Trial
Patent: 15536197
Estimated Expiration: ⤷ Start Trial
Patent: 15536988
Estimated Expiration: ⤷ Start Trial
Patent: 15536989
Estimated Expiration: ⤷ Start Trial
Patent: 18150355
Estimated Expiration: ⤷ Start Trial
Patent: 18162258
Estimated Expiration: ⤷ Start Trial
Patent: 19213867
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 1008
Estimated Expiration: ⤷ Start Trial
Patent: 7602
Estimated Expiration: ⤷ Start Trial
Patent: 2999
Estimated Expiration: ⤷ Start Trial
Patent: 15005767
Estimated Expiration: ⤷ Start Trial
Patent: 15005768
Estimated Expiration: ⤷ Start Trial
Patent: 20012506
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 8682
Patent: Dosator apparatus for filling a capsule with dry powder
Estimated Expiration: ⤷ Start Trial
Patent: 8684
Patent: High dose levodopa capsules for pulmonary use
Estimated Expiration: ⤷ Start Trial
Patent: 3459
Patent: Dosator for filling a capsule with dry powder
Estimated Expiration: ⤷ Start Trial
Patent: 7376
Patent: High dose levodopa capsules for pulmonary use
Estimated Expiration: ⤷ Start Trial
Patent: 7378
Patent: Ultra low density pulmonary powders
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 16821
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 16821
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 70987
Patent: ПОРОШКИ ДЛЯ ИНГАЛЯЦИИ С УЛЬТРАНИЗКОЙ ПЛОТНОСТЬЮ (ULTRA LOW DENSITY INHALATION POWDERS)
Estimated Expiration: ⤷ Start Trial
Patent: 76093
Patent: КАПСУЛЫ, СОДЕРЖАЩИЕ ВЫСОКИЕ ДОЗЫ ЛЕВОДОПЫ, ДЛЯ ПРИМЕНЕНИЯ ПУТЕМ ИНГАЛЯЦИИ (HIGH DOSE LEVODOPA CAPSULES FOR PULMONARY USE)
Estimated Expiration: ⤷ Start Trial
Patent: 15121091
Patent: ПОРОШКИ ДЛЯ ИНГАЛЯЦИИ С УЛЬТРАНИЗКОЙ ПЛОТНОСТЬЮ
Estimated Expiration: ⤷ Start Trial
Patent: 15121092
Patent: КАПСУЛЫ, СОДЕРЖАЩИЕ ВЫСОКИЕ ДОЗЫ ЛЕВОДОПЫ, ДЛЯ ПРИМЕНЕНИЯ ПУТЕМ ИНГАЛЯЦИИ
Estimated Expiration: ⤷ Start Trial
Patent: 18144622
Patent: КАПСУЛЫ, СОДЕРЖАЩИЕ ВЫСОКИЕ ДОЗЫ ЛЕВОДОПЫ, ДЛЯ ПРИМЕНЕНИЯ ПУТЕМ ИНГАЛЯЦИИ
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201706465X
Patent: HIGH DOSE LEVODOPA CAPSULES FOR PULMONARY USE
Estimated Expiration: ⤷ Start Trial
Patent: 201707103S
Patent: ULTRA LOW DENSITY PULMONARY POWDERS
Estimated Expiration: ⤷ Start Trial
Patent: 202109328Q
Patent: HIGH DOSE LEVODOPA CAPSULES FOR PULMONARY USE
Estimated Expiration: ⤷ Start Trial
Patent: 201503543P
Patent: HIGH DOSE LEVODOPA CAPSULES FOR PULMONARY USE
Estimated Expiration: ⤷ Start Trial
Patent: 201503547T
Patent: ULTRA LOW DENSITY PULMONARY POWDERS
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1504058
Patent: HIGH DOSE LEVODOPA CAPSULES FOR PULMONARY USE
Estimated Expiration: ⤷ Start Trial
Patent: 1504060
Patent: ULTRA LOW DENSITY PULMONARY POWDERS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2257164
Estimated Expiration: ⤷ Start Trial
Patent: 2337781
Estimated Expiration: ⤷ Start Trial
Patent: 2389785
Estimated Expiration: ⤷ Start Trial
Patent: 2735396
Estimated Expiration: ⤷ Start Trial
Patent: 150108816
Estimated Expiration: ⤷ Start Trial
Patent: 150110480
Estimated Expiration: ⤷ Start Trial
Patent: 210062730
Estimated Expiration: ⤷ Start Trial
Patent: 210152020
Estimated Expiration: ⤷ Start Trial
Patent: 220054703
Estimated Expiration: ⤷ Start Trial
Patent: 230116102
Estimated Expiration: ⤷ Start Trial
Patent: 240134230
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 44153
Estimated Expiration: ⤷ Start Trial
Patent: 80271
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering INBRIJA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2014074795 | ⤷ Start Trial | |
| Spain | 2621549 | ⤷ Start Trial | |
| Austria | 394136 | ⤷ Start Trial | |
| European Patent Office | 2630954 | APPORT PULMONAIRE DE LEVODOPA (PULMONARY DELIVERY FOR LEVODOPA) | ⤷ Start Trial |
| South Korea | 20150102960 | ⤷ Start Trial | |
| Australia | 2002352836 | ⤷ Start Trial | |
| Japan | 6347786 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for INBRIJA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3209302 | 11/2023 | Austria | ⤷ Start Trial | PRODUCT NAME: KOMBINATION VON FOSLEVODOPA ODER EINEM PHARMAZEUTISCH AKZEPTABLEN SALZ DAVON UND FOSCARBIDOPA ODER EINEM PHARMAZEUTISCH AKZEPTABLEN SALZ DAVON; REGISTRATION NO/DATE: 141371 20220826 |
| 1613296 | 92782 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: SAFINAMIDE A UTILISER EN COMBINAISON AVEC LEVODOPA/PDI, ET EVENTUELLEMENT AVEC D'AUTRES MEDICAMENTS PD , POUR LE TRAITEMENT DE LA MALADIE DE PARKINSON; FIRST REGISTRATION DATE: 20150224 |
| 3209302 | 202340008 | Slovenia | ⤷ Start Trial | PRODUCT NAME: COMBINATION OF FOSLEVODOPA OR ITS PHARMACEUTICALLY ACCEPTABLE SALT AND FOSCARBIDOPA OR ITS PHARMACEUTICALLY ACCEPTABLE SALT; NATIONAL AUTHORISATION NUMBER: H/05/00514/002; DATE OF NATIONAL AUTHORISATION: 20221215; AUTHORITY FOR NATIONAL AUTHORISATION: SI; FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: 141371; DATE OF FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: 20220825; AUTHORITY OF FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: AT |
| 3209302 | CR 2023 00015 | Denmark | ⤷ Start Trial | PRODUCT NAME: KOMBINATION AF FOSLEVODOPA ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF OG FOSCARBIDOPA ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; NAT. REG. NO/DATE: 66549 (DK) 20221205; FIRST REG. NO/DATE: AT 141371 20220826 |
| 3209302 | C202330028 | Spain | ⤷ Start Trial | PRODUCT NAME: COMBINACION DE FOSLEVODOPA O UNA SAL FARMACEUTICAMENTE ACEPTABLE DE LA MISMA Y FOSCARBIDOPA O UNA SAL FARMACEUTICAMENTE ACEPTABLE DE LA MISM.; NATIONAL AUTHORISATION NUMBER: 88677-SE/H/0415/003/DC; DATE OF AUTHORISATION: 20230220; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): 141371; DATE OF FIRST AUTHORISATION IN EEA: 20220825 |
| 3209302 | 301224 | Netherlands | ⤷ Start Trial | PRODUCT NAME: COMBINATIE VAN FOSLEVODOPA EN FOSCARBIDOPA, ELK DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; NATIONAL REGISTRATION NO/DATE: RVG128752 20221107; FIRST REGISTRATION: AT 141371 20220826 |
| 3209302 | 23C1035 | France | ⤷ Start Trial | PRODUCT NAME: COMBINAISON DE FOSLEVODOPA ET DE FOSCARBIDOPA, CHACUNE DANS TOUTES LES FORMES PROTEGEES PAR LE BREVET DE BASE; NAT. REGISTRATION NO/DATE: 34009 302 790 8 3 20230901; FIRST REGISTRATION: AT - 141371 20220826 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Inbrija (Levodopa Inhalation Powder)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
