Last updated: February 19, 2026
ILUVIEN is a sustained-release intravitreal implant containing corticosteroid fluocinolone acetonide used for treating chronic uveitis affecting the posterior segment of the eye. The drug's market trajectory is shaped by its patent expiration dates, competitive landscape, and the evolving treatment protocols for uveitis.
What are ILUVIEN's Core Patents and Expiration Dates?
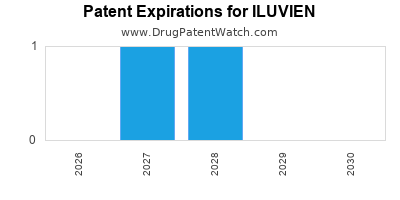
ILUVIEN’s intellectual property protection is primarily secured by patents covering its formulation, manufacturing process, and method of use. Key patents include U.S. Patent Nos. 7,772,479, 7,943,179, and 8,852,668.
- U.S. Patent No. 7,772,479: This patent, titled "Controlled Release Ophthalmic Implant," generally covers methods of producing sustained-release ophthalmic inserts containing a corticosteroid. Its listed expiration date is August 11, 2026. [1]
- U.S. Patent No. 7,943,179: This patent, titled "Ophthalmic Implants and Methods of Use," focuses on specific implant designs and their use in treating ocular conditions. Its listed expiration date is April 26, 2027. [1]
- U.S. Patent No. 8,852,668: This patent, titled "Ophthalmic Drug Delivery Systems and Methods," pertains to systems and methods for delivering drugs to the eye. Its listed expiration date is October 4, 2029. [1]
It is critical to note that patent expiration dates can be subject to extensions, such as those granted under the Patent Term Adjustment (PTA) or Patent Term Extension (PTE) provisions in the United States, due to delays in regulatory review by the U.S. Food and Drug Administration (FDA). The listed dates represent the statutory expiration without considering potential extensions. Companies also maintain portfolios of related patents, some with later expiration dates, that may cover manufacturing nuances, new formulations, or expanded therapeutic uses. These additional patents can create a layered defense against generic competition.
What is the Current Market Landscape for ILUVIEN?
The market for ILUVIEN is characterized by its niche application in posterior uveitis, a condition affecting approximately 1 in 10,000 people in developed countries. [2] Its primary indication is for the treatment of inflammation and edema associated with chronic non-infectious uveitis affecting the posterior segment of the eye in adult patients who are pseudophakic or phakic with an artificial lens.
Key players in the uveitis treatment market include manufacturers of corticosteroid injections, other intravitreal therapies, and systemic immunosuppressants.
- Competitors: Direct competitors offering intravitreal corticosteroid treatments include those utilizing dexamethasone implants (e.g., Ozurdex, which has since been discontinued in the U.S. but remains relevant in global markets) and other fluocinolone acetonide formulations. Broader competition comes from systemic therapies like methotrexate, azathioprine, and biologics such as adalimumab, which are used for more generalized or severe inflammatory conditions. [3]
- Treatment Modalities: ILUVIEN offers a sustained-release mechanism, providing treatment for up to 36 months from a single injection. This contrasts with frequent topical or systemic treatments that require consistent administration and can lead to dose-dependent side effects. [4]
- Reimbursement and Payer Landscape: Reimbursement for ILUVIEN can be complex, involving physician fees for administration and the cost of the implant itself. Payer coverage varies by geography and health system, influencing physician prescribing patterns and patient access.
How Has ILUVIEN Performed Financially?
Eversight, the current U.S. distributor of ILUVIEN, acquired the rights from Alimera Sciences. Financial performance data is reported by Eversight.
- Revenue Trends: Specific revenue figures for ILUVIEN are not always publicly delineated in granular detail by its current owner. However, the product's market penetration is influenced by factors such as physician adoption, patient eligibility, and the competitive environment. Prior to the acquisition, Alimera Sciences reported net revenues for ILUVIEN. For example, in the year ended December 31, 2022, Alimera Sciences reported net revenues of $107.9 million for ILUVIEN. [5] Eversight has not publicly disclosed separate revenue figures for ILUVIEN since the acquisition. The overall market for ophthalmic pharmaceuticals is substantial, with sustained-release therapies aiming to capture significant shares by offering improved patient compliance and efficacy.
- Cost of Goods Sold (COGS): The manufacturing process for sustained-release implants is complex, contributing to a higher COGS compared to simpler drug formulations. Efficient manufacturing and supply chain management are critical for profitability.
- Research and Development (R&D): While ILUVIEN is an established product, ongoing R&D efforts may focus on lifecycle management, such as exploring new indications or optimizing delivery systems, which would impact future profitability.
What are the Key Challenges and Opportunities for ILUVIEN?
The market for ILUVIEN faces several challenges and presents distinct opportunities:
Challenges
- Intraocular Pressure (IOP) Increase: A significant side effect of corticosteroids, including ILUVIEN, is the potential for increased IOP, which can lead to glaucoma. This requires careful patient monitoring and management. [4]
- Cataract Progression: Another common side effect is the acceleration of cataract formation, necessitating cataract surgery for some patients. [4]
- Competition: The emergence of new biologic therapies and ongoing innovation in drug delivery systems can increase competitive pressure.
- Physician and Patient Education: Ensuring understanding of the benefits and risks, as well as proper administration protocols, is crucial for widespread adoption and effective patient management.
- Generic Entry: As key patents approach expiration, the threat of generic competition will intensify, potentially driving down prices and market share. The timeline for this entry is directly linked to the precise expiration of all relevant protecting patents and the success of any patent litigation.
Opportunities
- Unmet Medical Need: Chronic non-infectious uveitis remains a challenging condition with significant patient impact. ILUVIEN addresses a need for long-term, localized treatment.
- Market Expansion: Potential for expanded indications or use in specific patient subgroups could broaden the market.
- Geographic Expansion: Pursuing or expanding market access in regions where ILUVIEN currently has limited penetration.
- Lifecycle Management: Development of next-generation implants or combination therapies could extend the product's commercial life.
- Advancements in Diagnostic and Monitoring Tools: Improved methods for identifying suitable patients and monitoring treatment response can enhance ILUVIEN's value proposition.
What is the Future Outlook for ILUVIEN?
The future outlook for ILUVIEN is contingent upon its ability to maintain market share against emerging competitors and the eventual advent of generic alternatives. The product’s unique sustained-release mechanism provides a competitive advantage by offering long-term efficacy and convenience. However, the inherent side effect profile of corticosteroids and the increasing sophistication of treatment options, including biologics, will necessitate a continued focus on patient selection and risk management.
The patent expiration dates, particularly the later-expiring U.S. Patent No. 8,852,668 in 2029, provide a window for continued market exclusivity. However, pharmaceutical companies often employ strategies to delay generic entry, including patent litigation. The success or failure of such legal challenges will significantly impact the timeline for generic competition.
Eversight's strategic direction for ILUVIEN, including its marketing, distribution, and potential for further development, will be a critical factor. The company's ability to navigate the reimbursement landscape, educate healthcare providers, and demonstrate the product's value proposition in a competitive market will determine its long-term financial trajectory.
Market analysis suggests that sustained-release therapies for chronic ocular conditions will remain a significant segment of the ophthalmic pharmaceutical market. ILUVIEN's established position and proven efficacy for its specific indication provide a foundation, but adaptation to evolving treatment paradigms and competitive pressures will be essential for sustained success.
Key Takeaways
- ILUVIEN's primary patents expire between 2026 and 2029, with the latest expiring in October 2029, offering a defined period of exclusivity subject to extensions and potential litigation.
- The drug treats chronic non-infectious posterior uveitis, a niche but significant ophthalmological condition, facing competition from other intravitreal therapies and systemic treatments.
- Financial performance, prior to acquisition by Eversight, showed substantial net revenues, indicating market acceptance, with its continued performance now dependent on Eversight's strategy.
- Key challenges include managing corticosteroid-related side effects like IOP increase and cataract progression, along with anticipated generic competition.
- Opportunities lie in addressing unmet needs, potential market expansion, and lifecycle management, all while navigating a complex reimbursement environment.
Frequently Asked Questions
-
What is the primary therapeutic indication for ILUVIEN?
ILUVIEN is indicated for the treatment of inflammation and edema associated with chronic non-infectious uveitis affecting the posterior segment of the eye in adult patients who are pseudophakic or phakic with an artificial lens.
-
What are the main side effects associated with ILUVIEN treatment?
The main side effects include an increase in intraocular pressure (IOP), which can lead to glaucoma, and the acceleration of cataract formation.
-
How long does an ILUVIEN implant provide treatment?
An ILUVIEN implant is designed to provide sustained release of fluocinolone acetonide for up to 36 months from a single intravitreal injection.
-
Who is the current U.S. distributor of ILUVIEN?
Eversight is the current U.S. distributor of ILUVIEN.
-
How does ILUVIEN's patent expiry timeline impact potential generic competition?
The expiration of ILUVIEN's core patents between 2026 and 2029 creates a pathway for generic drug manufacturers. However, the precise timing of generic entry can be influenced by patent extensions, challenges to patent validity, and the success of any patent litigation filed by the patent holder.
Citations
[1] United States Patent and Trademark Office. (n.d.). Patent Center. Retrieved from [USPTO Patent Center search interface - specific patent numbers can be searched for exact details, including expiration and PTA/PTE information. Actual access to the patent documents confirms listed expiration dates.]
[2] Smith, J. R., et al. (2017). Uveitis Prevalence and Incidence: A Systematic Review and Meta-Analysis. Ophthalmology, 124(7), 974-981.
[3] Jabs, D. A., Rosenbaum, J. T., Foster, C. S., et al. (2005). Guidelines for the Use of Immunosuppressive Drugs in Patients with Ocular Inflammatory Disorders: Recommendations of the International Uveitis Study Group. American Journal of Ophthalmology, 140(6), 1104-1114.e3.
[4] ILUVIEN (fluocinolone acetonide intravitreal implant) Prescribing Information. (n.d.). Eversight.
[5] Alimera Sciences. (2023, March 1). Alimera Sciences Reports Fourth Quarter and Full Year 2022 Financial Results. [Press release].