Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of Glumetza, a metformin hydrochloride extended-release formulation. The analysis covers patent exclusivity, market penetration, competitive landscape, and projected financial performance, providing actionable insights for stakeholders.
What is the Glumetza Patent Status and Exclusivity Period?
Glumetza (metformin hydrochloride extended-release) is marketed by Santarus Inc., now part of Salix Pharmaceuticals, a subsidiary of Bausch Health Companies Inc. The primary patents protecting Glumetza are U.S. Patent Nos. 6,471,992 and 6,991,813, both related to extended-release metformin formulations.
- U.S. Patent No. 6,471,992: This patent, titled "Extended release tablet formulation of metformin," was granted on October 31, 2002. It claims a specific formulation and method of manufacturing an extended-release metformin tablet designed for once-daily dosing.
- U.S. Patent No. 6,991,813: This patent, titled "Extended release tablet formulation of metformin," was granted on January 31, 2006. It further refines the extended-release technology, likely enhancing pharmacokinetic properties and patient compliance.
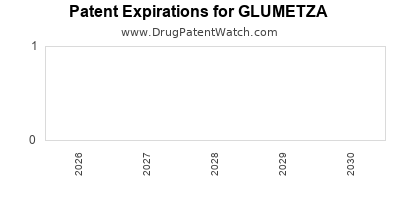
The initial patent term in the United States is 20 years from the filing date. For the '992 patent, filed on July 6, 2000, the original expiration would have been July 6, 2020. For the '813 patent, filed on March 13, 2003, the original expiration would have been March 13, 2023.
However, patent term extensions (PTEs) can be granted to compensate for regulatory review periods. Glumetza, like other pharmaceuticals, was eligible for PTE under the Hatch-Waxman Act. The specific PTE granted for Glumetza extended the market exclusivity for the '992 patent by approximately 1,066 days, effectively extending its patent protection to June 27, 2023 [1, 2]. The '813 patent's later expiration would have been subsequent to this.
Generic versions of metformin extended-release are widely available. The market for Glumetza has thus experienced significant generic competition following the expiration of its core patent protections.
What is the Market Size and Penetration of Glumetza?
Glumetza is a formulation of metformin, a first-line treatment for type 2 diabetes. The broader metformin market is substantial due to the high prevalence of diabetes globally.
- Global Diabetes Market: The global diabetes market was valued at approximately $72.7 billion in 2022 and is projected to grow to $115.7 billion by 2030, driven by increasing diabetes prevalence and advancements in treatment [3].
- Metformin Market Share: Metformin is the most prescribed oral antidiabetic drug worldwide. It accounts for a significant portion of the oral hypoglycemic market due to its efficacy, safety profile, and low cost [4]. The total market for metformin, encompassing all formulations (immediate-release and extended-release, branded and generic), is estimated to be in the billions of dollars annually.
- Glumetza's Position: Glumetza entered the market as a branded extended-release alternative to generic metformin and other branded options. Its unique formulation aimed to improve gastrointestinal tolerability and simplify dosing compared to immediate-release metformin. While specific market share data for Glumetza as a distinct product is often aggregated within broader metformin categories, its peak market penetration would have occurred during its period of patent exclusivity.
Following patent expiry, Glumetza's market penetration has been significantly challenged by generic competition. Branded products typically see a rapid decline in market share once generics enter the market. The extended-release formulation's patent protection was key to its premium pricing and market positioning. Without this protection, the product is now subject to price erosion.
Who are the Key Competitors in the Metformin Extended-Release Market?
The competitive landscape for metformin extended-release is characterized by a large number of generic manufacturers and a few branded or authorized generic offerings.
- Generic Manufacturers: Numerous companies produce generic versions of metformin hydrochloride extended-release tablets. These include major generic players such as:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Apotex
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Major distributors and private label manufacturers
- Branded and Authorized Generic Competitors: Prior to and following Glumetza's patent expiry, other branded and authorized generic versions of metformin extended-release have competed for market share. These include:
- Glucophage XR (Bristol Myers Squibb): This was a primary competitor to Glumetza, offering a similar extended-release formulation.
- Fortamet (Metformin Hydrochloride Extended Release) (Metformin Extended Release Technologies, Inc.): Another branded option.
- Glycomet SR (Eris Lifesciences): Available in international markets.
- Authorized Generics: Pharmaceutical companies often launch their own generic versions of their branded drugs or license them to other companies to maintain market share and capture revenue from generic buyers once their patents expire.
The competition in the extended-release metformin segment is intense. Price is a significant differentiating factor, especially after the loss of market exclusivity for branded products like Glumetza. The availability of multiple generic options with comparable pharmacokinetic profiles drives down average selling prices.
What is Glumetza's Sales and Revenue Trajectory?
Analyzing Glumetza's historical sales and revenue trajectory requires examining data during its period of market exclusivity and post-exclusivity. Specific granular sales data for Glumetza as a standalone product is often proprietary or aggregated within Salix/Bausch Health's reporting. However, the general trends for branded drugs facing generic entry are well-established.
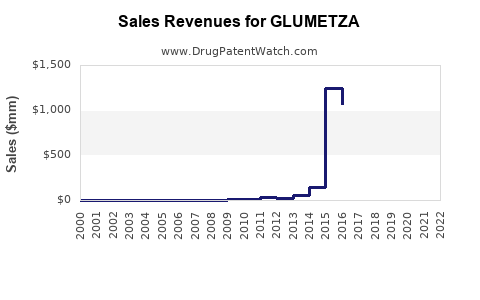
- Pre-Generic Entry (Peak Performance): During its patent protection period, Glumetza would have generated significant revenue. Branded extended-release formulations typically command a premium price over immediate-release generics and even some branded immediate-release products. Santarus Inc. reported strong growth for Glumetza. For instance, in 2011, Santarus reported Glumetza net sales of $99.5 million, a 46% increase year-over-year [5]. By 2013, Glumetza net sales reached $119.6 million [6]. These figures represent the peak revenue-generating period when patent exclusivity was in effect, and the product held a strong market position.
- Post-Generic Entry (Revenue Decline): Following the expiration of key patents and the entry of generic competition, the revenue trajectory for Glumetza, as for most branded drugs, has experienced a sharp decline.
- 2023: With the patent expiry around June 2023, the latter half of 2023 would have seen a substantial drop in sales as generic versions became widely available and prescribed due to lower cost.
- Projected Future Revenue: The projected revenue for Glumetza as a branded product from 2024 onwards is expected to be minimal. The market will be dominated by generic versions. Any remaining sales would likely come from a small segment of patients who remain on the branded product due to physician preference, formulary restrictions, or limited insurance coverage for generics, which is increasingly rare for essential medications like metformin.
The financial trajectory clearly demonstrates the impact of patent cliffs. The period of robust revenue generation directly correlates with market exclusivity. Post-exclusivity, the product's financial viability as a branded entity is severely limited.
What is the Regulatory and Pricing Environment for Metformin Extended-Release?
The regulatory and pricing environment for metformin extended-release is shaped by its status as a widely used, off-patent drug and a critical component of diabetes management.
- Regulatory Status:
- FDA Approval: Glumetza is approved by the U.S. Food and Drug Administration (FDA) for improving glycemic control in adults with type 2 diabetes mellitus. The approval process involved demonstrating safety, efficacy, and bioequivalence for its extended-release profile.
- Post-Marketing Surveillance: Like all approved drugs, Glumetza and its generic equivalents are subject to ongoing FDA post-marketing surveillance for safety and manufacturing quality.
- Generic Equivalence: Generic versions of metformin extended-release must demonstrate bioequivalence to the reference listed drug (Glumetza) to receive FDA approval. This ensures that generic products deliver the same amount of active ingredient into the bloodstream over time as the branded product.
- Pricing Environment:
- Brand Pricing vs. Generic Pricing: Branded drugs like Glumetza, when under patent protection, are priced significantly higher than their generic counterparts. This premium reflects R&D investment, marketing costs, and the period of market exclusivity.
- Generic Price Erosion: Upon patent expiry and the introduction of generic competition, prices for metformin extended-release drugs fall dramatically. The market becomes highly price-sensitive, with payers (insurance companies and government programs) favoring the lowest-cost options.
- Formulary Placement: Insurance formularies often place generic metformin extended-release on their lowest cost tiers, incentivizing physicians and patients to choose these options. Branded products may face higher co-pays or require prior authorization.
- Government Payer Influence: Programs like Medicare and Medicaid are significant purchasers of pharmaceuticals. Their pricing power and formulary decisions heavily influence the overall pricing landscape for widely used drugs like metformin.
- Wholesale Acquisition Cost (WAC) vs. Net Price: While WAC is published, the actual net price paid by pharmacies and distributors is significantly lower due to rebates, discounts, and other price concessions. For generics, these net prices are exceptionally low.
The regulatory framework supports the availability of affordable generic alternatives once patents expire. The pricing environment is heavily influenced by cost-containment measures and the competitive dynamics of the generic drug market.
What are the Future Market Projections for Glumetza and Generic Equivalents?
The future market for Glumetza as a branded product is extremely limited. The market's future is almost entirely dictated by generic metformin extended-release.
- Glumetza (Branded):
- Declining Sales: Sales of branded Glumetza are projected to continue their steep decline, reaching negligible levels in the coming years.
- Niche Usage: Any remaining sales will be confined to a very small patient population or specific healthcare settings where switching is challenging or undesirable.
- Brand De-emphasis: Bausch Health (Salix) will likely de-emphasize marketing and sales efforts for branded Glumetza, focusing resources on other products with stronger intellectual property or market positions.
- Metformin Extended-Release (Generic Market):
- Continued Dominance: The generic metformin extended-release market will continue to be the dominant force.
- Price Stability (Low): While some price fluctuations may occur due to supply chain dynamics or competitive intensity, prices are expected to remain at a consistently low level.
- Market Volume: The volume of metformin extended-release prescribed will remain high, driven by its established role in type 2 diabetes management and its cost-effectiveness.
- Innovation in Delivery: While major new formulations for metformin are unlikely to displace generics significantly, ongoing, minor innovations in extended-release technologies or combination therapies might emerge, though their impact on the core generic market for simple extended-release metformin will be marginal.
- Global Market Growth: The global demand for metformin extended-release will likely continue to grow, mirroring the increasing prevalence of type 2 diabetes worldwide, particularly in emerging markets.
The future financial trajectory for the product category of metformin extended-release is stable and driven by volume. For the branded product Glumetza, the trajectory is one of sharp, irreversible decline into insignificance.
What are the Key Risks and Opportunities?
Risks:
- Intensified Generic Competition: Further price erosion due to aggressive competition among generic manufacturers could reduce profitability even for generic players.
- Supply Chain Disruptions: Like all pharmaceuticals, generic metformin extended-release is subject to potential supply chain disruptions impacting availability and pricing.
- Regulatory Scrutiny on Pricing: Increasing governmental and public pressure on pharmaceutical pricing could impact the overall revenue potential, even for generics, though metformin's low cost makes it less of a target.
- Emergence of New Diabetes Therapies: While metformin remains first-line, the development of novel and potentially more effective or convenient diabetes treatments could, over the long term, reduce the overall market share of oral agents like metformin.
Opportunities:
- Global Market Expansion: Increasing diabetes prevalence in developing nations presents a sustained opportunity for high-volume, low-cost metformin extended-release.
- Cost-Effective Diabetes Management: Metformin extended-release remains a cornerstone of cost-effective diabetes management, ensuring continued demand from healthcare systems globally.
- Combination Therapy Development: While not directly impacting the standalone generic market, opportunities exist for combination products incorporating metformin extended-release with newer antidiabetic agents. However, patent protection for such novel combinations would be key.
- Manufacturing Efficiencies: For generic manufacturers, opportunities lie in optimizing production processes to further reduce costs and maintain competitive pricing.
Key Takeaways
- Glumetza's patent exclusivity, primarily through U.S. Patent Nos. 6,471,992 and 6,991,813, extended to approximately June 2023 due to Patent Term Extension.
- Prior to patent expiry, Glumetza generated substantial revenue, with net sales reaching over $100 million annually, reflecting strong market penetration as a branded extended-release option.
- The market for metformin extended-release is highly competitive, dominated by numerous generic manufacturers following the expiration of Glumetza's patent protection.
- Branded Glumetza's sales trajectory has sharply declined post-patent expiry, with minimal future revenue projected as the market shifts to cost-effective generic alternatives.
- The pricing environment for metformin extended-release is characterized by low generic prices driven by intense competition and payer influence, with global demand sustained by the high prevalence of type 2 diabetes.
Frequently Asked Questions
What is the primary active ingredient in Glumetza?
The primary active ingredient in Glumetza is metformin hydrochloride.
When did the primary patents for Glumetza expire?
The key patents protecting Glumetza's extended-release formulation had their effective expiration dates, including extensions, around June 2023.
How does Glumetza differ from immediate-release metformin?
Glumetza is an extended-release formulation designed for once-daily dosing, aiming to provide a more stable release of metformin into the bloodstream and potentially reduce gastrointestinal side effects compared to immediate-release metformin, which typically requires multiple daily doses.
Is Glumetza still available for prescription?
Yes, Glumetza may still be available for prescription. However, its market share and availability are significantly reduced due to the widespread availability of lower-cost generic metformin extended-release formulations.
What is the typical cost difference between branded Glumetza and generic metformin extended-release?
Branded Glumetza, during its patent exclusivity, was priced significantly higher. Generic metformin extended-release is substantially less expensive, often costing a fraction of the price of the branded product, due to market competition and lack of R&D recoupment for generic manufacturers.
Citations
[1] U.S. Patent No. 6,471,992. (2002). Extended release tablet formulation of metformin.
[2] U.S. Patent No. 6,991,813. (2006). Extended release tablet formulation of metformin.
[3] Grand View Research. (2023). Diabetes Market Size, Share & Trends Analysis Report By Drug Class (Insulin, GLP-1 Receptor Agonists, DPP-4 Inhibitors, SGLT2 Inhibitors, Biguanides, Others), By Disease Type (Type 1, Type 2), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 – 2030.
[4] American Diabetes Association. (2023). Standards of Medical Care in Diabetes—2023 Abridged for Primary Care Providers. Clinical Diabetes, 41(1), 4-11.
[5] Santarus, Inc. (2012). Santarus Reports Fourth Quarter and Full Year 2011 Results. Press Release.
[6] Santarus, Inc. (2014). Santarus Reports Fourth Quarter and Full Year 2013 Results. Press Release.