Last updated: February 19, 2026
Executive Summary
Glucophage (metformin hydrochloride) remains a cornerstone therapy for type 2 diabetes mellitus, demonstrating sustained market presence due to its efficacy, safety profile, and cost-effectiveness. The drug's market trajectory is characterized by significant generic competition following patent expirations, leading to price erosion but maintaining high prescription volumes. Financial performance is driven by broad market penetration and continued physician preference.
GLUCOPHAGE: Market Position and Competitive Landscape
Metformin, the active pharmaceutical ingredient in Glucophage, is a first-line oral antidiabetic agent recommended by major clinical guidelines, including those from the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) [1]. Its efficacy in lowering hemoglobin A1c (HbA1c) levels by an average of 1% to 1.5% is well-established [2]. The drug functions by decreasing hepatic glucose production, reducing intestinal glucose absorption, and improving insulin sensitivity [3].
The original patent for Glucophage expired in 2000. This led to the introduction of numerous generic versions, dramatically altering the market landscape. The branded Glucophage product, manufactured by Bristol Myers Squibb (now part of Bristol Myers Squibb), faces competition from multiple generic manufacturers.
Key Market Participants for Metformin:
- Branded: Glucophage (Bristol Myers Squibb)
- Generic Manufacturers: Teva Pharmaceuticals, Mylan N.V. (now Viatris), Sun Pharmaceutical Industries, Aurobindo Pharma, and numerous others.
The competitive intensity in the metformin market is high, characterized by a large number of suppliers offering the drug at significantly lower price points than the original branded product. This has resulted in substantial price reductions for metformin hydrochloride.
GLUCOPHAGE: Sales Performance and Revenue Generation
The financial trajectory of Glucophage, particularly the branded product, has been influenced by its patent status and the subsequent genericization. While precise, up-to-date revenue figures specifically for the branded Glucophage product are not always granularly disclosed by Bristol Myers Squibb due to its mature status and generic competition, industry data provides insight into the overall metformin market.
Historical and Current Revenue Trends:
- Pre-Patent Expiration: Branded Glucophage commanded significant market share and revenue, operating in a less competitive environment.
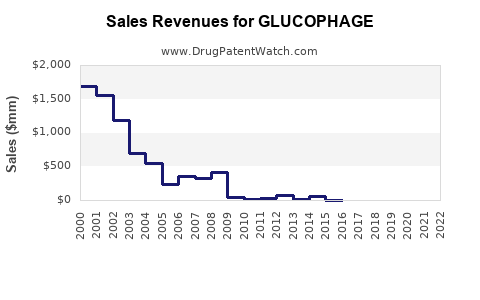
- Post-Patent Expiration (2000 onwards): Revenue for branded Glucophage experienced a sharp decline as generic alternatives entered the market.
- Current Market: The overall metformin market, including both branded and generic formulations, remains substantial in terms of volume due to its widespread use. However, revenue for the branded product is now a fraction of its peak, while generic sales drive the majority of the dollar value within the broader metformin market.
Market Size for Metformin (Global, Estimated):
- The global market for antidiabetic drugs, including metformin, is projected to reach over $60 billion by 2025 [4]. Metformin constitutes a significant portion of this market, primarily due to its high volume prescription rates.
- While exact figures for branded Glucophage are proprietary, the total metformin market (including generics) is estimated to generate billions of dollars annually. For instance, in 2019, the U.S. prescription drug spending report indicated that metformin was among the top 50 drugs by expenditure, with significant volume contributing to its overall economic impact [5].
The financial model for branded Glucophage has transitioned from high-margin exclusivity to volume-driven sales in a highly competitive generic market. Bristol Myers Squibb continues to market Glucophage, leveraging established brand recognition and physician familiarity.
GLUCOPHAGE: Prescription Data and Patient Access
Metformin is one of the most prescribed medications globally for type 2 diabetes. Its accessibility is further enhanced by its inclusion on formularies and its low cost, making it a preferred choice for healthcare systems and patients alike.
Prescription Volume Trends:
- High Volume: Metformin consistently ranks among the most frequently prescribed drugs in outpatient settings across major markets like the United States [5].
- Sustained Demand: Despite the availability of newer antidiabetic agents with novel mechanisms of action (e.g., SGLT2 inhibitors, GLP-1 receptor agonists), metformin's role as a first-line therapy ensures consistent, high prescription volumes.
- Geographic Penetration: Metformin is prescribed across all major geographic regions, with particularly high utilization in North America, Europe, and Asia.
Factors Influencing Prescription:
- Clinical Guidelines: Endorsement by leading diabetes organizations.
- Cost-Effectiveness: Significantly lower cost compared to newer agents, especially for generic versions.
- Safety Profile: Decades of clinical experience have established a well-understood safety profile.
- Efficacy: Proven ability to manage blood glucose levels.
- Combination Therapy: Widely used in combination with other antidiabetic agents.
The widespread adoption of metformin, including Glucophage, directly translates to substantial patient access and a continuous demand stream for the medication.
GLUCOPHAGE: Regulatory and Intellectual Property Status
The regulatory and intellectual property landscape for Glucophage has evolved significantly since its initial market introduction.
Regulatory Approvals:
- Glucophage was first approved by the U.S. Food and Drug Administration (FDA) in 1994.
- It has since received approvals in numerous other countries through national regulatory agencies.
- Extended-release formulations (e.g., Glucophage XR) were developed and approved to improve patient adherence and gastrointestinal tolerability, offering a differentiation point for the branded product before generic competition emerged for these formulations as well.
Intellectual Property:
- Primary Patents: The original composition of matter and use patents for metformin hydrochloride have long expired.
- Formulation Patents: Patents covering specific formulations, such as extended-release versions, may have had longer protection periods but have also largely expired or are nearing expiration in key markets.
- Generic Entry: The expiration of these key patents has allowed for widespread generic manufacturing and marketing. Regulatory agencies have procedures in place to facilitate the approval of generic versions once patent exclusivity is removed.
The absence of significant patent protection for the core metformin molecule means that the market for Glucophage is now characterized by generic competition, with limited opportunity for new patent-driven market exclusivity for the active ingredient itself.
GLUCOPHAGE: Future Outlook and Market Trends
The future market trajectory for Glucophage is expected to remain consistent with its current status: a high-volume, cost-effective, and widely prescribed medication.
Projected Market Dynamics:
- Sustained Prescription Volume: Metformin will continue to be a first-line therapy for type 2 diabetes due to its established clinical benefits and cost advantages.
- Price Pressure: Intense competition among generic manufacturers will maintain downward pressure on prices, particularly for generic metformin. Branded Glucophage will likely continue to occupy a niche, potentially commanding a slight premium for brand recognition or specific patient/physician preferences, but its market share will remain limited compared to generics.
- Competition from Newer Agents: While newer classes of antidiabetic drugs offer benefits beyond glycemic control (e.g., cardiovascular and renal protection), their higher cost often restricts their use as first-line therapy, especially in resource-constrained settings. Metformin is frequently used in combination with these newer agents, further solidifying its role.
- Reformulations and Combination Products: The market may see continued innovation in fixed-dose combination products that include metformin with other antidiabetic agents, though patent protection for such combinations would be tied to the newer, patented active ingredients.
- Biosimilarity Not Applicable: As a small molecule drug, metformin does not have biosimilars; the relevant term is generic.
The market for Glucophage and generic metformin is mature. Growth will primarily be driven by the increasing global prevalence of type 2 diabetes rather than market share expansion against competing drug classes or significant price increases for the branded product.
Key Takeaways
- Glucophage (metformin hydrochloride) maintains a dominant position as a first-line treatment for type 2 diabetes.
- Patent expirations have led to widespread generic competition, resulting in significant price erosion for the drug.
- The financial performance of branded Glucophage is now largely volume-driven within a highly competitive generic market.
- High prescription volumes persist due to clinical guidelines, cost-effectiveness, and a well-established safety profile.
- The future market trajectory is characterized by sustained demand, price pressure from generics, and continued use in combination with newer antidiabetic therapies.
Frequently Asked Questions
- What is the primary mechanism of action for Glucophage (metformin)?
Glucophage primarily functions by reducing hepatic glucose production, decreasing intestinal glucose absorption, and enhancing peripheral insulin sensitivity [3].
- When did the primary patents for Glucophage expire, allowing for generic competition?
The original patents for metformin hydrochloride expired around the year 2000, paving the way for generic versions of Glucophage to enter the market [1].
- How does Glucophage compare in cost to newer antidiabetic medications?
Glucophage, especially its generic formulations, is significantly more cost-effective than newer classes of antidiabetic drugs, such as SGLT2 inhibitors and GLP-1 receptor agonists [4].
- Is Glucophage still recommended as a first-line treatment for type 2 diabetes?
Yes, Glucophage remains a recommended first-line oral antidiabetic agent by major medical associations like the American Diabetes Association and the European Association for the Study of Diabetes due to its efficacy and safety profile [1].
- Are there any significant safety concerns associated with long-term use of Glucophage?
While generally safe, the most significant risk associated with metformin is lactic acidosis, though this is rare. Vitamin B12 deficiency can also occur with long-term use [2].
Citations
[1] American Diabetes Association. (2023). Standards of Care in Diabetes—2023. Diabetes Care, 46(Supplement_1), S1-S291.
[2] NICE. (2022). Type 2 diabetes in adults: management. NICE Guideline [NG28].
[3] Hundal, R., & Bhansali, S. (2020). Metformin: A comprehensive review. Diabetology & Metabolic Syndrome, 12(1), 96.
[4] Global Market Insights. (2021). Diabetes drugs market size to cross USD 60 billion by 2025. Retrieved from https://www.gminsights.com/pressrelease/diabetes-drugs-market
[5] IQVIA. (2020). The U.S. Pharmaceutical Market: 2019. Retrieved from https://www.iqvia.com/