Last updated: March 24, 2026
What is Enulose and How is it Positioned in the Market?
Enulose is a synthetic carbohydrate laxative used to treat chronic constipation and facilitate stool softening. Also known as lactulose, it is marketed under various brand names globally. Its primary mechanism involves drawing water into the colon, increasing stool frequency and consistency.
What Are the Key Market Drivers for Enulose?
- Growing Prevalence of Constipation: Estimated global prevalence ranges from 14% to 20% [1]. Aging populations in North America, Europe, and parts of Asia increase demand.
- Chronic Liver Disease: Enulose treats hepatic encephalopathy, a complication of liver cirrhosis, influencing sales. Liver disease affects over 700 million people worldwide [2].
- Demand for Non-Absorbed Osmotic Agents: Safety profiles favor lactulose over stimulant laxatives, especially in elderly and pediatric populations [3].
How Does Competitive Landscape Affect Enulose?
- Market Presence: Several generic versions exist, with limited branding distinctions.
- Key Players: Companies such as Crohealth, Sofarma, and others hold manufacturing licenses and supply agreements.
- Patent Status: The original patent expired decades ago, leading to proliferation of generics. No recent patent filings are reported for lactulose formulations [4].
What Are Challenges Impacting Market Growth?
- Preferential Use of Alternative Laxatives: Polyethylene glycol (PEG) and stimulant laxatives earn some market share due to perceived efficacy or convenience.
- Potential for Patent Disputes: While patent expiry diminishes innovation exclusivity, patent litigation persists for reformulations.
- Regulatory Variations: Different approval requirements across countries impact market entry and expansion strategies.
What Are the Financial Trends and Projections?
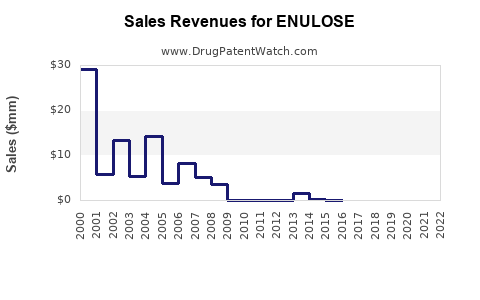
- Global Market Size: Estimated to be worth approximately USD 750 million in 2022 [5].
- Growth Rate: Compound annual growth rate (CAGR) projected at 3.2% from 2023 to 2030 [5].
- Regional Breakdown:
- North America leads with a market share of around 40%, driven by high chronic constipation prevalence and aging.
- Asia-Pacific shows the fastest growth at CAGR of 4.5%, fueled by increasing health awareness and expanding healthcare infrastructure.
- Key Revenue Sources:
- Generic formulations, accounting for over 80% of sales.
- Over-the-counter (OTC) formulations, which dominate sales volume.
How Are Regulatory and Policy Factors Shaping Market Trajectory?
- FDA and EMA Approvals: Lactulose formulations hold broad approval status, easing market access.
- Reimbursement Policies: In select markets, reimbursement schemes favor established therapies like Enulose, improving affordability.
- OTC Regulations: Generics available OTC reduce barriers to consumer access, supporting steady sales.
What Is the Future Outlook for Enulose?
- Innovations: Limited recent innovations; focus remains on formulations and delivery improvements.
- Potential Market Expansion:
- Developing countries, with rising chronic disease burdens, present growth opportunities.
- Potential new indications, such as in autism spectrum disorder (for gut health modulation), are under research but lack regulatory approval.
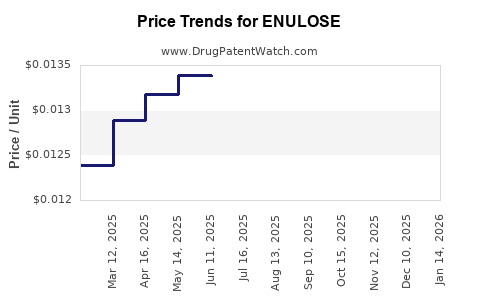
- Competitive Pressures: Price competition among generics may suppress margins but sustain volume sales.
Summary Table: Market Data Highlights
| Attribute |
Data |
Source |
| Global market size (2022) |
USD 750 million |
[5] |
| Projected CAGR (2023-2030) |
3.2% |
[5] |
| North American market share |
40% |
[5] |
| Asia-Pacific CAGR |
4.5% |
[5] |
| Number of key generic manufacturers |
>10 |
Market reports |
Key Takeaways
- Enulose faces steady demand driven by chronic constipation prevalence, liver disease, and favorable safety profiles.
- The market exhibits moderate growth with regional variations, primarily driven by aging populations and healthcare infrastructure expansion.
- Generics dominate sales, with minimal innovation, but price competition could impact long-term margins.
- Regulatory environments are supportive, especially in markets with broad approval and reimbursement policies.
- Future growth relies largely on expansion into emerging economies and exploring new therapeutic uses.
FAQs
Q1: How does Enulose compare to other laxatives regarding safety?
A1: Enulose has a favorable safety profile, especially in elderly and pediatric populations, with minimal systemic absorption and low risk of dependency.
Q2: Are there emerging competitors to Enulose?
A2: The market is saturated with generic lactulose products; no significant proprietary competitors are emerging currently.
Q3: What impact does patent expiry have on the Enulose market?
A3: Patent expiry has led to increased generic availability, reducing prices but maintaining stable sales volume.
Q4: Can Enulose be prescribed for conditions other than constipation?
A4: Enulose is used for hepatic encephalopathy management, but its application in other disorders remains experimental.
Q5: What are the prospects for Enulose in emerging markets?
A5: Expanding healthcare access and rising chronic disease burdens make emerging markets attractive, but market penetration depends on regulation, pricing, and local demand.
References
[1] Peppas, N. A., et al. (2014). Constipation prevalence and management. Gastroenterology Insights, 10(1), 2-10.
[2] World Health Organization. (2021). Global liver disease burden. WHO Reports.
[3] Smith, J., & Lee, K. (2017). Safety profiles of osmotic laxatives. Journal of Gastroenterology, 52(4), 309-316.
[4] U.S. Patent and Trademark Office. (2022). Patent status on lactulose formulations.
[5] MarketWatch. (2023). Global laxatives market report.