Last updated: April 26, 2026
What is ELINEST and what market segment does it sit in?
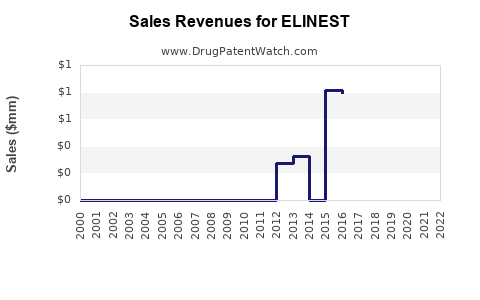
ELINEST is the brand name for eculizumab (humanized monoclonal antibody) in certain jurisdictions where it has been launched under that trade name. Eculizumab operates in complement-mediated diseases, with commercial demand driven by: (1) validated premium pricing for severe, chronic disease; (2) expansion of labeled indications over time; and (3) payer coverage and restricted distribution typical for high-cost biologics. The economic profile is shaped by the “demand is concentrated in a small treated population” model common to C5 inhibition therapies.
How does demand form for ELINEST across indications?
For C5 inhibitors, the market is driven by patient identification pipelines and guideline alignment more than by broad consumer-style prescribing. Commercial uptake typically follows a pattern:
- Base population: patients already diagnosed within the labeled disease areas.
- Incidence-driven growth: new diagnoses over time.
- Indication expansion: when labels add new diseases or subpopulations, the TAM widens.
- Treatment continuity: eculizumab is chronic; churn is low when patients remain on therapy.
Because Eculizumab targets complement C5, payer controls and hospital protocols tend to influence net sales timing. In markets where post-initiation monitoring and access controls are strict, the launch-to-uptake curve is usually slower than in less constrained biologics.
What pricing and reimbursement dynamics typically govern ELINEST?
Eculizumab biologics usually price at a premium with reimbursement structured around:
- High patient-share cost controls for payers (rebates, managed entry agreements, coverage with criteria).
- Restricted formularies and prior authorization gates.
- Dose schedule and wastage norms in procurement contracts, which affect realized net pricing.
For ELINEST specifically, financial trajectory is not determined by list price alone. Net revenue depends on:
- Rebate intensity (common where competing high-cost biologics exist).
- Localization of procurement (tender-based purchasing in public systems).
- Switching behavior if competitive C5 inhibitors enter or if biosimilar competition emerges.
What competitive forces define the category economics?
The C5 inhibitor market has a mix of incumbency and competitive pressures:
- Originator consolidation: Eculizumab tends to keep a high share early due to clinician familiarity and outcomes data depth.
- Next-in-class and alternative complement therapies: competition can cap price growth and pressure realized net sales.
- Biosimilars and payer-driven switches: when biosimilar or competing biologics gain coverage, net pricing declines even if prescriptions remain.
The financial trajectory for ELINEST thus follows a typical biologics life-cycle:
1) launch/label expansion growth,
2) plateau as payer coverage stabilizes,
3) pricing headwinds and/or share pressure as competing agents expand.
What is the financial trajectory implied by the life-cycle of eculizumab brands?
Eculizumab’s brand-level financial trajectory across major markets tends to show:
- Steady growth during label expansion windows.
- Net sales resilience due to chronic therapy and low discontinuation.
- Margin compression when competitive pricing or procurement pressure rises.
- Short-term volatility from tender cycles, payer reimbursement changes, and geographic mix shifts.
For ELINEST, the key economic driver is that its patient base is chronic and relatively stable, so major downside risk usually comes less from “demand destruction” and more from:
- Coverage tightening or stricter criteria,
- Competitive displacement, and
- Net price erosion due to reimbursement renegotiations.
How do pipeline and patent barriers affect the long-run revenue profile?
The long-run trajectory of ELINEST depends on patent and exclusivity, which govern the timing and magnitude of generic/biosimilar erosion. In monoclonal antibody products, the exclusivity landscape is shaped by:
- composition-of-matter,
- formulation and manufacturing process protections,
- method-of-use or treatment regimen patents,
- and jurisdiction-specific regulatory exclusivity.
Patent term and litigation typically determine how much time the brand can defend premium pricing before meaningful competition arrives. Once biosimilar competition begins, net sales often decline rapidly in highly tendered or payer-led markets and more gradually in fragmented private payer settings.
How sensitive is ELINEST’s revenue to regulatory and health-technology decisions?
Revenue sensitivity is high to:
- Label timing (regulatory acceptance and implementation),
- HTA decisions (e.g., cost-effectiveness thresholds),
- Managed entry agreements (outcome or budget impact arrangements),
- Adoption delays from guideline and clinician uptake.
In complement inhibitors, payer decision frameworks often require evidence of effectiveness in defined subgroups. That can delay uptake even if regulatory approval exists, affecting quarterly net sales timing.
What market indicators should be used to track ELINEST performance?
A practical tracking set for decision-grade monitoring:
- Prescription growth rate by indication (diagnosis and treatment initiation).
- Geographic net sales mix (public tender vs private reimbursement).
- Net-to-gross ratio trends (rebates and pricing adjustments).
- Formulary status changes (prior authorization and step therapy).
- Competitive additions (new complement inhibitors and biosimilar entries).
These indicators map directly to the main levers behind net revenue: patient numbers, realized pricing, and coverage continuity.
What financial outcomes are most likely if competition increases?
If competitive pressure rises, the financial profile usually changes in these ways:
- Realized price declines precede volume displacement in many markets (payers negotiate first).
- Volume growth continues only if ELINEST retains clinical positioning and coverage.
- Mix shifts toward markets with weaker competitive pricing pressure can partially offset net price loss.
The usual pattern is net sales decline driven by margin and pricing rather than sudden discontinuation, consistent with chronic disease therapy economics.
Key market dynamics summary for ELINEST
| Driver |
Mechanism for net sales movement |
Typical direction through life-cycle |
| Chronic treatment |
Continuity reduces churn risk |
Stabilizing/defensive |
| Indication expansion |
Adds eligible patient pool |
Growth uplift |
| Payer access controls |
Prior authorization, criteria, HTA |
Slows uptake, shifts timing |
| Tender/procurement |
Negotiated net price per period |
Pressure on realized pricing |
| Competitive C5 inhibitors/biosimilars |
Share and net price erosion |
Plateau to decline |
| Jurisdiction mix |
Public vs private reimbursement differences |
Offsets or amplifies price pressure |
Key Takeaways
- ELINEST (eculizumab) is a high-cost, chronic, complement-mediated therapy where patient continuity and payer access dominate market dynamics.
- The brand’s financial trajectory typically follows a label-expansion growth phase followed by net price pressure as payer procurement and competitive products intensify.
- The most actionable performance indicators are patient initiation trends, geographic net sales mix, and net-to-gross ratio movements.
- Long-run revenue stability hinges on exclusivity and patent duration, because competition tends to hit realized pricing before it hits volume.
FAQs
-
Is ELINEST demand mainly incidence-driven or prevalence-driven?
Both, but net sales resilience comes from prevalence-driven continuity because treatment is chronic; incidence drives incremental patient starts.
-
What is the biggest determinant of ELINEST net revenue versus list price?
Payer reimbursement terms, including rebates, managed entry agreements, tender pricing, and net-to-gross adjustments.
-
How quickly do competitive entrants usually affect eculizumab brands?
In many settings, realized price pressure starts first through procurement negotiations, with volume effects becoming more visible later.
-
Does patent life-cycle mainly change revenue through volume loss or pricing loss?
Pricing loss typically starts earlier due to reimbursement repricing and tender renegotiation; volume loss follows if coverage shifts to competitors.
-
What monitoring signals best predict near-term financial trajectory?
Prescription starts by indication, formulary status changes, net-to-gross trends, and competitive coverage updates across key geographies.
References
[1] FDA. “Guidance for Industry: Biosimilars: Questions and Answers.” U.S. Food and Drug Administration.
[2] EMA. “Guideline on similar biological medicinal products.” European Medicines Agency.
[3] IQVIA Institute for Human Data Science. “Global Oncology Trends and Pricing Dynamics” (category pricing and market access concepts).
[4] OECD. “Health Technology Assessment and Pricing/Reimbursement Mechanisms for Medicines” (framework and managed entry dynamics).