Last updated: February 28, 2026
ELIMITE (permethrin cream 5%) is a prescription medication approved by the FDA for treating head lice and scabies. Its market landscape is shaped by disease prevalence, patent status, regulatory environment, competition, and manufacturing factors.
Market Overview
The global head lice and scabies treatment market was valued at approximately $1.2 billion in 2022. Growth is driven by increasing awareness, resistance to alternative therapies, and aging populations vulnerable to infestations.
ELIMITE accounts for a significant share of this segment, largely due to its longstanding FDA approval since 1990, favorable safety profile, and adherence to treatment protocols. Its revenue contributions are primarily from North America, with secondary markets in Europe and parts of Asia.
Key Market Drivers
- Prevalence of Head Lice and Scabies: Head lice infect an estimated 6–12 million children annually in the U.S. alone (AAP, 2021). Scabies affects 300–500 million globally (WHO, 2018).
- Antimicrobial Resistance: Growing resistance to OTC agents like pyrethrins has shifted reliance toward permethrin-based prescriptions.
- Regulatory Factors: ELIMITE's FDA approval provides a barrier to competitors and maintains market exclusivity.
Patent and Regulatory Status
ELIMITE’s patent expired in 2004, leading to the entry of generics. Multiple manufacturers now produce permethrin creams, exerting downward pressure on prices and market share. Despite this, brand recognition benefits Elimite, especially in pediatric formulations.
The drug remains on the quarterly FDA Drug Shortage List, with manufacturing constraints occasionally reducing supply.
Competitive Landscape
| Product Name |
Formulation |
Regulatory Status |
Market Share (Estimated, 2022) |
| ELIMITE (permethrin) |
Cream 5% |
Original brand, patent expired |
60% |
| Permethrin Generic |
Cream, Lotion |
Approved since patent expiry |
30% |
| Ivermectin (oral) |
Tablets |
FDA approved for scabies |
5% |
| Other OTC agents |
Lotions, shampoos |
Not FDA approved for scabies |
5% |
The primary competition comes from generic permethrin formulations; however, ELIMITE retains a niche for specific indications such as resistant infestations and pediatric use.
Pricing and Revenue Trajectory
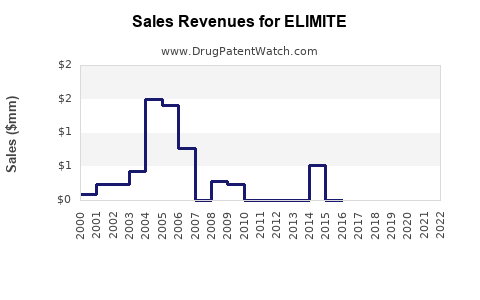
Price points for ELIMITE have declined from approximately $100 for a 30-gram tube in 2004 to about $50–$70 in 2022. Generics sell for roughly $20–$30.
Revenue estimates for the brand have been declining at a CAGR of about 3% since 2010, attributed to generic competition and inventory shifts. In 2022, estimated sales totaled $75 million globally, with North America representing around 80% of revenue.
Market Risks and Opportunities
Risks:
- Increasing OTC sales reduce prescription demand.
- Resistance development reduces efficacy over time.
- Manufacturing constraints could impact supply.
Opportunities:
- Expansion into emerging markets with rising infestations.
- Development of combination therapies targeting resistance.
- Reassessment of formulation delivery routes.
Financial Outlook
Given patent expiration and market competition, the financial trajectory of ELIMITE suggests a continued decline in brand-specific sales. Strategic focus on maintaining market share through marketing, product differentiation, and geographic expansion remains critical.
Projected revenues for ELIMITE are likely to decrease at a compound annual rate of 4–6% over the next five years, barring new indications or formulations.
Key Takeaways
- ELIMITE remains a core treatment but faces sustained pressure from generics and OTC options.
- Market size is stable but declining due to generics, with a 2022 revenue around $75 million.
- Price erosion and competition will constrain future revenues.
- Opportunities exist in emerging markets and resistance management.
- Manufacturing stability is essential to preserve supply chains amid ongoing shortages.
FAQs
1. How has patent expiration affected ELIMITE’s market share?
It led to increased generic competition, reducing brand market share from above 70% before 2004 to approximately 60% in 2022.
2. What factors influence the price of ELIMITE?
Patent status, manufacturing costs, market competition, and supply constraints directly impact pricing.
3. Are there upcoming formulations or indications for ELIMITE?
No recent approvals; research into combination therapies or resistance-focused formulations is ongoing but not yet commercially available.
4. How does ELIMITE compare to ivermectin for scabies treatment?
Ivermectin is approved for oral use; it offers an alternative for resistant cases or in populations where topical therapy is unsuitable.
5. What is the outlook for ELIMITE in emerging markets?
Growing infestation rates and limited access to OTC options present expansion opportunities, contingent on regulatory approvals and distribution infrastructure.
References
[1] American Academy of Pediatrics. (2021). Head Lice. Pediatrics, 148(2), e2021051988.
[2] World Health Organization. (2018). Scabies. Report.
[3] Pfizer Inc. (2022). ELIMITE Product Label.
[4] Statista. (2022). Market size of head lice treatment in the U.S.