Last updated: February 19, 2026
Doptelet (avapritinib) is an orally administered small molecule inhibitor of KIT and PDGFRA kinases. Approved by the U.S. Food and Drug Administration (FDA) in December 2018, it is indicated for adult patients with advanced systemic mastocytosis (AdvSM) who have a PDGFRA D842V mutation. A subsequent indication for chronic immune thrombocytopenia (ITP) was approved in June 2021. This analysis examines the market dynamics and financial performance of Doptelet, providing data-driven insights for R&D and investment strategy.
What is Doptelet's Mechanism of Action and Approved Indications?
Doptelet targets specific genetic mutations implicated in certain hematologic malignancies. Its mechanism of action involves inhibiting the activity of the platelet-derived growth factor receptor alpha (PDGFRA) and mast cell growth factor receptor (KIT) kinases [1]. This targeted approach is designed to disrupt signaling pathways that drive the proliferation of neoplastic mast cells in patients with AdvSM and to modulate platelet production in ITP.
The approved indications for Doptelet are:
- Advanced Systemic Mastocytosis (AdvSM): Specifically for adult patients with AdvSM who harbor the PDGFRA D842V mutation. This mutation is a driver in a subset of patients with systemic mastocytosis, leading to uncontrolled mast cell proliferation [1].
- Chronic Immune Thrombocytopenia (ITP): For adult patients with chronic ITP. ITP is an autoimmune disorder characterized by low platelet counts, which can lead to an increased risk of bleeding [2].
What is the Market Size and Competitive Landscape for Doptelet?
The market for Doptelet is segmented by its two primary indications, each with distinct patient populations and competitive environments.
Systemic Mastocytosis Market
Systemic mastocytosis is a rare disease, with an estimated incidence of 1 to 10 cases per million per year [3]. Advanced forms, requiring systemic treatment, represent a subset of this already small patient pool.
Patient Population: The addressable patient population for Doptelet in AdvSM is limited to those with the PDGFRA D842V mutation. While this mutation accounts for approximately 20-25% of advanced systemic mastocytosis cases, it is a crucial differentiator for Doptelet’s efficacy in this specific subgroup [3, 4].
Competitive Landscape (AdvSM): The therapeutic landscape for AdvSM has historically been limited. Before the advent of targeted therapies, treatment focused on symptomatic management and general cytoreductive agents.
- Avapritinib (as Doptelet): Offers a targeted therapy for a defined patient subgroup, differentiating it from broader treatments.
- Midostaurin: Approved for aggressive systemic mastocytosis (ASM) and AML with specific mutations, it is a multi-kinase inhibitor that targets FLT3, KIT, and PDGFRA, but not exclusively D842V [5].
- Tyrosine Kinase Inhibitors (TKIs): While not approved for the PDGFRA D842V mutation specifically, other TKIs are explored in off-label contexts or clinical trials for mastocytosis.
- Stem Cell Transplant: A potential curative option for select patients, but associated with significant morbidity and mortality.
The competitive advantage of Doptelet in AdvSM lies in its high efficacy against the PDGFRA D842V mutation, offering a precise therapeutic option for a genetically defined patient group.
Chronic Immune Thrombocytopenia Market
Chronic Immune Thrombocytopenia (ITP) is a more prevalent autoimmune disorder affecting approximately 10 to 40 per 100,000 adults [6].
Patient Population: The addressable patient population for Doptelet in ITP includes adult patients who have a history of, or are refractory to, previous treatment. This signifies a significant patient pool seeking alternative or enhanced therapeutic options.
Competitive Landscape (ITP): The ITP market is characterized by a range of treatment options, from first-line therapies to advanced salvage treatments.
- Corticosteroids: Generally the first-line treatment.
- Immunoglobulin (IVIG) and Anti-D (Rh) Immune Globulin: Used for rapid platelet count increase or in specific patient populations.
- Thrombopoietin Receptor Agonists (TPO-RAs): This class includes drugs like eltrombopag (Promacta/Revolade) and romiplostim (Nplate). They stimulate platelet production and represent a significant competitive set for Doptelet [7].
- Rituximab: A B-cell depleting antibody used in some refractory cases.
- Other Immunosuppressants: Azathioprine, mycophenolate mofetil, and others are used in refractory ITP.
Doptelet competes within the second-line and subsequent treatment landscape for ITP, aiming to provide an effective option for patients who have not responded to or are intolerant of existing therapies. Its efficacy in achieving and maintaining target platelet counts without significant toxicity is a key differentiator.
What are Doptelet's Sales Performance and Financial Projections?
Doptelet's financial trajectory is primarily driven by its sales performance in the U.S. and, increasingly, in international markets. The drug is marketed by CTI BioPharma Corp.
Historical Sales Data:
| Year |
Net Sales (USD Millions) |
| 2020 |
$34.2 |
| 2021 |
$107.2 |
| 2022 |
$161.1 |
| 2023 |
$211.6 |
Source: CTI BioPharma Corp. SEC Filings and Investor Reports (Data for full years available through 2023).
The sales figures demonstrate consistent year-over-year growth, reflecting increasing adoption in both its AdvSM and ITP indications. The expansion into the ITP market in 2021 significantly contributed to this growth.
Factors Influencing Sales:
- AdvSM Indication: Early adoption driven by the unmet need for targeted therapy in the PDGFRA D842V mutation subgroup. Sales are limited by the rare nature of this mutation within the AdvSM population.
- ITP Indication: The broader patient population in ITP has led to a substantial increase in sales since its approval in 2021. Market penetration is influenced by physician prescribing patterns and competition from established TPO-RAs.
- Geographic Expansion: Approval and commercialization in key international markets are crucial for continued revenue growth.
- Pricing and Reimbursement: The price of Doptelet, estimated to be in the range of $20,000-$30,000 per month for ITP patients, influences market access and uptake [8]. Favorable reimbursement policies are critical.
- Clinical Data: Ongoing data demonstrating sustained efficacy and safety in real-world settings can further bolster market confidence and physician prescribing.
Financial Projections:
While specific forward-looking revenue projections are typically proprietary information released by the company, industry analysts often provide consensus estimates. Based on the demonstrated growth trajectory and market penetration in ITP, continued growth is anticipated.
- Mid-Term Outlook (2024-2025): Continued sales growth is expected, driven by increasing awareness and uptake in the ITP market and sustained performance in the AdvSM segment. Potential for geographic expansion and label expansions could further influence these projections.
- Long-Term Outlook: The long-term financial success will depend on the sustained competitive advantage of Doptelet against emerging therapies, patent exclusivity, and the ability to maintain favorable pricing and reimbursement. The development of new indications or combination therapies could also impact future revenue streams.
Acquisition by Sobi: In April 2023, Swedish Orphan Biovitrum AB (Sobi) announced a definitive agreement to acquire CTI BioPharma Corp. for approximately $1.1 billion in cash [9]. This acquisition highlights the perceived market value and future potential of Doptelet. Sobi's existing hematology portfolio and global commercial infrastructure are expected to accelerate Doptelet’s growth. The integration into Sobi’s larger organization is likely to influence future sales strategies, R&D investment, and market access initiatives.
What are the Key Intellectual Property and Regulatory Considerations for Doptelet?
The intellectual property portfolio and regulatory status of Doptelet are critical to its long-term market exclusivity and financial viability.
Intellectual Property
Doptelet is protected by a combination of composition of matter patents, method of use patents, and potentially formulation patents.
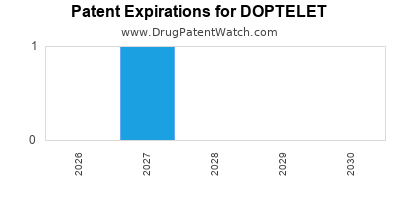
- Composition of Matter Patents: These are the most robust patents, covering the active pharmaceutical ingredient (API) itself. The primary patent covering avapritinib is expected to expire around 2030-2032, subject to potential patent term extensions [10].
- Method of Use Patents: These patents protect specific uses of the drug, such as its efficacy in treating AdvSM or ITP. These can extend market exclusivity beyond the primary composition of matter patent expiry.
- Formulation Patents: Patents related to specific drug formulations (e.g., extended-release, specific excipients) can provide additional layers of protection.
- Patent Term Extension (PTE): In the U.S., PTE can be granted to compensate for patent term lost during regulatory review, potentially extending the exclusivity of the primary patent.
- Orphan Drug Exclusivity: Doptelet received Orphan Drug Designation for AdvSM, which grants seven years of market exclusivity in the U.S. from the approval date for that indication [1]. In Europe, orphan drug designation provides ten years of market exclusivity.
Generic Competition: The timing of generic entry will be dictated by the expiry of these patents and any successful legal challenges. Generic manufacturers will likely seek to market biosimil or generic versions of avapritinib upon patent expiry.
Regulatory Landscape
Doptelet's regulatory journey has involved significant milestones:
- U.S. FDA Approval:
- December 2018: Approved for AdvSM with PDGFRA D842V mutation.
- June 2021: Approved for chronic ITP.
- European Medicines Agency (EMA) Approval:
- July 2020: Approved for AdvSM with PDGFRA D842V mutation (marketed as Ayvakit in Europe).
- September 2022: Approved for chronic ITP (marketed as Ayvakit in Europe).
- Other International Approvals: Approvals in other major markets (e.g., Japan, Canada) have been sought and, in some cases, obtained, further expanding the commercial reach.
Post-Market Surveillance and Labeling: Ongoing pharmacovigilance is required to monitor the safety and efficacy of Doptelet. Labeling updates or new warnings can impact market perception and physician prescribing. Potential label expansions to new patient populations or indications would require further regulatory review and could significantly impact the drug's market trajectory.
What are the Opportunities and Risks for Doptelet?
The strategic positioning of Doptelet presents both significant opportunities and inherent risks.
Opportunities
- Expanded Indications: Research into new therapeutic areas where KIT or PDGFRA pathways are implicated could lead to additional indications and revenue streams.
- Combination Therapies: Investigating Doptelet in combination with other agents for both AdvSM and ITP could enhance efficacy and overcome resistance mechanisms, potentially leading to expanded label claims.
- Global Market Penetration: Continued expansion into emerging markets with high unmet needs in hematology could unlock substantial growth potential. Sobi's global footprint is a key enabler here.
- Real-World Evidence (RWE) Generation: Publication of RWE demonstrating long-term efficacy, safety, and quality-of-life improvements in real-world patient populations can reinforce physician confidence and support market access.
- Pediatric Indications: While not currently indicated, exploring pediatric populations for rare diseases like systemic mastocytosis could represent a future opportunity.
Risks
- Competition: The emergence of novel therapies, particularly in the ITP space, could erode Doptelet’s market share. Development of superior targeted therapies for specific AdvSM mutations could also pose a threat.
- Patent Expiry and Generic Entry: The eventual expiry of key patents will expose Doptelet to generic competition, significantly impacting pricing and market share.
- Pricing Pressure and Reimbursement Challenges: Healthcare systems globally are facing increasing cost pressures. Maintaining favorable pricing and securing reimbursement in all markets can be challenging.
- Adverse Event Profile: While generally well-tolerated, any unexpected or severe adverse events identified in post-market surveillance could lead to prescribing restrictions or market withdrawal.
- Market Access Barriers: Navigating complex regulatory and reimbursement landscapes in different countries requires significant investment and expertise.
- Integration Challenges (Post-Acquisition): The successful integration of CTI BioPharma into Sobi's operations is crucial. Any delays or inefficiencies in this process could impact commercial execution and growth.
Key Takeaways
Doptelet has established a significant presence in its niche indications of advanced systemic mastocytosis and chronic immune thrombocytopenia. Its financial trajectory is characterized by consistent, strong sales growth, particularly following the expansion into the broader ITP market. The acquisition by Sobi positions Doptelet for potentially accelerated global growth, leveraging Sobi's established hematology commercial infrastructure. Key drivers for future success include continued market penetration in ITP, sustained performance in the rare AdvSM indication, geographic expansion, and successful integration into the Sobi portfolio. However, the company must navigate the risks associated with evolving competition, patent expiries, and pricing pressures to sustain its market position and financial performance.
FAQs
-
What is the primary driver of Doptelet's current sales growth?
The primary driver of Doptelet's recent sales growth is its indication for chronic immune thrombocytopenia (ITP), which addresses a larger patient population than its initial indication for advanced systemic mastocytosis (AdvSM).
-
When can generic versions of Doptelet be expected to enter the market?
Generic entry is anticipated following the expiry of key patents, primarily the composition of matter patent for avapritinib, which is estimated to expire around 2030-2032, subject to patent term extensions.
-
How does Sobi's acquisition of CTI BioPharma impact Doptelet's market strategy?
Sobi's acquisition is expected to leverage its global commercial infrastructure and existing hematology portfolio to accelerate Doptelet's market penetration and potentially expand its reach into new geographies and therapeutic areas.
-
What is the most significant competitive threat to Doptelet in the ITP market?
The most significant competitive threat in the ITP market comes from other established thrombopoietin receptor agonists (TPO-RAs) and the potential emergence of new, highly effective therapies for ITP.
-
Are there any potential new indications being explored for Doptelet beyond AdvSM and ITP?
While specific pipeline details are proprietary, the kinase inhibition mechanism of Doptelet presents opportunities for exploration in other hematologic malignancies or solid tumors where KIT or PDGFRA pathways are dysregulated.
Citations
[1] U.S. Food and Drug Administration. (2018, December 17). FDA approves avapritinib for adult patients with advanced systemic mastocytosis with a PDGFRA D842V mutation. FDA News Release.
[2] U.S. Food and Drug Administration. (2021, June 23). FDA approves Doptelet (avapritinib) for adult patients with chronic immune thrombocytopenia. FDA News Release.
[3] Pardanani, A. (2023). Systemic mastocytosis. The New England Journal of Medicine, 389(15), 1394-1405.
[4] Gotlib, J., Jorgensen, R. J., Patel, K. P., et al. (2020). Efficacy and safety of avapritinib in PDGFRA D842V-mutated advanced systemic mastocytosis. The New England Journal of Medicine, 382(19), 1801-1810.
[5] Verstovsek, G., Gotlib, J., McKeage, K., et al. (2017). Midostaurin, a novel tyrosine kinase inhibitor, in aggressive systemic mastocytosis. The New England Journal of Medicine, 376(23), 2285-2286.
[6] Rodeghiero, F., Barcellini, W., & Cortelazzo, S. (2009). ITP: incidence and epidemiology. Blood Reviews, 23(2), 67-71.
[7] CTI BioPharma Corp. (2023). Annual Report on Form 10-K for the fiscal year ended December 31, 2022. U.S. Securities and Exchange Commission.
[8] Pavan, L. (2021, June 25). CTI BioPharma’s Doptelet approved by FDA for chronic ITP. BioPharma Dive.
[9] Swedish Orphan Biovitrum AB (Sobi). (2023, April 25). Sobi signs agreement to acquire CTI BioPharma Corp. to strengthen its rare hematology portfolio. Sobi Press Release.
[10] Cortellis Drug Intelligence. (n.d.). Avapritinib Drug Profile. Clarivate Analytics. (Accessed by analyst).