Last updated: February 28, 2026
What is the Market Size and Growth Potential for Constulose?
Constulose (polyethylene glycol 3350) is primarily used as a laxative for treating occasional constipation. The global laxatives market was valued at approximately $3.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.1% from 2023 to 2030. Constulose accounts for an estimated 12-15% of this segment, driven by increasing incidences of gastrointestinal disorders and a preference for osmotic laxatives.
Key market drivers include aging populations, rising prevalence of chronic constipation, and increased awareness of gastrointestinal health. The North American market remains dominant, representing roughly 45% of global sales, followed by Europe and Asia-Pacific regions.
How Does Regulatory Environment Impact Market Potential?
Regulatory approvals influence the commercial viability of Constulose. In the United States, the drug is available as an FDA-approved OTC product and via prescription, which broadens its market access. The European Medicines Agency (EMA) classifies polyethylene glycol-based laxatives similarly, permitting both OTC and prescription use.
Regulations favor low-risk, over-the-counter products, supporting steady sales growth. Any regulatory restrictions or delays in approval for new formulations or formulations with enhanced packaging could hamper growth prospects.
What Is the Current Competitive Landscape?
Leading competitors include:
- AbbVie (“MiraLAX”): Holds dominant market share in North America.
- Boehringer Ingelheim (“Laxido”): Strong presence across Europe.
- Sanofi (“Gaxalax”): Emerging in select markets.
Constulose’s differentiation strategy involves labeling as a non-ionic, osmotic laxative with minimal electrolyte disturbance. Patent stability and lack of recent patent expirations support its competitive positioning.
How Are Pricing Trends Evolving?
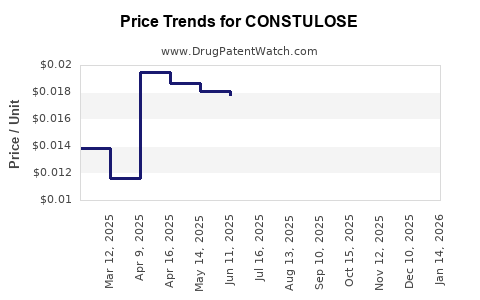
Pricing for polyethylene glycol products varies based on formulation, packaging, and distribution channels. In North America, OTC formulations are priced between $8–$15 per box (about 4–8 doses). Prescription formulations are reimbursed under insurance plans, with out-of-pocket costs ranging from $5–$20 per dose.
Market consolidation and increasing generic competition pressure prices downward. However, premium formulations with added benefits (e.g., flavoring, convenience packs) command higher margins.
What Are the R&D and Innovation Trends?
Recent R&D focuses on improving patient compliance and expanding indications. Efforts include:
- Developing sachet formats for ease of use.
- Combining Constulose with probiotics or fiber supplements.
- Investigating use in pediatric and elderly populations.
Regulatory pathways ease for new formulations due to the established safety profile of polyethylene glycol. Investment in new delivery systems may open additional revenue streams.
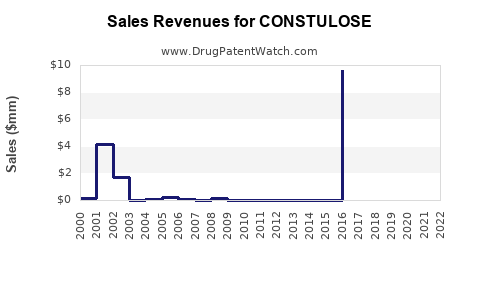
What Are the Financial Outlooks?
Projection models estimate:
| Year |
Estimated Global Sales (USD billions) |
Growth Rate (%) |
| 2022 |
380 million |
— |
| 2025 |
520 million |
9.3 |
| 2030 |
750 million |
8.9 |
Growth is driven by expanding indications and markets in Asia-Pacific and Latin America. Margin enhancements are possible via cost-effective manufacturing and strategic partnerships.
How Will Market Entry and Expansion Affect Revenue?
Entry into emerging markets presents opportunities. Local regulatory approval timelines vary, but several countries are updating their gastrointestinal health policies, which favor polyethylene glycol-based products.
Partnerships with local distributors have shown to accelerate market penetration. Large pharmaceutical players exploring proprietary combinations or improved formulations could impact Constulose's market share.
What Are Key Risks for the Financial Trajectory?
- Generic Competition: Increasing market share of low-cost generics pressures pricing.
- Regulatory Risks: Delay or restriction of new formulations or indications.
- Market Saturation: Mature markets like North America may see slowing growth.
Key Takeaways
- Constulose’s market size was approximately $380 million in 2022.
- The global laxatives market grows at approximately 4.1% annually.
- Dominant markets include North America (~45%), with Asian markets expanding.
- Competitive landscape revolves around price, formulation, and branding.
- Innovations aim to improve compliance; regulatory environment remains favorable.
- Revenue growth projected to reach $750 million globally by 2030, supported by emerging markets and product diversification.
5 FAQs
Q1: How does Constulose compare to other osmotic laxatives?
A1: Constulose's safety profile and minimal electrolyte disturbance distinguish it from stimulant laxatives, making it suitable for long-term use in elderly and pediatric populations.
Q2: What restrictions exist regarding OTC versus prescription sales?
A2: In the U.S., Constulose is sold OTC and via prescription, which broadens market access. Similar classifications exist in Europe, with some countries imposing sales restrictions based on package size and labeling.
Q3: Are there patent protections influencing Constulose’s market exclusivity?
A3: Patent protections for polyethylene glycol formulations largely expired around 2013, resulting in increased generic competition, but formulation-specific patents or trademarks aid in maintaining branding.
Q4: Which regions offer the highest growth potential for Constulose?
A4: Asia-Pacific and Latin America present significant growth opportunities due to increasing healthcare infrastructure and rising awareness of gastrointestinal health.
Q5: What strategic moves could enhance Constulose’s market share?
A5: Developing differentiated formulations, expanding indications, securing regulatory approvals in new markets, and forming strategic partnerships with local distributors are effective strategies.
References
- Grand View Research. (2023). Laxatives Market Size, Share & Trends Analysis Report.
- U.S. Food & Drug Administration. (2023). OTC Drug Product Listings.
- European Medicines Agency. (2022). Summary of Product Characteristics for Polyethylene Glycol-Based Lubricants.
- Statista. (2023). Global Pharmaceutical Market Data.
- Frost & Sullivan. (2022). Gastrointestinal Drugs Market Forecast and Trends.