Last updated: February 19, 2026
What is COARTEM and its Mechanism of Action?
COARTEM, a fixed-dose combination of artemether and lumefantrine, is an oral antimalarial medication. Its primary indication is the treatment of uncomplicated Plasmodium falciparum malaria in patients weighing at least 5 kg. The drug targets the parasite's erythrocytic stage. Artemether is a derivative of artemisinin, which rapidly reduces parasite biomass by cleaving the parasite's membrane. Lumefantrine acts more slowly by inhibiting parasite heme polymerase. This combination targets different stages of the parasite's life cycle and is designed to prevent the development of resistance. COARTEM is manufactured by Novartis. [1, 2]
What is the Global Market Size and Growth of COARTEM?
The global market for antimalarial drugs, including COARTEM, is substantial and driven by disease burden in endemic regions. While specific market size figures for COARTEM alone are not publicly disclosed by Novartis, the broader antimalarial market was valued at approximately USD 1.3 billion in 2022 and is projected to reach USD 1.7 billion by 2030, growing at a compound annual growth rate (CAGR) of 3.5% from 2023 to 2030. This growth is influenced by factors such as increased government spending on malaria control programs, the prevalence of malaria in sub-Saharan Africa, and the development of new treatment guidelines. [3, 4]
COARTEM has been a cornerstone of malaria treatment for years. Its market position is influenced by the global strategies of organizations like the World Health Organization (WHO), which recommends artemisinin-based combination therapies (ACTs) as first-line treatments for uncomplicated P. falciparum malaria. [5]
What are the Key Geographic Markets for COARTEM?
The primary geographic markets for COARTEM are countries with a high prevalence of malaria. Sub-Saharan Africa accounts for the largest share of malaria cases and deaths globally, making it the most significant market for COARTEM. Other endemic regions include parts of Southeast Asia, South America, and Oceania. [5]
Key countries with high malaria burdens that represent significant markets include:
- Nigeria: Consistently has the highest number of malaria cases and deaths.
- Democratic Republic of Congo: Another country with a very high disease burden.
- Uganda: Experiences a substantial number of malaria cases.
- India: While cases have declined, it remains an important market.
- Myanmar: Endemic region with significant malaria cases. [5, 6]
The market penetration in these regions is often influenced by national malaria control programs, donor funding, and the accessibility and affordability of treatments.
What is the Competitive Landscape for COARTEM?
COARTEM operates within a competitive market of antimalarial drugs. Its primary competitors are other artemisinin-based combination therapies (ACTs). The therapeutic choice is often guided by WHO recommendations and national treatment guidelines, which prioritize combinations demonstrating efficacy and minimizing resistance.
Major competitors and alternative treatment classes include:
- Other ACTs: Medications containing different artemisinin derivatives or fixed-dose combinations, such as artesunate-mefloquine, artesunate-amodiaquine, and dihydroartemisinin-piperaquine. Examples include Eurartesim (dihydroartemisinin-piperaquine) and Pyramax (artesunate-mefloquine). [7]
- Non-ACT antimalarials: While ACTs are preferred, other drugs like sulfadoxine-pyrimethamine (used for intermittent preventive treatment in pregnancy) and newer investigational compounds may play a role in specific contexts or as second-line treatments if resistance emerges. [5]
- Generic manufacturers: As patents expire, generic versions of artemisinin-based combinations enter the market, increasing price competition. [8]
The landscape is dynamic, with ongoing research into novel antimalarials and strategies to combat resistance.
What is the Patent Status and Exclusivity for COARTEM?
Novartis held primary patents protecting COARTEM. The patent landscape for established drugs like COARTEM is complex, involving composition of matter patents, formulation patents, and use patents. The original composition of matter patents for artemether and lumefantrine have expired in major markets. For example, patents related to the specific fixed-dose combination and manufacturing processes were crucial for maintaining market exclusivity for a period after the expiry of basic compound patents. [8]
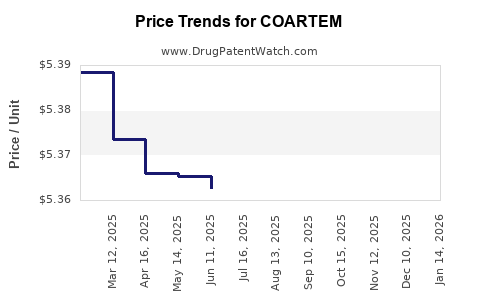
The expiry of key patents has allowed for the introduction of generic versions of artemether-lumefantrine. This has led to increased price competition and a shift in market dynamics, particularly in low- and middle-income countries where cost is a significant factor. [8]
What is the Financial Performance and Revenue Generation of COARTEM?
Specific revenue figures for COARTEM are not broken out by Novartis in its public financial reports. As a mature product in a critical therapeutic area, its financial contribution is part of Novartis's broader Global Health and Established Medicines divisions. However, its consistent use as a first-line treatment in high-burden countries indicates a stable revenue stream.
Factors influencing its financial performance include:
- Volume-driven sales: The vast majority of COARTEM sales occur in public health programs in malaria-endemic countries, often through tenders and large procurement deals with governments and NGOs.
- Price adjustments: In low-income settings, pricing is typically lower due to affordability considerations and competition from generics.
- Impact of generic competition: The entry of generic artemether-lumefantrine combinations has likely put downward pressure on pricing and market share in some regions. [8]
Despite generic competition, COARTEM remains a significant product due to its established efficacy, WHO recommendation, and Novartis's continued supply chain management in critical regions. The financial trajectory is characterized by consistent volume demand, tempered by price sensitivity and competitive pressures.
What are the Key R&D Trends and Future Outlook for COARTEM?
The R&D focus surrounding artemisinin-based therapies, including COARTEM, is primarily driven by the need to monitor and combat emerging drug resistance. While COARTEM has shown remarkable efficacy, the threat of P. falciparum resistance to artemisinins, particularly characterized by delayed parasite clearance, is a constant concern. [9]
Key R&D trends include:
- Resistance monitoring and surveillance: Ongoing studies track the prevalence and spread of resistance markers to artemisinins and their partner drugs. This includes monitoring parasite clearance times in clinical trials. [9]
- Development of novel ACTs: Research is underway to identify new artemisinin derivatives or novel partner drugs that can overcome existing resistance mechanisms or offer improved efficacy and safety profiles.
- Combination therapies beyond ACTs: Exploration of new drug combinations that include non-artemisinin compounds to provide alternative treatment options.
- Improved diagnostics and delivery: R&D in point-of-care diagnostics to ensure correct treatment for malaria and better drug delivery mechanisms. [5]
The future outlook for COARTEM is tied to global malaria control efforts. It is expected to remain a vital component of treatment regimens in many regions for the foreseeable future. However, its long-term dominance will depend on the sustained efficacy of its components and the ability to effectively manage emerging resistance through ongoing R&D and strategic deployment of alternative therapies.
Key Takeaways
- COARTEM (artemether-lumefantrine) is a WHO-recommended first-line treatment for uncomplicated Plasmodium falciparum malaria.
- Sub-Saharan Africa constitutes the primary geographic market, driven by high malaria prevalence.
- The competitive landscape includes other artemisinin-based combination therapies (ACTs) and generic manufacturers.
- Key patents for COARTEM have expired, leading to generic competition and price erosion.
- Specific revenue figures for COARTEM are not disclosed, but it contributes to Novartis's established medicines portfolio through volume-driven sales in public health programs.
- Future R&D is focused on monitoring and combating drug resistance and developing novel antimalarial therapies.
Frequently Asked Questions
What is the primary mechanism by which COARTEM fights malaria?
COARTEM works by targeting the erythrocytic stage of the Plasmodium falciparum parasite. Artemether rapidly reduces parasite biomass by damaging the parasite's membrane, while lumefantrine inhibits the parasite's heme polymerase, a crucial enzyme for its survival. [1, 2]
Has malaria parasite resistance to COARTEM been observed?
Yes, resistance to artemisinins, the core component of COARTEM, has been observed in several regions, primarily characterized by delayed parasite clearance. This necessitates ongoing monitoring and the development of alternative treatment strategies. [9]
How does the pricing of COARTEM compare to generic artemether-lumefantrine combinations?
Generic artemether-lumefantrine combinations are typically priced lower than branded COARTEM, particularly in low- and middle-income countries, due to reduced R&D recoupment costs and increased market competition. [8]
What is the role of COARTEM in global malaria eradication efforts?
COARTEM is a critical tool in current malaria control strategies, recommended by the WHO for treating uncomplicated P. falciparum malaria. Its widespread availability and effectiveness contribute to reducing morbidity and mortality, which are essential steps towards eventual eradication. [5]
Are there any significant side effects associated with COARTEM use?
Common side effects of COARTEM include nausea, vomiting, abdominal pain, headache, and dizziness. Serious adverse events are rare but can include neurological or cardiac effects. It is crucial to administer COARTEM under medical supervision. [1]
Citations
[1] World Health Organization. (2015). Guidelines for the treatment of malaria (3rd ed.). World Health Organization.
[2] Djimdé, A. A., & Taylor, T. E. (2009). Artemether–lumefantrine for the treatment of uncomplicated falciparum malaria. The New England Journal of Medicine, 361(11), 1075-1077.
[3] Grand View Research. (2023). Antimalarial Drugs Market Size, Share & Trends Analysis Report By Drug Class, By Disease Type, By Region, And Segment Forecasts, 2023-2030.
[4] Market Research Future. (2023). Antimalarial Drugs Market - Global Research Report 2030.
[5] World Health Organization. (n.d.). Malaria. Retrieved from https://www.who.int/news-room/fact-sheets/detail/malaria
[6] GBD 2019 Malaria Collaborators. (2021). Estimates of global, regional, and national burden of malaria in 2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet, 396(10244), 95-107.
[7] Pukrittayakamee, S., Domarle, B. L., & Singhasivanon, P. (2010). Artemisinin-based combination therapies in the treatment of malaria. Expert Opinion on Pharmacotherapy, 11(10), 1609-1620.
[8] Waako, J., & Orem, J. (2008). The introduction of artemisinin-based combination therapies in Africa. Tropical Medicine & International Health, 13(2), 242-248.
[9] Ashley, E. A., Walsh, D. S., & White, N. J. (2018). Malaria: artemisinin resistance-a real threat. Nature Reviews Diseases Primers, 4(1), 1-2.