Last updated: February 20, 2026
Candesartan cilexetil is a selective angiotensin II receptor blocker (ARB) approved for the management of hypertension and heart failure. Since its market entry, the drug has experienced steady growth, influenced by regulatory developments, competitive landscape shifts, and healthcare trends.
Market Overview
The global antihypertensive drugs market exceeded USD 34 billion in 2022. Candesartan cilexetil contributes a significant segment within ARBs, which accounted for approximately USD 8.2 billion in 2022, with continued growth projected at a Compound Annual Growth Rate (CAGR) of 4.2% through 2030.
Key Market Drivers
- Increased Hypertension Prevalence: Approximately 1.28 billion adults globally have hypertension, driving demand for ARBs.
- Cardiovascular Disease (CVD) Management: Candesartan cilexetil is prescribed for heart failure, post-myocardial infarction, and hypertensive crisis.
- Favorable Side-Effect Profile: Compared to ACE inhibitors, candesartan offers a lower incidence of cough and angioedema, supporting adherence.
- Generic Availability: Patent expirations have led to multiple generic versions, reducing costs and broadening access.
- Line Extensions and Combinations: Fixed-dose combinations (FDCs) with other antihypertensives expand therapeutic options.
Patent and Regulatory Landscape
Candesartan cilexetil's patent expired in many markets by 2015. Generic versions are now available in the US, EU, and emerging economies. Patent protections for some formulations (e.g., specific FDCs) extend until 2024-2026.
Regulatory agencies like the FDA and EMA oversee approvals. Recent guidelines emphasize evidence-based use for hypertensive and heart failure indications, impacting prescribing practices.
Competitive Landscape
Major suppliers include:
- Takeda Pharmaceuticals: Original developer, with a dominant market share prior to patent expiration.
- Generic Manufacturers: Teva, Mylan, Sandoz, and others hold substantial market portions due to cost advantages.
- Combination Product Makers: Firms developing candesartan-based FDCs with amlodipine, hydrochlorothiazide, or other agents.
Market share distribution reflects a shift toward generics, with branded sales declining post-patent expiry.
Financial Trajectory
Revenue Trends
In 2014, candesartan cilexetil-generated sales exceeded USD 1 billion globally. Post-patent loss, branded sales decreased by 45% in the US by 2016, with generics capturing majority market share.
Pricing and Market Penetration
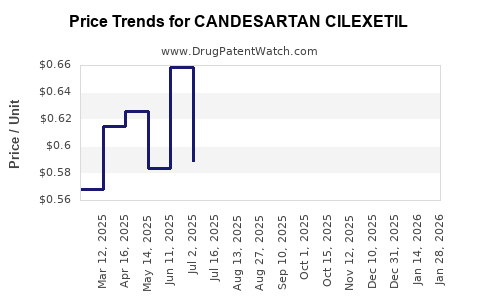
Generic pricing in developed markets has reduced by an average of 60-75% since patent expiry, leading to increased volume sales but decreased per-unit revenue. For instance, in the US, the average retail price for a 30-day supply fell from USD 120 in 2014 to below USD 30 in 2022.
Forecasted Sales
Between 2023 and 2030, global sales are projected to grow from USD 1.2 billion to USD 1.6 billion, driven by rising hypertension prevalence, especially in Asia-Pacific and Latin American markets, where generic penetration remains lower.
Regional Variations
- United States: Market saturation with generics, slower growth.
- Europe: Moderate growth, with increasing adoption of FDCs.
- Emerging Markets: Rapid sales growth, driven by expanding healthcare access and lower drug costs.
External Influences on Market and Financials
- Healthcare Policies: Price controls in Europe and Asia limit revenue growth.
- Reimbursement Policies: Favorability toward generics boosts uptake.
- New Therapeutics: Introduction of novel ARBs and combination therapies (e.g., sacubitril/valsartan) impacts market share.
Risks and Opportunities
Risks:
- Pricing pressure from generics.
- Regulatory delays for new formulations.
- Competition from novel antihypertensives and ARBs.
Opportunities:
- Development of novel formulations (extended-release, FDCs).
- Expansion into emerging markets.
- Potential patent filings for new uses or combinations.
Key Takeaways
- Candesartan cilexetil's revenue declined sharply post-2015 patent expiration, but volume sales compensated partially.
- Generic competition reduces per-unit prices, constraining revenue growth in developed markets.
- Global sales are forecasted to grow modestly (~3% CAGR) due to demographic trends and increasing hypertension diagnoses.
- Emerging markets present growth opportunities due to expanding healthcare infrastructure and affordability.
- Strategic focus on formulation innovation and market expansion can mitigate revenue pressures.
FAQs
1. What has been the impact of patent expiration on candesartan cilexetil revenues?
Patent expiration in 2015 led to widespread generic entry, decreasing branded sales and price points, shifting revenue reliance to volume.
2. How does generic competition influence the global market for candesartan cilexetil?
It reduces prices by 60-75% in mature markets, diminishes branded sales, and encourages wider access but limits profit margins.
3. Are there new formulations or combinations for candesartan cilexetil under development?
Yes. Fixed-dose combinations with amlodipine or hydrochlorothiazide are available, and research into extended-release formulations continues.
4. What regional factors affect the growth trajectory of candesartan cilexetil?
Price controls and reimbursement policies impact mature markets; rising hypertension prevalence and healthcare access drive growth in emerging economies.
5. How might upcoming patents influence the market in the next five years?
New patents for specific formulations or uses could temporarily boost revenues before generic competition increases.
References
[1] MarketWatch. (2022). Global antihypertensive drugs market size.
[2] IMS Health. (2014). Cardiovascular drug market data.
[3] European Medicines Agency. (2022). Approved antihypertensive drugs.
[4] FDA. (2020). Guidance on generic drug approval procedures.
[5] IQVIA. (2023). Emerging markets healthcare trends.