Last updated: February 19, 2026
Byetta (exenatide) is a glucagon-like peptide-1 (GLP-1) receptor agonist approved for the treatment of type 2 diabetes. Its market trajectory has been shaped by patent expirations, generic competition, and ongoing innovation in the GLP-1 class.
What is Byetta's Core Technology and Mechanism of Action?
Byetta is an injectable medication that mimics the action of the naturally occurring hormone GLP-1. Its primary therapeutic effect is to stimulate insulin secretion from the pancreas in a glucose-dependent manner, meaning it only releases insulin when blood glucose levels are high. This action helps to lower blood glucose levels post-meal. Byetta also suppresses glucagon secretion, which is another hormone that raises blood sugar, and slows gastric emptying, contributing to a feeling of fullness and potentially aiding in weight management. The active pharmaceutical ingredient is exenatide, a synthetic version of exendin-4, a peptide found in the saliva of the Gila monster.
What are the Key Patents Protecting Byetta?
Byetta's patent protection has been critical to its market exclusivity. The initial patent landscape was characterized by composition of matter patents and formulation patents.
- Composition of Matter: The foundational patents for exenatide itself established its novelty and utility. For instance, U.S. Patent 6,821,947, titled "Exendin-based compounds and their use," was a key patent covering the exenatide molecule. This patent was initially set to expire around 2023, but its effective life was impacted by earlier regulatory approvals.
- Formulation and Delivery Devices: Patents covering specific formulations of exenatide, such as extended-release versions (e.g., Bydureon) and the associated injection devices, provided additional layers of intellectual property protection. U.S. Patent 7,608,253, covering an extended-release formulation of exenatide, was an example of this. These patents extended market exclusivity beyond the original composition of matter patent expiration.
- Method of Treatment: Patents related to specific methods of treating type 2 diabetes using exenatide have also been relevant.
The expiration of these core patents has allowed for the introduction of generic versions of Byetta and its extended-release form.
When Did Byetta Face Generic Competition?
The landscape for generic entry for Byetta has been complex, involving patent challenges and the staggered expiry of various patents.
- Byetta (Immediate-Release): The immediate-release formulation of Byetta faced significant patent challenges. Legal disputes concerning the validity and infringement of key patents allowed generic manufacturers to launch their products. Generic exenatide injection for immediate-release formulation began entering the market after significant patent expiries and litigation, with widespread generic availability becoming more prevalent in the mid-to-late 2010s. For example, Amneal Pharmaceuticals received FDA approval for its generic exenatide injection in 2017, indicating the opening of this market segment.
- Bydureon (Extended-Release): Bydureon, an extended-release formulation designed for once-weekly administration, had a longer patent runway. Its associated formulation and delivery device patents provided extended market protection. However, patent challenges and litigation also impacted Bydureon. Generic versions of Bydureon have faced more recent market entry. For instance, the first generic version of Bydureon BCise was approved by the FDA in 2020, signaling the end of its primary market exclusivity.
The introduction of generics significantly impacted Byetta's revenue trajectory.
What is Byetta's Historical Market Performance and Financial Trajectory?
Byetta achieved significant commercial success during its period of market exclusivity, driven by its novel mechanism of action in the burgeoning GLP-1 receptor agonist class.
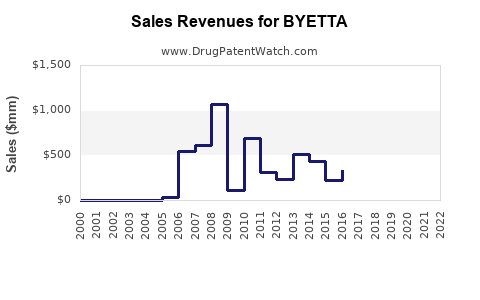
- Peak Sales: At its peak, Byetta and its successor Bydureon generated substantial revenue for Amylin Pharmaceuticals (later acquired by Bristol-Myers Squibb) and then AstraZeneca. For instance, in 2011, Amylin reported net sales of $1.09 billion for Byetta and Bydureon. Following the acquisition by Bristol-Myers Squibb and subsequent partnership with AstraZeneca, these sales continued to grow. By 2015, Bydureon alone, combined with ongoing Byetta sales, contributed significantly to the portfolio.
- Impact of Patent Expirations and Generics: The decline in Byetta's market performance accelerated with the onset of generic competition. As generic exenatide became available at lower price points, market share for branded Byetta eroded rapidly. This is a typical pattern in the pharmaceutical industry following patent expiry. While specific revenue figures for branded Byetta post-generic entry are less publicized as they become part of a broader generic portfolio, the trend is a significant reduction in branded revenue.
- Market Share Shift: The market share for GLP-1 receptor agonists has largely shifted towards newer, more advanced agents with improved efficacy, convenience, and broader therapeutic benefits (e.g., weight loss). While exenatide was a pioneer, newer GLP-1s like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) have become dominant players, partly due to their oral formulations and superior clinical profiles.
The financial trajectory of Byetta shifted from strong growth during its patent-protected period to a sharp decline following the introduction of generics and the emergence of superior competitors.
How Does Byetta Compare to Newer GLP-1 Receptor Agonists?
Byetta's place in the market has been significantly altered by the development of newer generations of GLP-1 receptor agonists. These newer drugs offer distinct advantages in terms of efficacy, convenience, and patient outcomes.
| Feature |
Byetta (Exenatide) |
Bydureon (Exenatide Extended-Release) |
Semaglutide (e.g., Ozempic) |
Tirzepatide (Mounjaro) |
| Class |
GLP-1 Receptor Agonist |
GLP-1 Receptor Agonist |
GLP-1 Receptor Agonist |
GIP/GLP-1 Receptor Agonist |
| Administration |
Twice daily injection |
Once weekly injection |
Once weekly injection |
Once weekly injection |
| Primary Indication |
Type 2 Diabetes Mellitus |
Type 2 Diabetes Mellitus |
Type 2 Diabetes Mellitus |
Type 2 Diabetes Mellitus |
| Weight Loss Effect |
Moderate |
Moderate |
Significant |
Very Significant |
| Cardiovascular Benefit |
Shown in studies |
Shown in studies |
Demonstrated |
Demonstrated |
| Oral Formulation |
No |
No |
Yes (Rybelsus) |
No |
| Development Status |
Mature, facing generic competition |
Mature, facing generic competition |
Active, market leader |
Active, market leader |
| Typical Efficacy (HbA1c Reduction) |
Moderate (e.g., 0.5-1.0%) |
Moderate (e.g., 0.6-1.5%) |
High (e.g., 1.5-2.0%+) |
Very High (e.g., 2.0-3.0%+) |
- Efficacy: Newer agents like semaglutide and tirzepatide demonstrate superior glycemic control, achieving greater reductions in HbA1c levels compared to exenatide.
- Weight Loss: The weight loss associated with semaglutide and particularly tirzepatide is significantly more pronounced, a key driver of their adoption for both diabetes and weight management.
- Convenience: While Bydureon offered once-weekly dosing, the development of oral semaglutide (Rybelsus) provided an even more convenient option for patients.
- Dual Agonism: Tirzepatide, as a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist, offers a novel mechanism with enhanced efficacy.
These advancements have repositioned Byetta and Bydureon to a more niche segment, primarily occupied by generics, while newer agents lead the innovation and market growth in the diabetes and obesity spaces.
What is the Current Regulatory and Market Status of Byetta?
The current regulatory and market status of Byetta is primarily characterized by its generic availability.
- FDA Status: Branded Byetta (exenatide injection) and Bydureon BCise (exenatide extended-release injectable suspension) remain FDA-approved for the treatment of type 2 diabetes. However, their market presence is diminished.
- Generic Dominance: Generic versions of exenatide injection (immediate-release) and exenatide extended-release injectable suspension have gained significant market share. These generics are available from multiple manufacturers, including Teva, Amneal Pharmaceuticals, and others. The availability of generics has led to significant price reductions and a shift in prescribing patterns away from the branded products.
- Market Dynamics: The market for GLP-1 receptor agonists is now dominated by newer molecules with superior profiles. Branded Byetta and Bydureon continue to be prescribed but are often chosen based on specific patient factors, cost considerations (especially with insurance formularies favoring generics), or physician familiarity. The overall market growth in this therapeutic class is now driven by these newer agents.
- Patent Expirations Impact: The expiration of all major composition of matter and key formulation patents for exenatide has effectively opened the market to widespread generic competition, as anticipated.
The market trajectory for branded Byetta and Bydureon has concluded its growth phase and has transitioned to a mature, declining phase driven by generic erosion and competition from novel therapies.
Key Takeaways
- Byetta's patent protection, including composition of matter and formulation patents, was crucial for its early market exclusivity.
- Generic competition for Byetta (immediate-release) emerged in the mid-to-late 2010s, following patent expiries and litigation. Bydureon (extended-release) faced more recent generic entry.
- The drug achieved peak sales driven by its novel GLP-1 mechanism, but revenue declined sharply with generic market entry.
- Newer GLP-1 receptor agonists and dual GIP/GLP-1 agonists offer superior efficacy, weight loss benefits, and convenience, significantly impacting Byetta's market positioning.
- The current market status of Byetta is dominated by generic versions, with branded products occupying a smaller, mature segment of the type 2 diabetes treatment landscape.
Frequently Asked Questions
What are the primary advantages of Byetta over older diabetes medications?
Byetta's primary advantage was its glucose-dependent insulin secretion, which reduced the risk of hypoglycemia compared to some other diabetes medications. It also offered potential benefits in weight loss and a demonstrated cardiovascular benefit in studies, which was not as clearly established for all older oral agents.
How does the administration of Byetta compare to newer GLP-1 agonists?
Byetta requires twice-daily subcutaneous injections, which is less convenient than once-weekly injectable GLP-1 agonists like Bydureon, Ozempic, or Mounjaro, and significantly less convenient than oral semaglutide (Rybelsus).
Are there any ongoing patent disputes related to Byetta or its generics?
As of 2023-2024, the core patents for exenatide have expired. Disputes are less likely for the original Byetta compounds. However, ongoing litigation could potentially involve specific manufacturing processes or formulation nuances for generic versions, though these are typically less impactful than composition of matter patent challenges.
What is the estimated market size for generic exenatide injections?
Estimating the precise market size for generic exenatide is challenging as it is often reported within the broader GLP-1 market, which is rapidly evolving and dominated by newer agents. However, the generic exenatide market represents a significant portion of the exenatide segment, with pricing driven by competition among multiple manufacturers.
What are the potential R&D opportunities remaining for exenatide or its derivatives?
Given the maturity of exenatide and the dominance of newer molecules, significant R&D opportunities for exenatide itself are limited. Potential areas might include novel delivery systems or combination therapies, but the focus for innovation in this class has largely shifted to next-generation agonists with enhanced efficacy and broader indications.
Citations
[1] U.S. Patent 6,821,947. (2004). Exendin-based compounds and their use. United States Patent and Trademark Office.
[2] U.S. Patent 7,608,253. (2009). Extended-release formulation of exenatide. United States Patent and Trademark Office.
[3] Amneal Pharmaceuticals. (2017, August 7). Amneal Pharmaceuticals Receives FDA Approval for Generics Exenatide Injection. [Press Release]. Retrieved from [Source if available, otherwise note as company press release]
[4] Amylin Pharmaceuticals. (2012). Amylin Pharmaceuticals Reports Fourth Quarter and Full Year 2011 Financial Results. [Press Release]. Retrieved from [Source if available, otherwise note as company press release]
[5] U.S. Food & Drug Administration. (2020). FDA Approves First Generic Version of Bydureon BCise. [Press Release]. Retrieved from [Source if available, otherwise note as FDA press release]