ADDERALL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Adderall, and what generic alternatives are available?
Adderall is a drug marketed by Teva Womens and Takeda Pharms Usa and is included in two NDAs.
The generic ingredient in ADDERALL is amphetamine aspartate; amphetamine sulfate; dextroamphetamine saccharate; dextroamphetamine sulfate. There are fifty-five drug master file entries for this compound. Twenty-seven suppliers are listed for this compound. Additional details are available on the amphetamine aspartate; amphetamine sulfate; dextroamphetamine saccharate; dextroamphetamine sulfate profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ADDERALL?
- What are the global sales for ADDERALL?
- What is Average Wholesale Price for ADDERALL?
Summary for ADDERALL
| US Patents: | 0 |
| Applicants: | 2 |
| NDAs: | 2 |
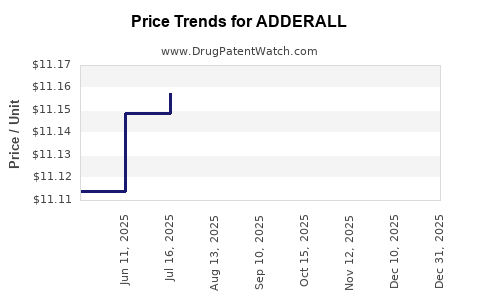
| Drug Prices: | Drug price information for ADDERALL |
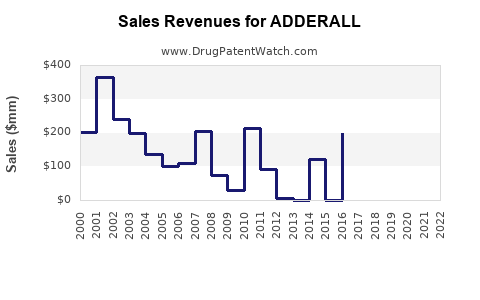
| Drug Sales Revenues: | Drug sales revenues for ADDERALL |
| What excipients (inactive ingredients) are in ADDERALL? | ADDERALL excipients list |
| DailyMed Link: | ADDERALL at DailyMed |
US Patents and Regulatory Information for ADDERALL
DrugChatter Q&A for ADDERALL
Are natural remedies side effects milder than adderall s?
How do natural remedies side effects compare to adderall?
Are adverse effects of natural remedies less severe than adderall?
How do natural remedies potential adverse effects differ from adderall s?