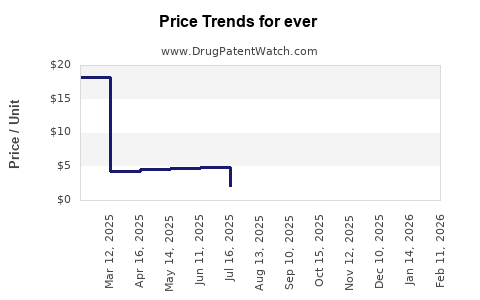
Ever - Generic Drug Details
✉ Email this page to a colleague
US Patents and Regulatory Information for ever
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Biocon Pharma | EVEROLIMUS | everolimus | TABLET;ORAL | 214182-004 | Feb 11, 2021 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Hikma | EVEROLIMUS | everolimus | TABLET;ORAL | 207486-001 | Jun 8, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Breckenridge | EVEROLIMUS | everolimus | TABLET;ORAL | 205432-003 | May 20, 2021 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Biocon Pharma | EVEROLIMUS | everolimus | TABLET;ORAL | 216140-002 | Apr 9, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Novugen | EVEROLIMUS | everolimus | TABLET;ORAL | 219955-002 | Oct 30, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |