Share This Page
VITAMIN A - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for vitamin a and what is the scope of freedom to operate?
Vitamin a
is the generic ingredient in eight branded drugs marketed by Astrazeneca, Banner Pharmacaps, Chase Chem, Everylife, Impax Labs, West Ward, Sterling Winthrop, Lilly, Del Ray Labs, Bayer Pharms, Bristol Myers Squibb, Elkins Sinn, Ivax Sub Teva Pharms, Mk Labs, Wharton Labs, Arcum, Teva, Casper Pharma Llc, and Bel Mar, and is included in thirty-one NDAs. Additional information is available in the individual branded drug profile pages.There are six drug master file entries for vitamin a.
Summary for VITAMIN A
| US Patents: | 0 |
| Tradenames: | 8 |
| Applicants: | 19 |
| NDAs: | 31 |
| Drug Master File Entries: | 6 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Patent Applications: | 7,396 |
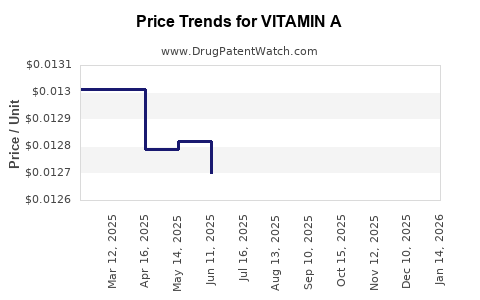
| Drug Prices: | Drug price trends for VITAMIN A |
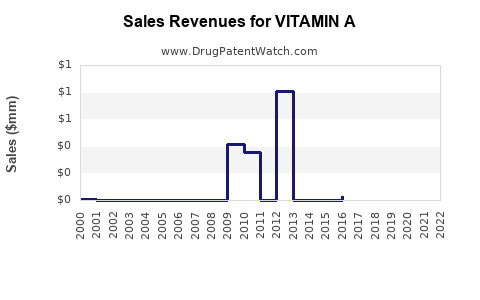
| Drug Sales Revenues: | Drug sales revenues for VITAMIN A |
| DailyMed Link: | VITAMIN A at DailyMed |
Medical Subject Heading (MeSH) Categories for VITAMIN A
US Patents and Regulatory Information for VITAMIN A
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ivax Sub Teva Pharms | VITAMIN A | vitamin a palmitate | CAPSULE;ORAL | 083035-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Everylife | VITAMIN A | vitamin a palmitate | CAPSULE;ORAL | 080943-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Bel Mar | VITAMIN A PALMITATE | vitamin a palmitate | INJECTABLE;INJECTION | 080819-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Sterling Winthrop | AFAXIN | vitamin a palmitate | CAPSULE;ORAL | 083187-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astrazeneca | AQUASOL A | vitamin a | CAPSULE;ORAL | 083080-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Vitamin A Pharmaceutical Market Analysis
This analysis assesses the market dynamics, patent landscape, and financial trajectory of Vitamin A as a pharmaceutical agent. The market is characterized by established therapeutic uses, a mature patent profile for basic formulations, and emerging patent activity around novel delivery systems and specific therapeutic applications. Regulatory hurdles, generic competition, and the development of advanced formulations are key factors influencing its commercial viability.
What are the primary pharmaceutical applications of Vitamin A?
Vitamin A, also known as retinol, is essential for vision, immune function, and cell growth. In pharmaceutical contexts, its primary applications include:
- Ophthalmology: Treatment of nyctalopia (night blindness) and xerophthalmia, conditions often stemming from Vitamin A deficiency. High-dose Vitamin A is a standard therapy in preventing blindness in children in regions with widespread deficiency [1].
- Dermatology: Isotretinoin, a synthetic derivative of Vitamin A, is a highly effective treatment for severe nodular acne and other dermatological conditions like psoriasis. Other topical retinoids are used for photoaging and acne vulgaris.
- Oncology: Research is ongoing into the role of retinoids in cancer prevention and treatment, particularly for certain leukemias and head and neck cancers. Their ability to induce differentiation and apoptosis in cancer cells is of significant interest [2].
- Reproductive Health: Vitamin A plays a role in male and female fertility and fetal development. However, excessive intake during pregnancy is teratogenic.
- Immunology: Vitamin A supports immune system function, and supplementation has been studied for its impact on infectious diseases, particularly in developing countries.
What is the current patent landscape for Vitamin A-based pharmaceuticals?
The patent landscape for Vitamin A itself is largely characterized by expired foundational patents covering its isolation, synthesis, and basic therapeutic uses. However, new patent filings are concentrated in specific areas:
- Novel Formulations and Delivery Systems: Patents exist for enhanced bioavailability formulations, sustained-release preparations, and targeted delivery mechanisms for Vitamin A and its derivatives. Examples include liposomal encapsulation, nanoparticle formulations, and transdermal patches designed to improve efficacy and reduce side effects [3].
- Specific Therapeutic Indications: Patents are being filed for the use of Vitamin A derivatives in treating specific diseases beyond the well-established ones. This includes novel applications in autoimmune disorders, neurodegenerative diseases, and specific cancer types, often based on new understandings of retinoid signaling pathways [4].
- Combination Therapies: Patents may cover the synergistic effects of Vitamin A or its derivatives when combined with other active pharmaceutical ingredients for enhanced therapeutic outcomes [5].
- Manufacturing Processes: While less common for basic Vitamin A, patents may emerge for novel, more efficient, or environmentally friendly synthesis routes for specific retinoids or their precursors.
The majority of expired patents relate to the chemical entity of Vitamin A (retinol) and its fundamental physiological roles. For instance, patents for the basic oral supplementation of Vitamin A for deficiency have long since expired. However, patents for specific synthetic analogs like isotretinoin, and for novel uses and formulations of these compounds, remain active.
Table 1: Key Patent Categories for Vitamin A Pharmaceuticals
| Patent Category | Description | Status of Core Technology | Example Focus Areas |
|---|---|---|---|
| Basic Synthesis & Isolation | Patents covering the initial methods of producing and purifying Vitamin A. | Largely expired | N/A |
| Established Therapeutic Uses | Patents for treating conditions like night blindness, xerophthalmia, acne. | Largely expired | N/A |
| Synthetic Analogs (e.g., Isotretinoin) | Patents for specific derivatives with enhanced or altered properties. | Varies; some core patents expired, new ones pending | Acne vulgaris, psoriasis, other dermatological conditions |
| Novel Formulations | Patents for improved drug delivery, stability, and bioavailability. | Active and growing | Liposomal encapsulation, nanoparticles, controlled-release systems, topical delivery |
| New Medical Indications | Patents for treating diseases beyond established uses, based on new research. | Active and growing | Cancer therapy, autoimmune disorders, neuroprotection, inflammatory conditions |
| Combination Therapies | Patents for using Vitamin A compounds with other drugs. | Active | Synergistic treatments for cancer, infectious diseases |
What are the regulatory considerations and market approval pathways for Vitamin A-based drugs?
The regulatory pathway for Vitamin A-based drugs depends on their intended use and formulation.
- Dietary Supplements: Vitamin A for general nutritional supplementation is regulated as a dietary supplement in many jurisdictions, including the U.S. under the Dietary Supplement Health and Education Act (DSHEA) of 1994. Manufacturers are responsible for ensuring safety and accurate labeling, but pre-market approval by regulatory bodies like the U.S. Food and Drug Administration (FDA) is not required for claims of general well-being.
- Pharmaceuticals for Specific Indications: Drugs containing Vitamin A or its derivatives for treating specific diseases (e.g., isotretinoin for severe acne) follow the full pharmaceutical regulatory pathway. This requires extensive pre-clinical and clinical trials to demonstrate safety and efficacy, followed by a New Drug Application (NDA) or equivalent submission to regulatory agencies such as the FDA, European Medicines Agency (EMA), or Pharmaceuticals and Medical Devices Agency (PMDA) in Japan.
- Orphan Drug Designation: For rare diseases where Vitamin A derivatives might be explored, orphan drug designation can provide incentives, including market exclusivity for a period after approval.
- Teratogenicity Concerns: For retinoids like isotretinoin, stringent risk management programs (e.g., iPLEDGE in the U.S.) are mandated due to their known teratogenicity. These programs involve strict controls on prescribing, dispensing, and patient monitoring to prevent fetal exposure. Such regulatory burdens significantly impact market access and commercialization strategies.
The approval process for novel indications or advanced formulations is time-consuming and costly, often spanning several years and requiring substantial investment in clinical research.
What is the global market size and projected growth for Vitamin A pharmaceutical products?
The global market for Vitamin A as a pharmaceutical product is segmented, with distinct market sizes and growth trajectories for its various applications.
- Nutritional Supplementation: The market for Vitamin A in dietary supplements is mature, with steady demand driven by general health awareness and deficiency correction in specific populations. This segment is valued in the hundreds of millions of dollars globally and experiences low single-digit annual growth.
- Dermatology (Isotretinoin and topical retinoids): This segment represents a significant portion of the pharmaceutical Vitamin A market. The market for isotretinoin alone is substantial, with sales driven by its efficacy in treating severe acne. The global isotretinoin market was estimated to be approximately $1.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4-5% through 2028 [6]. The market for topical retinoids for acne and anti-aging is also considerable and growing.
- Oncology and Other Emerging Indications: These segments are currently smaller but represent areas of significant growth potential, driven by ongoing research and development. While difficult to quantify precisely due to the research-stage nature of many applications, the therapeutic potential in these areas could unlock multi-billion dollar markets in the future.
Table 2: Estimated Global Market Size and Growth for Key Vitamin A Pharmaceutical Segments
| Pharmaceutical Segment | Estimated 2022 Market Size (USD Billion) | Projected 2023-2028 CAGR | Key Drivers |
|---|---|---|---|
| Nutritional Supplementation | $0.3 - $0.5 | 2-3% | General health awareness, deficiency management |
| Dermatology (Isotretinoin & Topicals) | $1.5 - $2.0 | 4-5% | High efficacy for acne, anti-aging demand, expanding indications |
| Oncology & Emerging Indications | $0.1 - $0.3 (current) | 8-12%+ (projected) | Advancements in retinoid signaling research, novel cancer therapies, unmet needs |
| Total Pharmaceutical Vitamin A Market | $1.9 - $2.8 | 4-6% | Diverse applications, ongoing R&D, increasing healthcare access |
Note: Figures are estimates and can vary based on market research reports.
The overall pharmaceutical Vitamin A market is projected to grow from an estimated $1.9 billion to $2.8 billion in 2022 to approximately $2.6 billion to $4.0 billion by 2028, with a blended CAGR of 4-6%. Growth will be primarily fueled by the established dermatology segment and the high-potential, albeit nascent, oncology and emerging therapeutic areas.
What are the key competitive forces and market challenges?
The Vitamin A pharmaceutical market faces several competitive forces and challenges:
- Generic Competition: For established Vitamin A drugs and their widely used derivatives like isotretinoin, generic competition is intense. Once primary patents expire, multiple generic manufacturers enter the market, driving down prices and squeezing profit margins for originators.
- Price Pressure: Healthcare cost containment measures by governments and insurers globally put pressure on drug prices, impacting the profitability of even novel Vitamin A formulations.
- Development of Alternative Therapies: For conditions treated by Vitamin A derivatives, new therapeutic modalities are constantly being developed. For example, in acne treatment, new antibiotics, anti-androgens, and biologics compete with isotretinoin. In oncology, a wide array of targeted therapies and immunotherapies present alternatives.
- Regulatory Scrutiny and Safety Concerns: The known teratogenicity of certain retinoids necessitates stringent regulatory oversight and risk management programs. This adds complexity and cost to market access and patient treatment. Any new safety signals or adverse events can lead to significant market withdrawal or restrictions.
- R&D Investment and Risk: Developing novel formulations or new therapeutic indications for Vitamin A requires substantial investment in research and clinical trials. The high failure rate in drug development and the lengthy approval timelines represent significant financial risks.
- Supply Chain and Manufacturing: Ensuring a consistent and high-quality supply of Vitamin A and its derivatives, especially for highly regulated pharmaceutical applications, requires robust manufacturing processes and a resilient supply chain. Fluctuations in raw material costs can also impact profitability.
- Physician and Patient Awareness: For new indications or advanced formulations, educating healthcare professionals and patients about the benefits and risks is crucial for market adoption.
What are the financial considerations for investing in Vitamin A-based pharmaceutical companies?
Investing in companies involved with Vitamin A pharmaceuticals requires a nuanced approach, considering the diverse nature of its applications and the maturity of its market segments.
- Established Players: Companies focused on generic Vitamin A supplements or established drugs like isotretinoin are likely to offer lower growth but stable revenue streams, with profitability dependent on manufacturing efficiency and market share. Their financial performance is often tied to volume and cost management.
- Specialty Pharmaceutical Companies: Firms developing novel formulations or patented delivery systems for Vitamin A derivatives may offer higher growth potential. Investments here carry higher risk, dependent on successful clinical trials, regulatory approvals, and market penetration against existing treatments. Their financial models rely on premium pricing for innovative products.
- Biotechnology and R&D Focused Firms: Companies exploring Vitamin A derivatives for novel indications like oncology or autoimmune diseases represent the highest risk and potentially highest reward. Their financial trajectory is heavily tied to R&D pipeline progress, partnership deals, and eventual drug approvals. Valuations are speculative until late-stage clinical data emerges.
- Valuation Metrics: Key metrics to consider include:
- Revenue Growth: Differentiate between growth in mature segments (low single digits) and emerging segments (high double digits if successful).
- Profit Margins: Examine gross and net profit margins, which will vary significantly between generic manufacturers and innovators with patent protection.
- R&D Expenditure: For companies focused on new indications, R&D spending as a percentage of revenue is critical. Sustained, well-managed R&D is essential.
- Patent Portfolio Strength: Assess the breadth, depth, and remaining lifespan of patents covering key compounds, formulations, and indications.
- Market Access and Reimbursement: Analyze a company's ability to secure favorable market access and reimbursement for its products, especially for novel uses.
- Cash Burn Rate and Funding: For R&D-heavy companies, evaluate their cash reserves, burn rate, and ability to secure future funding rounds or partnerships.
Companies with a diversified portfolio across different Vitamin A applications, or those with strong patent protection on novel delivery systems or late-stage clinical assets for unmet medical needs, are likely to present more attractive investment opportunities. However, thorough due diligence on specific product pipelines, competitive landscapes, and regulatory risks is paramount.
Key Takeaways
- Vitamin A's pharmaceutical applications are diverse, ranging from essential nutritional supplementation and dermatology to emerging roles in oncology and immunology.
- The patent landscape is bifurcated, with expired foundational patents for basic Vitamin A and active, growing patent activity in novel formulations, delivery systems, and specific therapeutic indications.
- Regulatory pathways vary significantly from dietary supplements to prescription drugs, with stringent risk management programs required for certain retinoids due to teratogenicity.
- The global market for pharmaceutical Vitamin A is projected to reach $2.6 billion to $4.0 billion by 2028, driven by the established dermatology segment and growth in emerging therapeutic areas.
- Competitive forces include genericization, price pressure, development of alternative therapies, and stringent regulatory oversight.
- Investment in Vitamin A-based pharmaceuticals requires evaluating companies based on their position within specific market segments, the strength of their patent portfolios, R&D pipeline, and ability to navigate regulatory and market access challenges.
Frequently Asked Questions
- Are there any major new therapeutic breakthroughs anticipated for Vitamin A in the next five years? Research into retinoid signaling pathways continues to identify potential new applications in areas like autoimmune diseases, neurodegenerative disorders, and specific types of cancer. While breakthroughs are difficult to predict, ongoing clinical trials may yield positive results for novel indications.
- What is the primary risk associated with investing in companies developing Vitamin A-based cancer therapies? The primary risk is the high failure rate in oncology drug development, coupled with intense competition from numerous other drug classes. Demonstrating superior efficacy and safety compared to existing standards of care is a significant hurdle.
- How do advancements in drug delivery systems specifically impact the market for Vitamin A? Improved drug delivery systems, such as nanoparticles or sustained-release formulations, can enhance therapeutic efficacy, reduce side effects (like skin irritation from topical retinoids), and improve patient compliance. These innovations create opportunities for patent protection and premium pricing for new product generations.
- What impact does the expiry of key patents for isotretinoin have on market dynamics? Patent expiry for isotretinoin has led to significant generic competition, driving down prices and increasing market accessibility. Companies that historically manufactured branded isotretinoin must focus on developing newer formulations, combination therapies, or exploring different therapeutic indications to maintain market share and profitability.
- What is the regulatory status of Vitamin A in over-the-counter versus prescription products? Vitamin A for general nutritional supplementation is typically regulated as a dietary supplement, requiring manufacturers to ensure safety and good manufacturing practices but not pre-market approval for efficacy claims. Vitamin A derivatives used to treat specific medical conditions, like isotretinoin for severe acne, are regulated as prescription drugs, demanding rigorous clinical trials and FDA/EMA approval.
Citations
[1] Sommer, A. (2008). Vitamin A. In Modern Nutrition in Health and Disease (Vol. 10, pp. 267-281). Lippincott Williams & Wilkins. [2] Dimas, K., & Jones, P. A. (2004). Vitamin A analogs in cancer prevention and treatment. Current Opinion in Investigational Drugs, 5(7), 748-757. [3] Torchilin, V. P. (2005). Targeted drug delivery—the concept of a macromolecular drug carrier. Pure and Applied Chemistry, 77(11), 921-929. [4] Psaroudakis, G., & Athanasiou, E. (2016). Retinoids and the immune system. Immunopharmacology and Immunotoxicology, 38(1), 71-79. [5] Huang, K., Huang, L., Li, Y., & Xu, C. (2019). Vitamin A and its role in the treatment of tumors. Journal of Cancer, 10(6), 1462-1470. [6] Grand View Research. (2023). Isotretinoin Market Size, Share & Trends Analysis Report By Application (Acne, Others), By Region, And Segment Forecasts, 2023 - 2030. Retrieved from [Grand View Research website] (Note: Specific URL omitted for brevity as it's a direct report reference).
More… ↓