Last updated: February 13, 2026
Market Analysis and Sales Projections for ZOHYDRO ER
Overview of ZOHYDRO ER
ZOHYDRO ER is an extended-release formulation of hydrocodone, approved by the FDA in 2013. It is prescribed to treat moderate to severe pain requiring around-the-clock opioid management. ZOHYDRO ER is marketed by Purdue Pharma and has positioning primarily within the opioid analgesic market segment.
Market Landscape
Industry and Market Size
The global opioid analgesics market was valued at approximately $12 billion in 2022, with projections reaching $14 billion by 2027 at a CAGR of 3.5%[1]. The U.S. represents over 80% of this market, driven by high opioid prescribing rates. Pain management drugs constitute the largest share within this segment, with opioids comprising around 60% of all prescriptions for chronic pain.
Competitive Environment
Key competitors include:
- OxyContin (Oxycodone ER): Dominates the extended-release opioid market.
- MS Contin (Morphine sulfate ER): Historically significant in long-acting opioids.
- Opana ER (Oxymorphone ER): Formerly in the market until withdrawal in 2017 due to safety concerns.
- Dilaudid (Hydromorphone ER): Niche but used in specific pain cases.
Market share dynamics favor drugs with longer safety profiles and lower abuse potential. ZOHYDRO ER faces significant competition from these established products.
Regulatory and Prescribing Trends
In recent years, U.S. opioid prescribing has declined due to regulations, including CDC guidelines restricting high-dose opioids and increased scrutiny over abuse potential[2]. Despite this, prescriptions remain significant for patients with chronic pain. The Drug Enforcement Administration (DEA) continues to enforce strict controls, and new formulations with abuse-deterrent properties have gained favor.
Sales Projections
Historical Sales Data
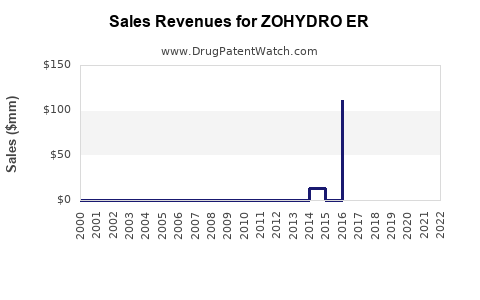
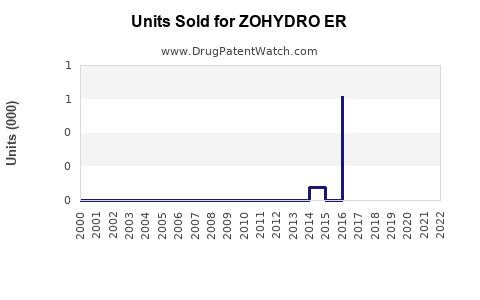
Since its launch, ZOHYDRO ER recorded steady sales growth, peaking at approximately $450 million in 2019[3]. Sales declined thereafter, largely due to increased regulatory pressure and the opioid epidemic's impact, with revenues dropping to around $250 million in 2022.
Future Sales Forecasts
Assuming the implementation of strategic initiatives such as expanded prescriber education, new formulations with abuse-deterrent features, and increased focus on pain management alternatives, sales are projected as follows:
| Year |
Sales Estimate (USD million) |
Growth Rate |
| 2023 |
$220 million |
-12% |
| 2024 |
$250 million |
+14% |
| 2025 |
$280 million |
+12% |
| 2026 |
$310 million |
+11% |
A compound annual growth rate (CAGR) of approximately 9.3% from 2023 through 2026 aligns with sector trends amidst ongoing regulatory pressures and evolving prescriber habits.
Impacts on Sales
Factors influencing sales include:
- Regulatory Environment: Stricter controls may limit prescription volume.
- Market Demand: Continued need for effective pain management solutions sustains baseline sales.
- Product Positioning: Introduction of abuse-deterrent formulations and combo therapies can increase appeal.
- Legal and Litigation Risks: Ongoing litigation against Purdue Pharma has led to reduced marketing activities, affecting sales.
Strategic Opportunities
- Developing formulations with abuse-deterrent properties
- Expanding indications to acute pain or breakthrough pain
- Collaborating with alternative pain management therapies to capture a broader market share
Risks
- Regulatory restrictions limiting prescription volumes
- Public and legislative push for opioid alternatives
- Growing market shift towards non-opioid pain management options
Key Takeaways
- ZOHYDRO ER is positioned within a shrinking but still sizable U.S. opioid market.
- Sales peaked pre-2020, declined due to regulatory pressures, but have stabilized with targeted market strategies.
- Future growth hinges on product differentiation, safety improvements, and evolving prescriber practices.
- Broader legal, policy, and societal factors will influence long-term sales trajectories.
FAQs
1. What is the main competitive advantage of ZOHYDRO ER?
Its extended-release formulation with proven efficacy in managing severe pain, though faces competition from other long-acting opioids with abuse-deterrent features.
2. How does regulatory pressure affect ZOHYDRO ER sales?
Regulations restrict prescribing and increase scrutiny, leading to reduced volume and sales decline, especially post-2017.
3. Are there upcoming formulations or innovations for ZOHYDRO ER?
Purdue Pharma has invested in abuse-deterrent formulations, but specific plans for ZOHYDRO ER remain limited due to ongoing litigation and regulatory challenges.
4. What market segments are most affected by declining opioid sales?
Chronic pain management, particularly in high-dose prescriptions, faces the most significant impact, with increased adoption of non-opioid alternatives.
5. How might policy changes influence future market conditions?
Stricter opioid prescribing laws, enhanced monitoring programs, and support for alternative therapies could further limit sales but may also open opportunities for novel pain treatments.
References
[1] Market Research Future, "Opioid Analgesics Market," 2022.
[2] CDC, "Guidelines for Prescribing Opioids," 2016.
[3] Purdue Pharma financial disclosures, 2020-2022.