Share This Page
Drug Sales Trends for TELMISARTAN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TELMISARTAN (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
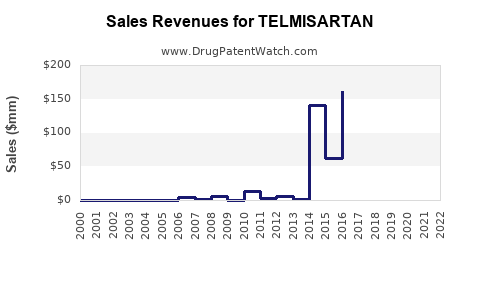
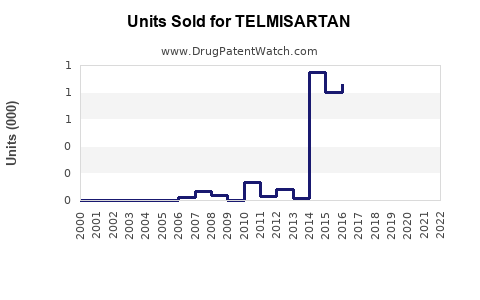
Annual Sales Revenues and Units Sold for TELMISARTAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TELMISARTAN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Telmisartan: Patent Landscape and Market Outlook
Telmisartan, a potent angiotensin II receptor blocker (ARB), is established in the treatment of hypertension and cardiovascular risk reduction. The drug's patent exclusivity has expired in key markets, paving the way for generic competition and influencing its market trajectory. This analysis examines the current patent landscape, competitive environment, and future sales projections for telmisartan.
What is the Current Patent Status of Telmisartan?
The foundational patents protecting telmisartan have expired globally. The primary patent for telmisartan, U.S. Patent No. 5,591,753, which claimed the compound itself, expired in October 2012 [1]. Related formulation and method of use patents have also largely lapsed. This has led to the widespread availability of generic versions of telmisartan in major pharmaceutical markets.
Key Patent Expirations:
- Composition of Matter Patent: Expired in 2012 in the U.S. and corresponding periods internationally based on priority dates.
- Formulation Patents: Many of these also expired, allowing for diverse generic product offerings.
- Method of Use Patents: Patents related to specific indications, such as cardiovascular risk reduction (e.g., the ONTARGET study outcomes), have also faced challenges and eventual expiration or limited enforceability against generic entrants.
The absence of active patent protection allows generic manufacturers to produce and market telmisartan, intensifying price competition. This has significantly impacted the revenue generated by branded telmisartan products.
Who are the Key Players in the Telmisartan Market?
The telmisartan market is characterized by a mix of originator companies, generic manufacturers, and contract manufacturing organizations (CMOs).
Originator:
- Boehringer Ingelheim: The original developer of telmisartan, marketed under the brand name Micardis. While brand sales have declined due to generic erosion, Boehringer Ingelheim may still hold market share through authorized generics or residual branded sales in specific regions.
Major Generic Competitors:
The generic space is highly fragmented. Key players include:
- Teva Pharmaceutical Industries: A significant global generics player with a strong presence in various therapeutic areas.
- Mylan N.V. (now Viatris): Another large generics company with a broad product portfolio.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with substantial generic drug offerings.
- Sun Pharmaceutical Industries: India's largest pharmaceutical company, known for its generic and branded generics.
- Lupin Limited: A global pharmaceutical company with a focus on generics.
- Aurobindo Pharma: An Indian company with a significant presence in the U.S. and European generics markets.
Contract Manufacturing Organizations (CMOs):
Many smaller generic companies or even larger ones may rely on CMOs for the manufacturing of telmisartan active pharmaceutical ingredients (APIs) or finished dosage forms. These CMOs operate under strict regulatory oversight.
The competitive landscape is driven by price, supply chain reliability, and regulatory approvals in different countries. Generic manufacturers continually seek to optimize production costs to maintain profitability in a competitive market.
What are the Current Sales and Market Size for Telmisartan?
The global market for telmisartan has experienced a significant decline in revenue from its peak due to the influx of generic competition. However, the volume of prescriptions remains substantial due to its established efficacy and affordability as a generic drug.
Global Market Size (Estimated):
- Peak Branded Sales (Micardis): Estimated to have reached over $1 billion annually before significant generic entry.
- Current Generic Market Value: While exact figures are difficult to pinpoint due to the fragmented nature of the generics market and varying regional pricing, the global market value for telmisartan, encompassing all generic versions, is estimated to be in the range of $500 million to $800 million annually. This figure represents the total revenue generated by all manufacturers selling telmisartan products worldwide.
Regional Market Dynamics:
- United States: The largest pharmaceutical market, with a high volume of telmisartan prescriptions. The average selling price (ASP) of generic telmisartan is low, driven by intense competition among multiple suppliers.
- Europe: Similar to the U.S., with a mature generics market and significant price pressures.
- Emerging Markets (Asia, Latin America, Africa): These markets represent growing volumes for telmisartan, driven by increasing access to healthcare and the affordability of generic medications. Pricing in these regions can vary significantly based on local healthcare policies and payer structures.
Volume vs. Value:
It is crucial to distinguish between sales volume and sales value. While the total dollar value of the telmisartan market has decreased from its branded peak, the number of tablets dispensed and patients treated has likely remained high or even increased due to the accessibility of generics.
What are the Future Sales Projections for Telmisartan?
Future sales projections for telmisartan are expected to be characterized by continued volume growth in emerging markets, offset by persistent price erosion in developed markets. The overall market value is unlikely to see substantial increases, primarily driven by generic competition.
Key Factors Influencing Future Sales:
- Generic Competition: This remains the dominant factor. The absence of patent protection will ensure ongoing price pressure and limit any potential for significant price increases. Competition among numerous generic manufacturers will keep prices low.
- Emerging Market Growth: As healthcare infrastructure and access improve in countries across Asia, Africa, and Latin America, the demand for affordable antihypertensive medications like telmisartan is projected to rise. This will contribute to the overall volume sold.
- Established Clinical Use: Telmisartan is a well-established therapy with a strong evidence base for both hypertension and cardiovascular risk reduction. Its proven efficacy and safety profile ensure its continued use by physicians and patients.
- Combination Therapies: Telmisartan is frequently used in combination with other antihypertensive agents. The demand for these fixed-dose combinations (FDCs) will indirectly support telmisartan sales.
- Regulatory Policies: Government-imposed pricing regulations, tender systems, and formulary restrictions in various countries will continue to influence pricing and market access.
Projected Market Value:
- Short-Term (1-3 years): The market value is expected to remain relatively stable, with minor fluctuations.
- Medium-Term (3-5 years): A slight decline in overall market value might occur as price pressures intensify, even with volume increases in emerging markets.
- Long-Term (5+ years): The market will likely stabilize at a lower revenue level, sustained by its role as an essential, affordable generic medication.
Estimated Annual Sales Projection (Global Market Value):
- 2024-2026: $500 million - $700 million
- 2027-2029: $450 million - $650 million
These projections are based on current market trends, anticipated generic pricing behavior, and projected volume growth in key regions.
What is the Competitive Landscape for Telmisartan Combinations?
Telmisartan is often prescribed in fixed-dose combination (FDC) therapies to improve patient adherence and manage blood pressure more effectively. The competitive landscape for these combinations is dynamic and includes branded and generic products.
Key Telmisartan Combination Products:
- Telmisartan + Hydrochlorothiazide (HCTZ): A common combination for hypertension.
- Branded: Micardis HCT (Boehringer Ingelheim).
- Generics: Numerous manufacturers offer generic versions of telmisartan/HCTZ. The market is highly competitive, with multiple suppliers driving down prices.
- Telmisartan + Amlodipine: Another widely used combination.
- Branded: Twynsta (Boehringer Ingelheim).
- Generics: Generic telmisartan/amlodipine is available from various companies.
- Telmisartan + Ramipril: While less common than the other two, this combination is also available.
- Generics: Generic versions exist.
Competitive Dynamics of FDCs:
- Patent Expirations: Similar to monotherapy telmisartan, the patent protection for many FDCs has expired, leading to generic availability.
- Pricing: FDCs face intense price competition. Generic manufacturers leverage their ability to produce cost-effectively.
- Market Penetration: The uptake of FDCs is driven by physician preference, patient convenience, and formulary inclusion.
- Newer Combinations: While telmisartan combinations are established, ongoing research explores novel combinations for more challenging hypertension profiles or co-morbidities, which could shift the competitive landscape over time. However, significant new branded FDCs based on telmisartan are unlikely given the drug's mature lifecycle.
The market for telmisartan FDCs is expected to follow a similar trajectory to telmisartan monotherapy, with stable to declining revenue in developed markets and volume growth in emerging economies.
What are the Regulatory Considerations for Telmisartan?
Regulatory considerations for telmisartan are critical for both originator and generic manufacturers. Compliance with evolving pharmaceutical regulations impacts market access, pricing, and product quality.
Key Regulatory Aspects:
- Good Manufacturing Practices (GMP): All manufacturers must adhere to stringent GMP guidelines established by regulatory bodies like the FDA (U.S.), EMA (Europe), and PMDA (Japan). This ensures the quality, safety, and efficacy of the drug product.
- Abbreviated New Drug Applications (ANDAs) / Marketing Authorizations: Generic manufacturers must submit ANDAs (in the U.S.) or equivalent marketing authorization applications in other regions, demonstrating bioequivalence to the reference listed drug.
- Bioequivalence Studies: These studies are essential to prove that the generic drug performs comparably to the brand-name drug in terms of absorption and bioavailability.
- Post-Marketing Surveillance: Regulatory agencies require ongoing monitoring of drug safety once it is on the market. This includes pharmacovigilance activities to detect and report adverse events.
- Intellectual Property and Patent Litigation: While core patents have expired, disputes can arise over secondary patents (e.g., formulations, manufacturing processes). Litigation can delay generic entry or impact market exclusivity for generic players.
- Labeling Requirements: Generic drug labels must be consistent with the approved labeling of the reference drug, with certain permitted differences.
- Quality Control and Impurity Profiling: Manufacturers must meticulously control impurities in the API and finished product to meet regulatory limits. Changes in impurity profiles can trigger regulatory scrutiny.
- Emerging Market Regulations: Regulatory frameworks in developing countries can vary, requiring manufacturers to navigate different approval processes and quality standards.
The ongoing scrutiny of pharmaceutical quality and manufacturing processes by regulatory bodies means that manufacturers must maintain robust quality management systems to remain compliant and ensure continued market access for telmisartan.
Key Takeaways
- Telmisartan's foundational patents have expired globally, enabling widespread generic competition.
- The global telmisartan market, currently valued at an estimated $500 million to $800 million annually, is dominated by generic manufacturers.
- Future market growth will be driven by increasing volume in emerging markets, while developed markets will experience persistent price erosion.
- Telmisartan is frequently used in fixed-dose combinations, with telmisartan/HCTZ and telmisartan/amlodipine being the most common.
- Regulatory compliance, including GMP and bioequivalence demonstration, is critical for all market participants.
Frequently Asked Questions
- Will there be any new patent-protected indications for telmisartan? While secondary patents for specific formulations or combination therapies might exist, the development of entirely new, patent-protected indications for telmisartan is unlikely given its mature lifecycle and well-established therapeutic profile.
- What is the primary driver for generic telmisartan sales volume? The primary driver is its established efficacy in managing hypertension and reducing cardiovascular risk, coupled with its affordability as a generic medication, making it accessible to a broad patient population.
- How does the price of generic telmisartan compare to its branded counterpart at its peak? Generic telmisartan is significantly less expensive, often by 80-90%, compared to the peak prices of the branded product (Micardis), reflecting the impact of generic competition.
- Are there any significant supply chain risks for telmisartan API? The supply chain for telmisartan API is global and involves multiple manufacturers, primarily in India and China. While generally robust, risks can include geopolitical events, raw material shortages, or regulatory enforcement actions impacting key API producers.
- How do fixed-dose combinations of telmisartan impact the overall market? Telmisartan FDCs contribute to the overall telmisartan volume and market value by offering convenience and improved adherence, thereby sustaining demand for telmisartan as a component.
Sources
[1] U.S. Patent No. 5,591,753. (1996). Substituted benzimidazoles. Assignee: Boehringer Ingelheim Pharma KG.
More… ↓
