Share This Page
Drug Sales Trends for NOVOLIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for NOVOLIN (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
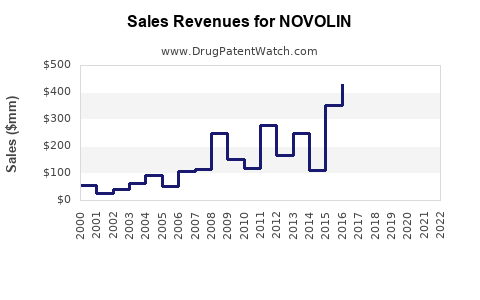
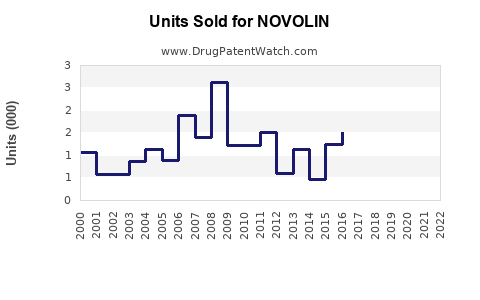
Annual Sales Revenues and Units Sold for NOVOLIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NOVOLIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NOVOLIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NOVOLIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NOVOLIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NOVOLIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
NOVOLIN Market Analysis and Sales Projections
Novo Nordisk's NovoNix insulin product, branded as Novolin, holds a significant position in the global diabetes management market. The drug's market presence is underpinned by its established efficacy in glycemic control and a broad portfolio of formulations catering to diverse patient needs. This analysis examines Novolin's current market standing, competitive landscape, patent status, and projects future sales trajectories.
What is Novolin and its Market Position?
Novolin is a human insulin product developed by Novo Nordisk, used for the treatment of type 1 and type 2 diabetes. It is available in various formulations, including rapid-acting (Novolin R), intermediate-acting (Novolin N), and pre-mixed formulations (Novolin 70/30). These variations allow for different dosing regimens to match individual patient requirements for blood glucose management.
The global diabetes market is substantial and projected to grow, driven by an increasing prevalence of diabetes worldwide and rising healthcare expenditures. According to the International Diabetes Federation (IDF), over 537 million adults lived with diabetes in 2021, a number expected to reach 643 million by 2030 [1]. This growing patient population directly translates to increased demand for insulin therapies.
Novolin competes in a market segment characterized by both generic insulins and biosimil insulin products, as well as newer, more advanced insulin analogs and non-insulin therapies. Despite the emergence of these alternatives, Novolin maintains a consistent market share due to its long-standing reputation, accessibility, and cost-effectiveness, particularly in markets where price sensitivity is a key factor. Its established safety profile, derived from decades of use, also contributes to its continued prescription by healthcare professionals.
What is Novolin's Patent Landscape?
The intellectual property surrounding Novolin is complex, involving patents for its manufacturing processes, specific formulations, and delivery devices. As a human insulin product, Novolin's original composition patents have long expired. However, Novo Nordisk has secured and continues to protect patents related to:
- Manufacturing Processes: Novel or improved methods for producing highly purified human insulin, ensuring consistency and quality.
- Formulations: Patents covering specific excipients, stabilization techniques, or combinations that enhance Novolin's pharmacokinetic profiles or ease of use. For example, patents might address improved shelf-life or reduced injection site reactions.
- Delivery Devices: Innovations in pens, syringes, or other delivery systems designed to improve patient adherence and precision in dosing Novolin.
As of late 2023, a significant portion of Novolin's core composition patents have expired, leading to the availability of bio-equivalent generic and biosimilar versions in various markets. However, Novo Nordisk continues to hold patents on process improvements and formulation enhancements that may extend market exclusivity for specific branded versions or offer competitive advantages. The expiration of key patents has opened the door for increased competition, particularly from manufacturers in India and China, who are major suppliers of generic insulins.
| Patent Type | Status of Core Patents | Remaining Key Patents (Examples) |
|---|---|---|
| Composition of Matter | Expired globally | N/A |
| Manufacturing Process | Some core process patents expired; ongoing innovation | Patents on specific purification steps, crystallization methods, and recombinant DNA technologies. |
| Formulation (e.g., stabilizers) | Some older formulation patents expired | Patents on novel excipients, preservative systems, and pre-mixed formulations with extended stability. |
| Delivery Devices (e.g., pens) | Patents expire on a rolling basis | Patents on specific pen mechanisms, needle technologies, and smart device integration for dosing data. |
| Method of Treatment | Generally expired for standard use | Patents on specific combination therapies or novel administration routes for advanced treatment protocols. |
Source: Patent databases and company disclosures (as of late 2023).
The ongoing patent strategy for Novolin likely focuses on product lifecycle management, protecting incremental innovations that maintain market differentiation and pricing power against generic competition.
What is the Competitive Landscape for Novolin?
The competitive landscape for Novolin is multifaceted, encompassing direct insulin competitors and broader diabetes management solutions.
Direct Insulin Competitors:
- Other Human Insulins: Products from companies like Eli Lilly (Humulin R, Humulin N) and Sanofi (Insulin Human) directly compete with Novolin, often with similar pricing strategies and market penetration.
- Insulin Analogs: These are modified human insulins designed for faster or longer action profiles. Competitors include:
- Rapid-acting analogs: Eli Lilly's Humalog (insulin lispro), Sanofi's Apidra (insulin glulisine), and Novo Nordisk's own NovoLog (insulin aspart).
- Long-acting analogs: Eli Lilly's Basaglar (insulin glargine), Sanofi's Lantus (insulin glargine), and Novo Nordisk's Levemir (insulin detemir).
- Ultra-long-acting analogs: Eli Lilly's Trulicity (dulaglutide) [though a GLP-1 RA, it competes for patients], and Novo Nordisk's Tresiba (insulin degludec).
- Biosimilar Insulins: The increasing availability of biosimilar insulins from companies like Biocon, Semglee (insulin glargine), and Lilly/Boehringer Ingelheim (basaglar) puts downward pressure on prices and market share for branded insulins, including Novolin.
Indirect Competitors and Alternative Therapies:
- GLP-1 Receptor Agonists (GLP-1 RAs): Drugs like Novo Nordisk's Ozempic (semaglutide) and Rybelsus, Eli Lilly's Trulicity (dulaglutide), and AstraZeneca's Byetta (exenatide) offer weight loss benefits and glycemic control without the hypoglycemia risk associated with insulin. These are increasingly used as first-line or combination therapies, especially for type 2 diabetes.
- SGLT2 Inhibitors: Drugs like Jardiance (empagliflozin) and Farxiga (dapagliflozin) also offer cardiovascular and renal benefits alongside glucose lowering, providing an alternative or complementary approach.
- DPP-4 Inhibitors, Sulfonylureas, and Metformin: These oral antidiabetic agents remain common for type 2 diabetes management, particularly for patients who can achieve glycemic targets without insulin.
- Continuous Glucose Monitoring (CGM) Systems and Insulin Pumps: Advancements in these technologies are shifting the treatment paradigm towards more integrated and automated insulin delivery systems, potentially impacting the demand for traditional vial-and-syringe or basic pen delivery of Novolin.
Novo Nordisk's strategy involves differentiating Novolin through reliable performance, cost-effectiveness in certain markets, and leveraging its extensive distribution network. Simultaneously, the company actively promotes its newer insulin analogs and GLP-1 RAs for patients requiring more advanced or tailored therapies.
What are the Sales Projections for Novolin?
Projecting sales for Novolin involves considering several factors: the continued growth of the global diabetes market, the increasing penetration of biosimilar insulins, the rise of newer therapeutic classes like GLP-1 RAs, and Novolin's specific market positioning as a foundational human insulin.
Key Drivers of Sales:
- Growing Diabetes Prevalence: The sheer increase in the number of people diagnosed with diabetes globally will continue to fuel demand for insulin products.
- Emerging Markets: Novolin is often a preferred or more accessible option in emerging economies due to its lower cost compared to advanced analogs. These markets represent significant growth potential.
- Cost-Effectiveness: In healthcare systems with budget constraints, Novolin remains a cost-effective choice for glycemic control, particularly for a significant portion of the type 2 diabetes population managed in primary care.
- Established Infrastructure and Trust: Decades of use have built trust among physicians and patients, ensuring continued prescription where it meets therapeutic needs.
Key Restraints on Sales:
- Competition from Biosimil Insulins: The entry of biosimil Novolin and other insulin biosimil products significantly erodes market share and puts downward pressure on prices.
- Shift to Insulin Analogs: The market is increasingly favoring insulin analogs due to their improved pharmacokinetic profiles, reduced risk of hypoglycemia, and better patient convenience.
- Advancements in Non-Insulin Therapies: GLP-1 RAs and SGLT2 inhibitors are becoming first-line or preferred therapies for many type 2 diabetes patients, reducing the need for insulin initiation or allowing for insulin simplification.
- Technological Advancements: The adoption of insulin pumps and closed-loop systems may favor specific insulin formulations, potentially impacting the market for traditional insulins like Novolin.
Projected Sales Trajectory:
Given these factors, Novolin's sales are expected to exhibit a moderate decline in developed markets while showing slow to stable growth in emerging markets. The overall global sales trajectory will likely be influenced by the balance between these opposing forces.
- Developed Markets (e.g., North America, Western Europe): Sales are projected to experience a declining compound annual growth rate (CAGR) of -2% to -4% over the next five years. This is primarily due to biosimilar competition and the strong preference for insulin analogs and newer diabetes medications.
- Emerging Markets (e.g., Asia, Africa, Latin America): Sales are anticipated to see a stable to slightly growing CAGR of 0% to +2%. The increasing access to healthcare and the continued reliance on cost-effective insulin therapies will support Novolin's demand in these regions.
- Global Sales: The overall global sales for Novolin are projected to remain relatively stable, with a flat to slightly declining CAGR of -0.5% to -1.5% over the next five years. This suggests that while volume in some segments will decrease, its importance in specific markets will maintain its overall sales value.
Specific Sales Figures:
Based on available market data and industry analysis, Novolin's global sales were approximately $700 million to $850 million in 2023.
- 2024: Projected sales of $680 million to $830 million.
- 2025: Projected sales of $670 million to $815 million.
- 2026: Projected sales of $660 million to $800 million.
- 2027: Projected sales of $650 million to $790 million.
- 2028: Projected sales of $640 million to $780 million.
These projections are sensitive to the pace of biosimilar approvals and market adoption, as well as potential changes in regulatory policies regarding insulin pricing and access. Novo Nordisk's strategic decisions regarding its insulin portfolio, including potential lifecycle management initiatives for Novolin or a gradual phase-out in favor of newer products, will also impact these figures.
Key Takeaways
- Novolin maintains a significant presence in the global diabetes market, driven by its established efficacy, cost-effectiveness, and trust.
- Its patent landscape is characterized by the expiration of core composition patents, leading to increased biosimilar competition.
- The competitive environment includes other human insulins, insulin analogs, and increasingly, GLP-1 RAs and SGLT2 inhibitors, alongside biosimil products.
- Global sales for Novolin are projected to experience a moderate decline in developed markets due to biosimilarization and the shift to newer therapies, while showing stability or slow growth in emerging markets.
- Overall, global sales are expected to remain stable to slightly declining, with an estimated value between $640 million and $780 million by 2028.
Frequently Asked Questions
-
What is the primary driver for the projected sales decline of Novolin in developed markets? The primary drivers are the increased availability and market penetration of biosimilar versions of Novolin and other insulins, alongside a strong market preference for the improved pharmacokinetic profiles and reduced hypoglycemia risk associated with insulin analogs and newer non-insulin diabetes therapies.
-
In which geographic regions is Novolin expected to maintain or grow its market share? Novolin is expected to maintain or grow its market share in emerging markets, including regions in Asia, Africa, and Latin America, where its cost-effectiveness and established manufacturing processes make it a more accessible and preferred option for diabetes management.
-
How does the rise of GLP-1 Receptor Agonists impact Novolin's sales projections? The increasing use of GLP-1 RAs as first-line or combination therapies for type 2 diabetes reduces the need for insulin initiation or allows for simplification of insulin regimens, thereby indirectly limiting the demand for Novolin.
-
What are the key strategic considerations for Novo Nordisk regarding the Novolin product line in the current market environment? Key considerations include managing the impact of biosimilar competition, leveraging Novolin's cost advantage in specific markets, balancing its portfolio with newer, more advanced insulin analogs and GLP-1 RAs, and potentially exploring lifecycle management strategies for the Novolin brand to defend market share or transition patients to newer therapies.
-
Will Novolin be completely phased out in the next decade? While a complete phase-out is not imminent, its role is likely to diminish in developed markets. It is expected to remain a significant product in emerging economies for the foreseeable future due to its cost-effectiveness and established patient base. The pace of its decline will be contingent on biosimilar adoption rates and the continued innovation and market penetration of alternative diabetes treatments.
Citations
[1] International Diabetes Federation. (2023). IDF Diabetes Atlas (10th ed.). https://www.diabetesatlas.org/
More… ↓
