Last updated: February 20, 2026
What is the current market position of HUMIRA?
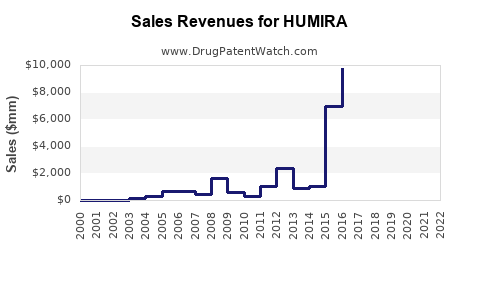
HUMIRA (adalimumab), developed by AbbVie, remains one of the top-selling biologic drugs globally. As of 2022, it generated $21.2 billion in revenue, representing approximately 23% of AbbVie's total sales. It holds the position as the world's best-selling drug, with a dominant share in multiple therapeutic areas, including autoimmune diseases.
How does HUMIRA's market share compare across key indications?
HUMIRA targets several autoimmune conditions: rheumatoid arthritis, psoriatic arthritis, Crohn's disease, ulcerative colitis, hidradenitis suppurativa, and ankylosing spondylitis. It holds roughly:
- 70% of the global TNF-alpha inhibitor market (2022).
- 50-60% market share within each indication in the U.S., depending on the condition.
The drug faces competition from biosimilars introduced in markets like the EU, where biosimilars launched in 2018 and 2019 have eroded its revenue share.
What are the key drivers influencing HUMIRA's sales?
Patent expirations and biosimilar competition
HUMIRA's primary patent expired in the U.S. in January 2023, enabling biosimilar competition. However, AbbVie employed legal strategies to extend market exclusivity, including patent litigation and agreements with biosimilar manufacturers that limited immediate launches.
Launch of adalimumab biosimilars
Biosimilars are launched in Europe, India, and other regions, capturing substantial market share. In the EU, biosimilars have captured over 80% of HUMIRA's original market within two years of entry, significantly reducing sales.
Growth in emerging markets
Emerging markets are experiencing increased biologic adoption, driven by rising autoimmune disease prevalence, healthcare infrastructure expansion, and government policies promoting biosimilars.
New indications and label expansions
AbbVie continues to expand the approved uses, supporting sales. For example, approval for hidradenitis suppurativa in 2015 and ulcerative colitis in 2020 opened additional revenue streams.
What are the sales projections for the next five years?
Assumptions for projections
- Biosimilar competition will increase globally, especially in key markets like the U.S. and EU.
- Patent litigation and exclusivity strategies will delay biosimilar impact in the U.S. until late 2024 or early 2025.
- Growth in emerging markets will offset some loss from mature markets.
Revenue forecasts (2023-2027)
| Year |
Estimated Revenue (USD billion) |
Notes |
| 2023 |
12.5 - 14.0 |
Partial biosimilar entry in the U.S. and EU; sales decline expected. |
| 2024 |
10.0 - 12.0 |
Increased biosimilar competition, especially outside the U.S. |
| 2025 |
8.0 - 10.0 |
Biosimilars gain substantial market share; impact in North America. |
| 2026 |
7.0 - 9.0 |
Continued biosimilar adoption; potential new indications support residual sales. |
| 2027 |
6.5 - 8.0 |
Maturation of biosimilar markets; steady decline expected. |
Key factors influencing projections
- Biosimilar market penetration rates.
- Regulatory and patent litigation outcomes.
- Launch timing in different regions.
- Expanded use of HUMIRA for approved indications.
What strategies does AbbVie use to sustain HUMIRA sales?
- Patent filings to extend exclusivity.
- Securing new indications.
- Developing next-generation products, including biosimilars with improved profiles.
- Pricing adjustments and patient assistance programs.
- Market expansion into less saturated regions.
How will biosimilar entry impact the global market?
Biosimilars are projected to cut HUMIRA's global revenue by 50–60% over the next five years. This is driven by:
- Price reductions of 20-30% upon biosimilar entry.
- Increased access and adoption, especially in cost-sensitive markets.
- Manufacturer strategies such as aggressive marketing and partnership deals.
United States biosimilar market share of adalimumab is expected to reach 60–70% by 2026, with European markets showing similar trends earlier.
Key Takeaways
- HUMIRA remains a top-selling biologic, but patent expirations and biosimilar competition will reduce revenues significantly over the next few years.
- Revenue declines will accelerate from 2024 onward, with estimates showing a 25-35% drop annually.
- Growth strategies include expanding indications, entering new markets, and developing next-generation biologics.
- Biosimilar market penetration varies geographically, heavily impacting the U.S. and Europe.
- The long-term outlook involves transitioning from high-revenue monotherapy to diversified portfolio strategies, including new biologics and small molecules.
FAQs
1. How quickly will biosimilars erode HUMIRA’s sales in the U.S.?
Biosimilars are projected to capture 60-70% of market share by 2026, leading to substantial revenue loss starting from 2023.
2. Which regions will see the fastest biosimilar adoption?
Europe and India already have significant biosimilar presence, with adoption rates exceeding 80%. The U.S. adoption will accelerate from 2024 onwards.
3. How does AbbVie's patent strategy impact sales?
Legal litigation and patent extensions delay biosimilar launches, extending HUMIRA’s market exclusivity until late 2024 or early 2025.
4. Can new indications reverse sales decline?
While they can prolong sales, new indications alone are unlikely to offset biosimilar-driven revenue decreases in the long term.
5. What is the outlook for next-generation biologics?
AbbVie’s development pipeline includes biosimilars and other biologics designed to compete with or replace HUMIRA, aiming to stabilize long-term revenue streams.
References
[1] AbbVie. (2022). HUMIRA revenue and market data. Retrieved from https://www.abbvie.com/investors.html
[2] IQVIA. (2022). Global biologic market analysis. IQVIA Reports.
[3] European Medicines Agency. (2022). Biosimilar adalimumab approvals and market penetration. https://www.ema.europa.eu/
[4] U.S. Food and Drug Administration. (2023). Transparency on biosimilar approvals. https://www.fda.gov/
[5] Statista. (2023). Biologics and biosimilars market forecast. https://www.statista.com