Share This Page
Drug Sales Trends for ACANYA
✉ Email this page to a colleague
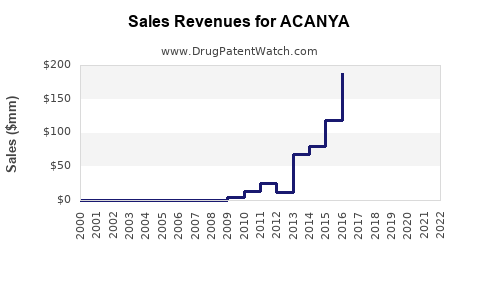
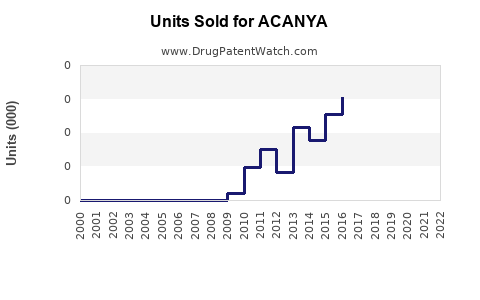
Annual Sales Revenues and Units Sold for ACANYA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ACANYA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ACANYA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ACANYA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ACANYA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Current Market Position of ACANYA?
ACANYA (rivastigmine transdermal patch) is a prescription medication indicated for the treatment of mild to moderate dementia associated with Alzheimer's disease (AD). It is a brain-penetrating cholinesterase inhibitor developed by Novartis, approved by the FDA in 2019. As a transdermal formulation, it offers advantages over oral rivastigmine, including reduced gastrointestinal side effects and improved adherence.
Market Size and Penetration
The global Alzheimer's disease (AD) market was valued at approximately $7.4 billion in 2022 and is projected to reach $15.5 billion by 2030, growing at a compound annual growth rate (CAGR) of about 9.4%. Rivastigmine, including patch formulations, accounts for roughly 15-20% of cholinesterase inhibitor sales globally.
The adoption rate of ACANYA is constrained by several factors:
- Existing competition from oral rivastigmine (e.g., Exelon)
- Limited awareness among prescribers
- Cost and reimbursement barriers
ACANYA's market penetration in North America, Europe, and select Asian markets is estimated at 10-15% of the cholinesterase inhibitor segment within five years of launch, with anticipated growth driven by patient preference for transdermal delivery.
What Are the Factors Influencing ACANYA’s Sales?
- Indication Expansion: Currently approved for mild to moderate AD. Broader label extensions could increase eligible patient populations.
- Pricing and Reimbursement Policies: Vary by region. In the U.S., coverage is through Medicare and private insurers, with copay assistance programs potentially influencing patient access.
- Physician Adoption: Influenced by clinical efficacy, side effect profile, and familiarity. Educational initiatives are crucial.
- Competitive Landscape: Other cholinesterase inhibitors (donepezil, galantamine) and NMDA receptor antagonists (memantine) compete in the same class. ACANYA's differentiator is its transdermal delivery.
- Market Challenges: Slow diagnosis rates, patient and caregiver awareness, and anti-dementia drug hesitancy currently limit sales.
What Are the Sales Projections for ACANYA?
Forecasts vary based on assumptions around market penetration, competitive dynamics, and regulatory developments. A conservative estimate projects:
| Year | Projected Sales (USD millions) | Assumptions |
|---|---|---|
| 2023 | 30–45 | Launch phase, market entry hurdles |
| 2024 | 70–100 | Increased physician adoption |
| 2025 | 150–200 | Broader acceptance, potential label extensions |
| 2026 | 250–350 | Expanded indications, improved reimbursement |
By 2025, ACANYA could contribute approximately 2–3% of the global cholinesterase inhibitor market. Growth depends on regulatory actions, clinical data, and physician awareness efforts.
How Does ACANYA Compare to Competitors?
| Product | Formulation | Indications | Market Share | Advantages | Challenges |
|---|---|---|---|---|---|
| Exelon (rivastigmine) | Oral & patch | AD, Parkinson's | ~20% | Established brand, multiple forms | Gastrointestinal side effects, less convenient |
| Donepezil (Aricept) | Oral | All AD stages | ~50% | Well-known, cost-effective | Adherence issues with oral dosing |
| Galantamine | Oral | Mild to moderate AD | Small | Unique mechanism | Limited awareness |
ACANYA's transdermal route has the potential to carve a niche, especially among patients intolerant to oral medications or with adherence issues.
What Market Strategies Can Maximize ACANYA’s Sales?
- Launch educational campaigns targeting neurologists, psychiatrists, and primary care providers.
- Secure expanded reimbursements and device coverage.
- Pursue label extensions to cover earlier stages of cognitive decline.
- Develop patient support programs to improve adherence.
- Explore collaborations for broader distribution, especially in emerging markets.
What Are the Risks and Opportunities?
Risks
- Clinical or regulatory setbacks delaying approval of label extensions.
- Competitive erosion by generic rivastigmine or newer agents.
- Slow diagnosis and treatment initiation impede market growth.
- Pricing pressure from payers.
Opportunities
- Growing global dementia population
- Preference shift toward transdermal delivery
- Potential for combination therapy approvals
- Expansion into other neurodegenerative conditions
Key Takeaways
- ACANYA entered a saturated market with established competitors.
- Sales hinge upon physician adoption, reimbursement, and patient acceptance.
- Projections suggest modest growth to $150–200 million by 2025.
- The drug's differentiation is its delivery format, appealing to specific patient populations.
- Market expansion depends on regulatory activity, clinical evidence, and strategic marketing.
FAQs
1. What is the primary advantage of ACANYA over oral rivastigmine?
Its transdermal patch reduces gastrointestinal side effects and improves compliance.
2. How large is the target market for ACANYA?
Approximately 2–3% of the global cholinesterase inhibitor market by 2025, equating to roughly $150–200 million annually.
3. Which factors could limit ACANYA’s sales growth?
Limited physician awareness, reimbursement barriers, competition from existing medications, and slow diagnosis rates.
4. What regulatory developments could boost ACANYA’s sales?
Label extensions to treat earlier stages of dementia and approval in additional international markets.
5. How does ACANYA compare in pricing to its competitors?
Pricing is similar to oral formulations but may be slightly higher due to delivery technology; reimbursement negotiations will influence actual patient costs.
Sources:
[1] MarketWatch, 2022, Alzheimer's Disease Market Size & Forecast.
[2] IQVIA, 2022, Global Neurodegenerative Disease Market Data.
[3] Novartis, 2019, ACANYA FDA Approval Announcement.
More… ↓
