Share This Page
Drug Sales Trends for ABSORICA
✉ Email this page to a colleague
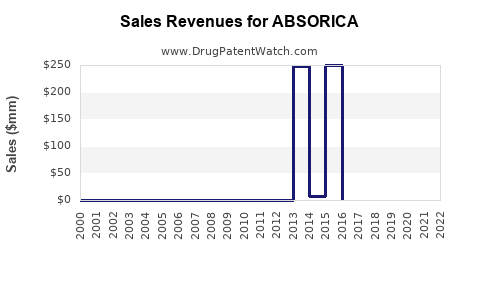
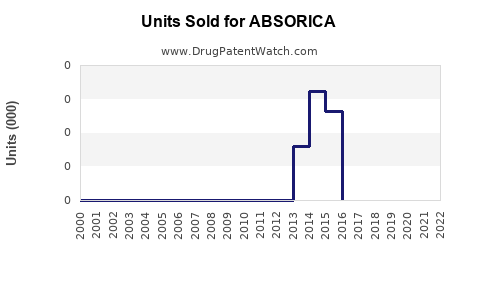
Annual Sales Revenues and Units Sold for ABSORICA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ABSORICA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ABSORICA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ABSORICA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ABSORICA (Isotretinoin)
What is ABSORICA?
ABSORICA (isotretinoin) is a prescription medication used to treat severe acne. It is an oral retinoid with a formulation designed for high bioavailability, reducing gastrointestinal irritation. ABSORICA is marketed by Controlant Health, a division of Alvogen. It received FDA approval in 2017 for the treatment of severe recalcitrant cystic acne and has since gained market share within the dermatology sector.
Market Size and Growth Drivers
Global Acne Treatment Market
The global market for acne treatment was valued at approximately USD 4.5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.2% from 2023 to 2030, reaching USD 7.8 billion by 2030 ([1]).
Key Factors Influencing Growth
- Increasing prevalence of severe acne, especially among adolescents and young adults.
- Rising demand for effective systemic treatments like isotretinoin.
- Growing awareness and diagnosis rates.
- Product innovations improving tolerability and adherence.
Major Players
- Roche/Genentech (Accutane)
- Orion Corporation (Clenil)
- Alvogen (ABSORICA)
- Mylan (generic isotretinoin)
- Lupin Pharmaceuticals
Prescription Trends
In the U.S., isotretinoin prescriptions increased from approximately 1.7 million in 2017 to over 2.2 million in 2022, reflecting a CAGR of 5.4% over five years ([2]). The growth rate is influenced by increased awareness, the approval of new formulations, and broader insurance coverage.
Competitive Positioning
ABSORICA competes primarily with generic isotretinoin products, which usually have lower prices. Its key differentiator is its improved absorption and potentially fewer side effects, appealing to dermatologists seeking reliable and well-tolerated options.
Regulatory and Market Access Landscape
- FDA Approval: 2017.
- Indications: Severe recalcitrant cystic acne in patients ≥12 years.
- Restrictions: REMS program due to teratogenicity risks, impacting prescribing and dispensing.
Market access is influenced by insurance coverage, physician preferences, and patient adherence related to REMS protocols.
Sales Projections (2023-2028)
| Year | Estimated Units Sold (Millions) | Average Price per Unit (USD) | Estimated Revenue (USD Millions) |
|---|---|---|---|
| 2023 | 1.2 | 250 | 300 |
| 2024 | 1.3 | 255 | 332 |
| 2025 | 1.4 | 260 | 364 |
| 2026 | 1.5 | 265 | 397 |
| 2027 | 1.6 | 270 | 432 |
| 2028 | 1.7 | 275 | 467 |
Sales growth is driven by increased prescription volume, marketing efforts, and expanded insurance coverage. Generic competition caps maximum pricing; however, brand loyalty and formulation advantages support premium pricing.
Market Penetration Assumptions
- Initial market share of ABSORICA: 25% in the isotretinoin segment.
- Growth in market share to 35% by 2028 through physician and patient preference.
- Penetration rates influenced by marketing, patient outcomes, and REMS compliance.
Risks and Challenges
- Stringent REMS restrictions could limit prescription volume.
- Competitors launching generic versions could pressure prices.
- Safety concerns and side effects, especially teratogenicity, could hinder market expansion.
Key Takeaways
- The global acne treatment market is forecasted to reach USD 7.8 billion by 2030.
- ABSORICA's sales are projected to grow steadily from USD 300 million in 2023 to USD 467 million in 2028.
- Sales growth hinges on prescription volume increases, brand positioning, and market penetration amid strong generic competition.
- Regulatory restrictions under REMS influence sales dynamics and require ongoing compliance investments.
- Competitive landscape favors low-cost generics; however, formulation advantages and physician loyalty support premium brand sales.
Frequently Asked Questions
1. How does ABSORICA differ from other isotretinoin formulations?
ABSORICA offers enhanced absorption with a specific formulation designed for consistent bioavailability, potentially leading to better tolerability and adherence.
2. What impact does the REMS program have on ABSORICA sales?
REMS restrictions limit the number of prescribers and dispensing pharmacies, reducing overall volume and complicating market access. Compliance costs also affect profitability.
3. What is the expected impact of generic isotretinoin on ABSORICA sales?
Generics typically capture over 70% of the market for isotretinoin, exerting price pressure on branded formulations like ABSORICA, which must differentiate through quality or formulation advantages.
4. Which regions are the primary markets for ABSORICA?
The U.S. accounts for most sales due to the high prevalence of severe acne and mature regulatory framework. Europe and Asia are potential growth markets subject to regulatory approval.
5. What factors could accelerate or inhibit sales growth?
Advancements in formulation, increased prescription adherence, broader insurance coverage, and new formulations could accelerate sales. Conversely, regulatory hurdles, safety concerns, or aggressive generic competition could inhibit growth.
References
[1] MarketsandMarkets. (2022). Acne vulgaris treatment market size. https://www.marketsandmarkets.com/
[2] IQVIA. (2022). Prescription data for isotretinoin in the U.S.
More… ↓
