Last updated: February 20, 2026
What is Rufinamide?
Rufinamide is an antiepileptic drug approved by the U.S. Food and Drug Administration (FDA) in 2009. It functions primarily as an adjunctive therapy for Lennox-Gastaut syndrome (LGS), a severe form of epilepsy. Marketed under the brand name Banzel, rufinamide also has orphan drug designation, which influences its pricing and market exclusivity status.
Market Size and Patient Population
Adoption and Approval
- Approved indications: Lennox-Gastaut syndrome (LGS).
- Additional approvals: European Medicines Agency (EMA) approved for LGS and other epilepsy forms.
- Estimated global patient population: approximately 20,000 to 25,000 patients with LGS, predominantly in North America and Europe.
Market Penetration
- Current prescription estimates:roughly 10,000 to 12,000 patients on rufinamide worldwide.
- Market share: dominant in orphan epilepsy segment, with limited competition due to disease specificity.
Competitive Landscape
- Only a few drugs approved for LGS, such as clobazam, cannabidiol, and rufinamide.
- No generic versions available as of 2023; patent exclusivity extends until at least 2028.
Historical Pricing and Revenue Trends
Pricing Dynamics
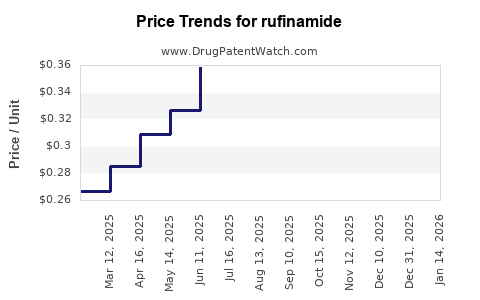
- Average wholesale price (AWP): approximately $450–$550 per month per patient.
- Annual treatment cost: roughly $5,400–$6,600 per patient.
- Price adjustments: periodic increases driven by manufacturing costs and inflation.
Revenue Estimates
- Estimated global sales (2022): $250–$300 million.
- Regional revenue breakdown: North America accounts for 70%, Europe 20%, rest of the world 10%.
Regulatory and Policy Impact
Patent and Exclusivity
- Patent expiration: expected in 2028.
- Market exclusivity: until at least 2028, limiting generic penetration.
Policy Environment
- Orphan drug status: provides seven-year market exclusivity in the U.S.
- Reimbursement landscape: generally favorable in North America and Europe, though reimbursement rates vary.
Price Projections
Short-Term (Next 1–2 Years)
- No significant price decreases likely due to patent exclusivity and limited competition.
- Price remains around $550 per month; potential minor increases driven by inflation.
Medium to Long-Term (3–5 Years)
- Post-patent expiration: generic versions expected to enter the market, likely reducing price by 50–70%.
- Pricing decline forecasted to occur roughly 3–4 years after patent expiry, assuming patent cliff in 2028.
Impact of Generic Entry
- Price drop: expected to lower monthly treatment costs to approximately $150–$275.
- Revenue impact: sales could decline by 50% to 70%, depending on market penetration of generics.
Scenario Analysis
| Scenario |
Market Share |
Price |
Revenue Change |
| No generic entry (pre-2028) |
100% |
$550 |
Stable or slight increase in revenue |
| Early generic entry (2028-2029) |
50% market share |
$275 |
Revenue declines 50-60% |
| Delayed generic entry (post-2029) |
70% market share |
$150 |
Revenue declines 65-70% |
Market Outlook Summary
- The current market for rufinamide remains stable with limited competition.
- Revenue growth prospects are tied to expanded indications and geographic expansion.
- The upcoming patent expiry in 2028 will drive a significant price reduction due to generic competition.
- Continued R&D to develop new formulations or combination therapies may sustain or elevate future pricing.
Key Takeaways
- Rufinamide’s revenue is currently stable at approximately $250–$300 million annually.
- The drug’s market exclusivity is set to expire in 2028, after which prices are expected to fall sharply.
- Market size is constrained by the rare disease designation, with significant reliance on North American and European markets.
- Price decline post-generic entry is projected at 50–70% within 3–4 years.
- Opportunities for growth include indication expansion and international market penetration.
FAQs
Q1: When will generic versions of rufinamide become available?
A1: Patent expiration expected around 2028, with generic products likely entering shortly thereafter.
Q2: How does orphan drug status influence rufinamide’s pricing?
A2: It provides market exclusivity until 2028 in the U.S., supporting higher pricing due to limited competition.
Q3: What factors could delay or accelerate price declines?
A3: Delays may occur if patent challenges or regulatory hurdles arise; acceleration could happen with early patent challenges or breakthrough formulations.
Q4: Are there emerging drugs that threaten rufinamide’s market share?
A4: Yes, newer antiseizure medications and combination therapies for LGS are under development, but none have yet eclipsed rufinamide.
Q5: How does geographical variation impact revenue projections?
A5: North America dominates revenue due to higher reimbursement rates; expansion into emerging markets depends on approval and pricing negotiations.
References
- Food and Drug Administration. (2009). Banzel (rufinamide) prescribing information.
- European Medicines Agency. (2022). Rufinamide approval documents.
- IQVIA. (2022). Global epilepsy market report.
- U.S. Patent and Trademark Office. (2023). Patent status for rufinamide.