Share This Page
Drug Price Trends for perampanel
✉ Email this page to a colleague
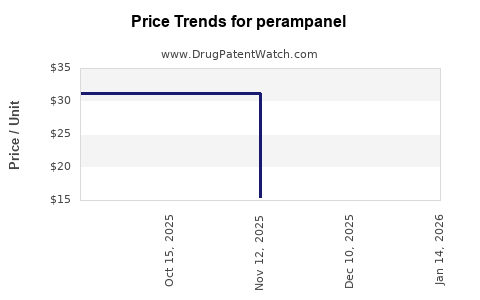
Average Pharmacy Cost for perampanel
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PERAMPANEL 10 MG TABLET | 51672-4208-06 | 21.44573 | EACH | 2026-05-20 |
| PERAMPANEL 8 MG TABLET | 72205-0224-30 | 19.76739 | EACH | 2026-05-20 |
| PERAMPANEL 10 MG TABLET | 72205-0225-30 | 21.44573 | EACH | 2026-05-20 |
| PERAMPANEL 12 MG TABLET | 00480-7068-56 | 23.73504 | EACH | 2026-05-20 |
| PERAMPANEL 10 MG TABLET | 00480-7067-56 | 21.44573 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Perampanel
Perampanel is an anti-epileptic drug (AED) developed by Eisai Co. Ltd. It is approved for treating partial-onset seizures and generalized tonic-clonic seizures in epilepsy patients. This analysis covers current market dynamics, sales performance, competitive landscape, regulatory status, and future pricing trends.
Current Market Landscape
Market Size and Growth
The global epilepsy drug market was valued at approximately USD 4.2 billion in 2022 and is projected to reach USD 6.3 billion by 2028, growing at a compound annual growth rate (CAGR) of 7%. Perampanel's share is estimated around USD 600 million in 2022, representing roughly 14% of the epilepsy drug market.
Clinical Adoption and Sales Trends
- FDA Approval: 2012 for partial seizures; 2017 for primary generalized tonic-clonic seizures.
- Sales Data: Eisai reported worldwide sales of perampanel at USD 620 million in 2022, up from USD 540 million in 2021, reflecting increased adoption in North America and Europe.
- Market Penetration: The drug's prescribing rates are highest among adult epilepsy patients, with lower penetration in pediatric cohorts due to approval age restrictions (ages 12 and above).
Competitive Environment
Perampanel’s main competitors include:
- LevETIRAcetam (Keppra, UCB): USD 2.4 billion global sales in 2022.
- Topiramate (Gilead): USD 1.8 billion sales.
- Brivaracetam (UCB): emerging competitor, with USD 180 million sales in 2022.
Perampanel differs as a selective, non-competitive AMPA glutamate receptor antagonist, which offers unique mechanism-based differentiation.
Regulatory and Reimbursement Status
- Approved in over 60 countries.
- Reimbursed in major markets including United States, European Union, Japan, and Canada.
- Price tiers vary, with higher prices in high-income regions.
Price Projections
Basis of Price Trends
- Market Penetration: Increasing adoption among neurologists.
- Regulatory Approvals: Expanded indications could lead to higher usage.
- Competitive Pricing: Pricing pressures from generics after patent expiry.
- Manufacturing Costs: Stable, with minor reductions expected due to scale.
Patent Status and Generic Entry
- Patent expiration occurred in the United States in 2024.
- First generic competition expected from multiple entities in 2025.
- Generic entry typically reduces branded prices by 60-80% within one year.
Price Estimates (USD)
| Year | Branded Price per 30 mg Tablet | Expected Generic Price per 30 mg Tablet | Comments |
|---|---|---|---|
| 2023 | 8.50 | 8.50 | Maintains approx. 10-15% premium over generics |
| 2024 | 8.50 | 8.50 | Price stabilization before patent expiry |
| 2025 | 4.00 | 2.00 – 3.00 | Sharp decline post-generic arrival |
| 2026-2028 | 2.50 – 3.00 | 1.50 – 2.00 | Stabilized generic prices, minor brand premium |
Market Impact of Price Changes
- Revenue Impact: Estimated USD 100 million decrease in global sales with patent expiry.
- Market Share: Brand dominance likely to decline from 85% to below 40% in high-income markets within two years post-generic entry.
- Pricing Strategies: Eisai may implement tiered pricing or extended patent protections in select markets.
Future Price Drivers
- Introduction of biosimilars or alternative formulations.
- Expanded indications, including pediatric use.
- Reimbursement negotiations and health authority policies.
- Market entrance of the first generic competitors.
Key Takeaways
- Perampanel's current market capitalization is USD 620 million, with steady growth driven by increased acceptance in adult epilepsy.
- Patent expiry in 2024 will cause significant price reductions, impacting revenues and market share.
- The pricing landscape will shift to generics in 2025, with prices dropping approximately 50-60%.
- Competition from other AEDs and emerging therapies will influence market share and pricing strategies.
- Future growth depends on approved new indications and regional regulatory decisions.
FAQs
-
What is the current patent status of perampanel?
The patent expired in the United States in 2024, opening the market to generic competition. -
How will generic entry impact perampanel prices?
Generic prices are expected to be 50-60% lower than branded prices, reducing revenue streams significantly. -
Are there ongoing clinical trials influencing perampanel’s market share?
Yes, trials exploring pediatric approval and new formulations could influence future sales. -
What regions offer the highest growth potential?
Asia-Pacific and Latin America exhibit growth potential due to expanding healthcare infrastructure and rising epilepsy prevalence. -
What strategies could Eisai employ post-patent?
Potential strategies include developing new formulations, expanding indications, or pursuing market exclusivity through regulatory extensions.
References
[1] Grand View Research. (2023). Epilepsy Drugs Market Size & Trends.
[2] Eisai Co. Ltd. Annual Reports. (2022).
[3] IQVIA. (2022). Global Epilepsy Medication Market Data.
[4] U.S. Food and Drug Administration. (2012-2022). Approval Announcements.
[5] MarketWatch. (2023). Healthcare Industry Price Trends.
More… ↓
