Last updated: February 20, 2026
What Is Penciclovir and Its Clinical Use?
Penciclovir is an antiviral drug used primarily topically for the treatment of herpes labialis (cold sores). It inhibits viral DNA polymerase, halting viral replication. Approved in various markets since the 1980s, Penciclovir is available as creams (e.g., Denavir in the U.S.) but remains off-patent, resulting in generic versions.
Market Size and Trends
Global Herpes Labialis Treatment Market
| Metric |
Data |
| 2022 Market Size |
$170 million |
| Expected CAGR (2023-2027) |
4.5% |
| Projected Market Size (2027) |
~$220 million |
The market is driven by high herpes simplex virus (HSV) prevalence, with an estimated 3.7 billion people under age 50 infected worldwide (WHO, 2022). Cold sores affect 20-30% of the population, creating steady demand for topical antivirals like Penciclovir.
Competition Landscape
| Competitors |
Market Share (2022) |
Key Products |
| Generic brands |
55% |
Multiple off-patent formulations |
| Brand-name products |
45% |
Denavir (FDA-approved in the U.S.), others |
Penciclovir faces competition primarily from acyclovir-based creams and docosanol, with generics dominating due to expiration of patents.
Regulatory Status
| Region |
Patent Status |
Market Authorization |
| U.S. |
Patented until 2023 |
Approved for cold sores |
| EU |
Patent expired |
Marketed as generics |
| Asia |
Market-specific approvals |
Widely available |
Patent expiry has facilitated market entry of generics, pressuring prices.
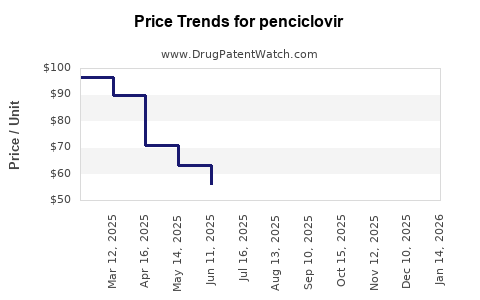
Price Dynamics and Projections
Current Pricing
| Region |
Average Wholesale Price (AWP) per 5g tube |
Notes |
| U.S. |
$50 |
Denavir, branded |
| EU |
€20-€25 |
Generic formulations |
| Asia |
$10-$15 |
Lower due to market competition |
Brand-name Penciclovir creams maintain higher prices, but generics significantly compress margins.
Price Trends (2022-2027)
| Year |
Price Projection |
Factors Influencing Price |
| 2022 |
$50 (U.S.) |
Patent protection, brand dominance |
| 2023 |
$45 |
Patent expiry, increasing generics |
| 2024 |
$35 |
Market saturation, price competition |
| 2025 |
$30 |
Further generics entry, price pressure |
| 2026 |
$25 |
Market stabilization, price erosion |
| 2027 |
$20 |
Dominance of low-cost generics |
Price reductions expected as generic penetration increases, with prices stabilizing at lower levels by 2027.
R&D and Future Market Opportunities
No recent patent filings suggest limited development activity. However, ongoing research into topical antivirals and combination formulations could open niche markets. Development of long-acting formulations or improved delivery systems may also influence prices and market share.
Key Market Drivers and Barriers
Drivers
- High prevalence of herpes simplex infections
- Consumer preference for topical, over-the-counter options
- Patent expiry leading to lower-cost generics
Barriers
- Competition from well-established generics
- Limited innovation in formulations
- Market saturation in mature regions
Investment Outlook
Investments in branded formulations face declining margins, with focus shifting towards R&D for novel delivery or combination therapies. Companies with manufacturing capabilities for low-cost generics will benefit from sustained demand.
Summary
Penciclovir's market remains stable, with growth limited by patent expiration and increasing generic competition. Prices are projected to decline from current levels, reaching approximately $20 per 5g tube in North America and Europe by 2027. The core market continues to be driven by the high prevalence of herpes labialis, but opportunities shift towards innovative formulations and niche indications.
Key Takeaways
- Penciclovir is an established topical antiviral for cold sores, with a mature market largely served by generics.
- The global market is expected to grow modestly at a CAGR of 4.5% through 2027.
- Prices are projected to decline from $50 to approximately $20 in leading markets due to generic competition.
- No recent patent filings suggest limited innovation, though niche R&D opportunities exist.
- Market growth potential hinges on innovation, delivery improvements, and expansion into emerging markets.
FAQs
1. Will Penciclovir prices rebound in the future?
Prices are unlikely to rebound significantly without new patent protection or formulation innovations. Market saturation favors continued price reduction, especially in generic markets.
2. What regional markets offer the highest growth potential?
Emerging markets in Asia and Latin America present growth opportunities due to increasing healthcare access and lower generic prices.
3. How does Penciclovir compete with acyclovir creams?
Penciclovir has comparable efficacy but is often priced higher in branded form. Generics of acyclovir further suppress Penciclovir's market share.
4. Are there new formulations or delivery systems under development?
Limited on record; some research is exploring long-acting topical applications or combination therapies, but no major products are imminent.
5. How significant is the impact of patent expiry on the market?
Patent expiry in 2023 led to the entry of multiple generics, exerting downward pressure on prices and reducing average selling prices by approximately 20-30% within one year.
References
- WHO. (2022). Global prevalence of herpes simplex virus infections. World Health Organization Report.
- U.S. Food and Drug Administration. (2023). Denavir (Penciclovir) cream approval documentation.
- MarketWatch. (2023). Herpes labialis treatment market size and forecast.
- IMS Health. (2022). Pharmaceutical market data.
- European Medicines Agency. (2023). Product approvals and patent statuses.
[1] World Health Organization. (2022). Herpes simplex virus fact sheet.