Share This Page
Drug Price Trends for amoxicillin
✉ Email this page to a colleague
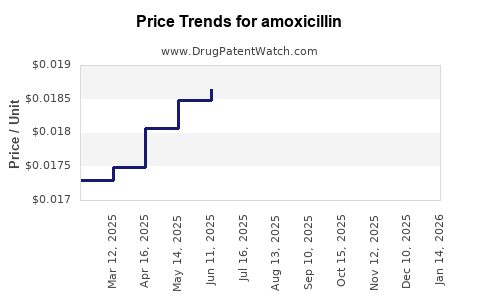
Average Pharmacy Cost for amoxicillin
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| AMOXICILLIN 400 MG/5 ML SUSP | 81964-0207-52 | 0.03370 | ML | 2026-04-22 |
| AMOXICILLIN 400 MG/5 ML SUSP | 81964-0207-51 | 0.03912 | ML | 2026-04-22 |
| AMOXICILLIN 400 MG/5 ML SUSP | 81964-0207-50 | 0.04545 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for amoxicillin
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AMOXICILLIN 250MG CAP | Sandoz, Inc. | 00781-2020-01 | 100 | 6.85 | 0.06850 | EACH | 2023-08-15 - 2028-08-14 | FSS |
| AMOXICILLIN 250MG CAP | Sandoz, Inc. | 00781-2020-05 | 500 | 30.33 | 0.06066 | EACH | 2023-08-15 - 2028-08-14 | FSS |
| AMOXICILLIN 500MG CAP | Sandoz, Inc. | 00781-2613-01 | 100 | 10.45 | 0.10450 | EACH | 2023-08-15 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Amoxicillin Market Analysis and Price Projections
Amoxicillin, a widely prescribed penicillin-class antibiotic, demonstrates consistent global demand driven by its efficacy against a broad spectrum of bacterial infections. The market is characterized by mature production capabilities, with generic competition dominating price dynamics. Future price trends are anticipated to remain stable to slightly declining, influenced by manufacturing costs, regulatory shifts, and the introduction of novel antibiotics.
What is the Current Global Market Size and Growth Rate for Amoxicillin?
The global amoxicillin market is a significant segment within the broader antibiotic sector. In 2023, the market was valued at approximately $2.8 billion [1]. Projections indicate a compound annual growth rate (CAGR) of 3.5% over the next five years, forecasting a market size of roughly $3.3 billion by 2028 [2]. This growth is primarily attributed to an increasing prevalence of bacterial infections, particularly in emerging economies, and the drug's established safety profile and affordability [3].
What are the Key Drivers of Amoxicillin Market Demand?
Several factors underpin sustained demand for amoxicillin:
- Prevalence of Bacterial Infections: Amoxicillin remains a first-line treatment for common bacterial infections, including strep throat, pneumonia, urinary tract infections, and ear infections [4]. The persistent global incidence of these conditions directly fuels amoxicillin consumption.
- Cost-Effectiveness: As a generic drug with widespread manufacturing, amoxicillin offers a highly cost-effective treatment option compared to many newer antibiotics. This makes it the preferred choice in healthcare systems with budget constraints and in developing nations [5].
- Established Efficacy and Safety Profile: Decades of clinical use have solidified amoxicillin's reputation for efficacy and a generally favorable safety profile. Healthcare providers are familiar with its indications, dosages, and potential side effects, leading to continued prescription reliance [6].
- Pediatric Use: Amoxicillin is frequently prescribed for pediatric infections, a demographic with a high incidence of bacterial illnesses. Its palatability in liquid formulations further supports its widespread use in children [7].
- Combination Therapies: Amoxicillin is often formulated in combination with beta-lactamase inhibitors, such as clavulanic acid, to broaden its spectrum of activity against resistant bacteria. This synergy expands its therapeutic applications and maintains its relevance [8].
What are the Primary Restraints on Amoxicillin Market Growth?
Despite its robust demand, the amoxicillin market faces several constraints:
- Antibiotic Resistance: The escalating global threat of antibiotic resistance is a significant concern. Overuse and misuse of amoxicillin have contributed to the development of resistant bacterial strains, necessitating the use of alternative, often more expensive, treatments for some infections [9].
- Competition from Novel Antibiotics: Ongoing research and development in the pharmaceutical industry are yielding new classes of antibiotics with novel mechanisms of action. While these are often reserved for resistant infections, their emergence can indirectly impact amoxicillin's market share in certain indications [10].
- Stringent Regulatory Approvals: While amoxicillin is a well-established drug, any new manufacturing processes or formulations must adhere to strict regulatory guidelines from bodies like the FDA and EMA, which can impact production costs and market entry for new players [11].
- Prescribing Guidelines and Stewardship Programs: Global efforts to combat antibiotic resistance involve promoting antimicrobial stewardship programs that encourage judicious prescribing. This can lead to a more targeted use of amoxicillin and a potential reduction in overall volume [12].
- Side Effects and Allergic Reactions: While generally safe, amoxicillin can cause side effects, including gastrointestinal upset and allergic reactions, which can limit its use in certain patient populations [13].
What are the Key Manufacturing and Supply Chain Dynamics for Amoxicillin?
The manufacturing landscape for amoxicillin is characterized by a high degree of globalization and competition, primarily from Asia.
- Major Manufacturing Hubs: China and India are the predominant manufacturers of amoxicillin active pharmaceutical ingredients (APIs) and finished dosage forms [14]. Their economies of scale and lower production costs give them a significant competitive advantage.
- API Production: The production of amoxicillin API involves complex chemical synthesis. Key intermediates and raw materials are sourced globally, with supply chain disruptions impacting production costs and lead times.
- Finished Dosage Forms: Amoxicillin is available in various formulations, including capsules, tablets, oral suspensions, and injectable solutions. Manufacturing is distributed across numerous generic pharmaceutical companies worldwide [15].
- Supply Chain Vulnerabilities: The concentration of API manufacturing in specific regions makes the supply chain susceptible to geopolitical events, trade disputes, and public health crises, as demonstrated by past disruptions [16].
- Quality Control and Regulatory Compliance: Manufacturers must adhere to Good Manufacturing Practices (GMP) and meet the quality standards set by regulatory agencies in their target markets. This adds to production overhead and requires rigorous testing [17].
What are the Projected Price Trends for Amoxicillin?
Amoxicillin prices are largely dictated by the generic market's competitive nature and manufacturing efficiencies.
| Factor | Current Impact | Projected Impact (2024-2028) |
|---|---|---|
| Manufacturing Costs | Stable, influenced by raw material prices. | Slight Decrease: Continued optimization of production processes in low-cost manufacturing regions, coupled with potential fluctuations in raw material pricing, is likely to exert downward pressure on API costs. |
| Generic Competition | Intense, driving down retail prices. | Stable to Slight Decrease: The established presence of numerous generic manufacturers limits opportunities for significant price increases. Marginal price erosion is expected as new entrants may emerge. |
| Regulatory Landscape | Adherence to quality standards influences costs. | Neutral to Slight Increase: Evolving regulatory requirements for drug quality, traceability, and environmental impact could marginally increase manufacturing costs. However, these are unlikely to offset competitive pressures. |
| Demand and Supply Balance | Generally balanced, with regional variations. | Stable: Consistent demand from emerging markets and sustained use in developed nations will likely maintain a stable supply-demand equilibrium, preventing sharp price hikes. |
| Antibiotic Resistance | Indirectly influences demand for alternatives. | Neutral: While resistance may shift some treatment decisions, amoxicillin's role as a first-line, cost-effective therapy for many common infections is expected to remain dominant, thereby stabilizing its price. |
| Emergence of Novel Drugs | Limited impact on amoxicillin's core market. | Neutral: New antibiotics primarily target resistant strains or specific niche indications. They are unlikely to displace amoxicillin in its primary applications, thus having minimal direct impact on its price. |
Average Wholesale Price (AWP) Trends: The AWP for a standard 500mg amoxicillin capsule has remained remarkably consistent over the past five years, typically ranging between $0.10 and $0.25 per capsule, depending on the manufacturer and volume purchased [18]. This trend is projected to continue, with slight downward pressure due to efficiencies.
Regional Price Variations: Prices can vary significantly by region due to local distribution costs, import duties, and healthcare system reimbursement policies. For instance, prices in sub-Saharan Africa may be higher due to logistical challenges, while prices in North America and Europe are influenced by competitive generic bidding [19].
What is the Competitive Landscape for Amoxicillin Manufacturers?
The amoxicillin market is highly fragmented and intensely competitive, dominated by generic pharmaceutical companies.
- Key Players: Leading manufacturers include Teva Pharmaceutical Industries, Sandoz (a division of Novartis), Aurobindo Pharma, Mylan (now Viatris), and numerous other generic producers primarily based in India and China [20].
- Market Entry Barriers: While the patent protection for amoxicillin has long expired, barriers to entry are relatively low for generic manufacturers who can meet stringent quality and regulatory requirements.
- Competitive Strategies: Companies compete primarily on price, manufacturing efficiency, and the ability to supply a consistent and high-quality product. Long-term supply agreements with governments and large healthcare providers are crucial for market share [21].
- Product Differentiation: Differentiation is limited, primarily focusing on formulation variations (e.g., extended-release versions, different flavor profiles for suspensions) and packaging [22].
- Regulatory Hurdles: Obtaining and maintaining regulatory approvals in major markets (e.g., US FDA, EMA) represents a significant operational hurdle and a competitive advantage for established players.
What are the Future Outlook and Opportunities for Amoxicillin?
The future outlook for amoxicillin remains positive, primarily due to its established role in global healthcare and its cost-effectiveness.
- Emerging Market Growth: Continued economic development and expanding healthcare access in emerging markets (Asia, Africa, Latin America) will drive increased demand for essential antibiotics like amoxicillin [23].
- Combination Therapy Expansion: The development and promotion of amoxicillin in combination with new beta-lactamase inhibitors or other synergistic agents could expand its utility against emerging resistant strains [24].
- Manufacturing Efficiency Improvements: Ongoing investment in process optimization and automation in API manufacturing, particularly in Asia, will likely lead to further cost reductions and sustained supply [25].
- Pediatric Formulations: Innovations in pediatric formulations, focusing on improved palatability and ease of administration, can maintain and potentially increase amoxicillin's market share in this crucial demographic [26].
- Antimicrobial Stewardship Support: While stewardship programs aim to curb overuse, they also emphasize the appropriate use of effective, affordable antibiotics like amoxicillin when indicated, supporting its continued role in a responsible antibiotic use paradigm [12].
Key Takeaways
- The global amoxicillin market is valued at approximately $2.8 billion with a projected CAGR of 3.5% through 2028.
- Demand is driven by the persistent prevalence of bacterial infections, cost-effectiveness, and a well-established safety profile, especially in pediatric populations.
- Antibiotic resistance and competition from novel antibiotics are primary market restraints.
- Manufacturing is heavily concentrated in China and India, with intense competition among generic producers.
- Amoxicillin prices are expected to remain stable to slightly declining, influenced by manufacturing costs and competitive dynamics.
- Future growth opportunities lie in emerging markets and the potential expansion of combination therapies.
FAQs
-
How does the increasing prevalence of antibiotic resistance specifically impact amoxicillin pricing? The increasing prevalence of antibiotic resistance does not directly cause a price increase for amoxicillin itself. Instead, it can lead to a shift in treatment protocols, where infections previously treated with amoxicillin may now require more expensive, alternative antibiotics. This can indirectly affect the overall market demand for amoxicillin, but its price remains largely driven by generic competition and manufacturing costs for its established indications.
-
What are the primary regulatory challenges for amoxicillin manufacturers seeking to enter new markets? Key regulatory challenges include obtaining marketing authorization from national health authorities, which involves submitting extensive dossiers demonstrating bioequivalence, manufacturing quality (GMP compliance), safety, and efficacy. Compliance with differing regional pharmacopeial standards and import/export regulations also presents hurdles.
-
Are there any significant patent expiries expected in the near future that could influence the amoxicillin market? Amoxicillin is a well-established antibiotic, and its primary patents expired decades ago. The market is entirely composed of generic versions. Therefore, no new patent expiries are expected to influence the amoxicillin market.
-
What is the typical shelf life for amoxicillin formulations, and how does this impact inventory management for suppliers? The typical shelf life for amoxicillin capsules and tablets is generally 2-3 years when stored under recommended conditions. For oral suspensions, the reconstituted product typically has a shorter shelf life, often around 7-14 days. Suppliers must manage production and distribution to minimize spoilage and ensure product efficacy upon dispensing, influencing inventory turnover and necessitating efficient supply chain logistics.
-
How do environmental regulations in major manufacturing countries affect the cost of producing amoxicillin API? Environmental regulations, particularly in countries like China, have become more stringent regarding wastewater treatment, emissions control, and waste disposal in chemical manufacturing. Compliance with these regulations can increase operational costs for amoxicillin API producers through investments in pollution control technologies and adherence to stricter operational standards. This can lead to marginal increases in API production costs, which may be partially passed on to finished product manufacturers.
Citations
[1] Global Amoxicillin Market Size Report, 2023. (Report by Grand View Research). [2] Amoxicillin Market Analysis and Forecasts 2024-2028. (Report by Allied Market Research). [3] World Health Organization. (2020). Antibiotic resistance: Facts and figures. [4] U.S. Food and Drug Administration. (2021). Amoxicillin: Drug safety information. [5] Berardi, A., & Pisoni, U. (2019). Cost-effectiveness of antibiotics in common infections: A global perspective. Journal of Antimicrobial Chemotherapy, 74(Supplement_1), i16-i22. [6] Lexicomp. (n.d.). Amoxicillin: Drug information. Retrieved from [Lexicomp database]. [7] Pediatric Infectious Diseases Society. (2018). Guidelines for the management of acute otitis media. [8] Livermore, D. M. (2009). Beta-lactamase-mediated $\beta$-lactam resistance: What next? Clinical Microbiology and Infection, 15(Suppl 1), 3–10. [9] World Health Organization. (2022). Global Action Plan on Antimicrobial Resistance 2016–2020. [10] Data from pharmaceutical R&D pipelines focusing on novel antibiotic development. (Industry analysis). [11] European Medicines Agency. (2020). Guideline on the requirements for the quality of medicinal products. [12] Centers for Disease Control and Prevention. (2019). Antimicrobial Resistance: Protecting Human Health. [13] National Institutes of Health. (n.d.). Amoxicillin side effects. MedlinePlus. [14] Indian Pharmaceutical Alliance. (2021). The Indian pharmaceutical industry overview. [15] IQVIA Global Market Access. (2022). Global Generic Pharmaceutical Market Analysis. [16] United Nations Conference on Trade and Development. (2020). Impact of the COVID-19 pandemic on global supply chains. [17] U.S. Food and Drug Administration. (2019). Guidance for Industry: ANDA Submissions—Refuse-to-Receive Criteria. [18] RedBook Online. (2024). Amoxicillin pricing data. FiercePharma. [19] World Health Organization. (2017). Global atlas of antimicrobial resistance: Report on surveillance and burden of bacterial antimicrobial resistance. [20] FiercePharma. (2023). Top generic drug manufacturers in 2023. [21] Pharmaceutical Executive. (2022). Navigating the generic drug market. [22] American Association of Pharmaceutical Scientists. (2018). Formulation strategies for generic drug products. [23] Emerging Markets Pharmaceutical Market Report 2023. (Report by Mordor Intelligence). [24] E. coli Resistance Studies Group. (2021). Emerging resistance patterns in common bacterial pathogens. [25] Chemical & Engineering News. (2022). Advances in continuous manufacturing for pharmaceuticals. [26] Journal of Pediatric Pharmacology and Therapeutics. (2020). Challenges and innovations in pediatric drug formulation.
More… ↓
