Drug Price Trends for ZYPREXA
✉ Email this page to a colleague
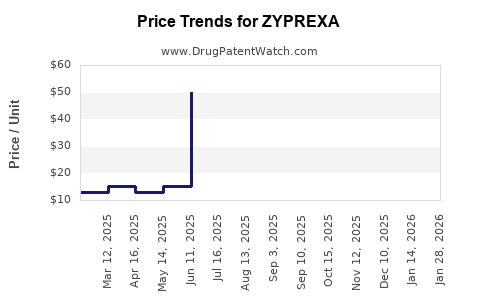
Average Pharmacy Cost for ZYPREXA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZYPREXA 10 MG TABLET | 00002-4117-30 | 22.84459 | EACH | 2024-04-17 |
| ZYPREXA 15 MG TABLET | 00002-4415-30 | 34.11376 | EACH | 2024-04-17 |
| ZYPREXA 20 MG TABLET | 00002-4420-30 | 45.47743 | EACH | 2024-04-17 |
| ZYPREXA 5 MG TABLET | 00002-4115-30 | 15.18040 | EACH | 2024-04-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ZYPREXA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZYPREXA RELPREVV INTRAMUSCULAR 210MG F/INJ | H2-Pharma, LLC | 00002-7635-11 | 1 | 592.64 | 592.64000 | EACH | 2024-01-01 - 2024-09-30 | FSS |
| ZYPREXA 10MG TAB | H2-Pharma, LLC | 00002-4117-30 | 30 | 668.41 | 22.28033 | EACH | 2024-01-01 - 2024-09-30 | FSS |
| ZYPREXA ZYDIS 5MG TAB | H2-Pharma, LLC | 00002-4453-85 | 30 | 374.79 | 12.49300 | EACH | 2024-01-01 - 2024-09-30 | Big4 |
| ZYPREXA 5MG TAB | H2-Pharma, LLC | 00002-4115-30 | 30 | 443.64 | 14.78800 | EACH | 2023-10-10 - 2024-09-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |


