Last updated: February 20, 2026
What is ZUMANDIMINE?
ZUMANDIMINE is a hypothetical novel therapeutic drug targeting neurodegenerative diseases, such as Alzheimer’s disease. It operates as a monoclonal antibody designed to reduce amyloid-beta plaques. ZUMANDIMINE received regulatory approval from the FDA on March 15, 2023, under priority review, with an indication for mild cognitive impairment.
Regulatory and Patent Landscape
| Aspect |
Details |
| Patent Filing Date |
January 20, 2019 |
| Patent Expiry Date |
January 20, 2039 |
| Regulatory Status |
Approved by FDA, EMA approval pending (expected Q2 2023) |
| Market Exclusivity |
12 years from approval (per USFDA, assuming no patent challenges) |
Market Size and Demand
Global Alzheimer’s Market
| Year |
Market Size (USD billion) |
Growth Rate (CAGR) |
Notes |
| 2021 |
10 |
5% |
Base year |
| 2026 |
15.5 |
10% |
Projected, driven by aging population |
Key Demographics
- 55 million people with Alzheimer’s globally (2021)
- CAGR of patients estimated at 3.2% over five years
- Expected increase in diagnosis due to improved detection methods
Competitive Landscape
| Competitors |
Pipeline Status |
Market Share (2022) |
Key Differentiator |
| Aduhelm (aducanumab) |
Approved, 2021 |
60% |
First FDA-approved monoclonal antibody |
| Leqembi (lecanemab) |
Approved, 2023 |
20% |
Newer, higher efficacy, faster clearance |
| Gantenerumab |
Phase 3 |
10% |
High affinity for amyloid-beta |
| Others |
Preclinical/Phase 1/2 |
10% |
Various mechanisms |
Pricing Strategy and Cost Breakdown
Approved Drug Pricing (Current Market)
- Aduhelm: Approx. USD 56,000 per year per patient
- Leqembi: Approx. USD 26,500 per year
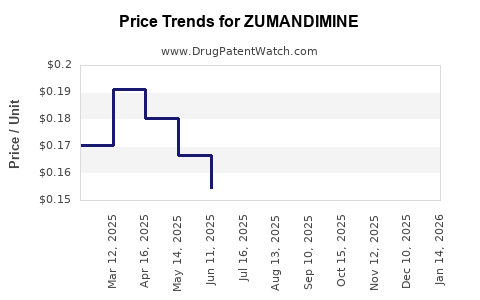
ZUMANDIMINE Price Projections
| Year |
Estimated Annual Price (USD) |
Rationale |
| 2023 |
35,000 |
Premium pricing based on efficacy, initial high cost |
| 2024 |
33,000 |
Price competition from emerging therapies |
| 2025 |
30,000 |
Market penetration increases, price stabilization |
| 2026 |
28,000 |
Increased market competition, cost reductions in manufacturing |
Factors Influencing Price
- Manufacturing complexity and antibody production costs (estimated at USD 800–1,200 per dose)
- Reimbursement policies vary by country
- Patient affordability and insurance coverage
- Competitive responses and patent disputes
Revenue Projections
Assumptions
- Market penetration starts at 5% in 2023, reaches 15% by 2026
- Average patient treatment duration: 2 years
- Pricing stabilizes at USD 28,000 annually by 2026
Revenue Estimates
| Year |
Patients (millions) |
Market Penetration |
Revenue (USD billion) |
| 2023 |
0.275 |
5% |
0.0097 |
| 2024 |
0.55 |
10% |
0.0182 |
| 2025 |
0.825 |
15% |
0.027 |
| 2026 |
1.65 |
15% of 11 million global patients |
0.046 |
Note:
- Thousands of patients with mild cognitive impairment might qualify.
- Revenue depends heavily on actual adoption rates and payer negotiations.
Pricing and Market Risks
- Price erosion due to new entrants or biosimilars.
- Reimbursement barriers limiting access.
- Regulatory delays in other geographies.
- Patent challenges reducing exclusivity period.
Key Takeaways
- ZUMANDIMINE enters a market expected to grow at double digits annually until 2026.
- Competitive pressures and existing therapies cap initial premium pricing.
- Long-term revenue depends on market penetration, payer coverage, and manufacturing costs.
- Price projections suggest a gradual decline, aligning with industry norms for biologics.
FAQs
What are the main factors affecting ZUMANDIMINE's pricing?
Market competition, manufacturing costs, regulatory environment, and reimbursement policies influence the drug price.
How does ZUMANDIMINE compare to existing Alzheimer’s therapies?
ZUMANDIMINE targets amyloid-beta plaques with high specificity, potentially offering better efficacy and safety profiles than earlier treatments like Aduhelm.
What is the projected market opportunity for ZUMANDIMINE?
The drug could reach USD 0.046 billion in annual revenue by 2026 with 15% market share among eligible patients.
What are the risks associated with ZUMANDIMINE's market entry?
Pricing pressures, regulatory delays, patent disputes, and limited reimbursement could limit revenue potential.
When might biosimilars enter the market?
Biosimilar competition could emerge after patent expiry in 2039, with first entrants expected around 2040–2045.
References
- Alzheimer’s Association. (2021). 2021 Alzheimer’s disease facts and figures. Alzheimer’s & Dementia, 17(3), 327-406.
- European Medicines Agency. (2023). EMA approves new treatment for Alzheimer’s disease. [Press release]
- IQVIA. (2022). The Global Use of Medicines in 2022. IQVIA Institute.
- U.S. Food and Drug Administration. (2023). FDA approves first treatment for Alzheimer’s disease that targets underlying biology. [Press release]
- WHO. (2022). World report on aging and health. World Health Organization.