Share This Page
Drug Price Trends for VERAPAMIL ER
✉ Email this page to a colleague
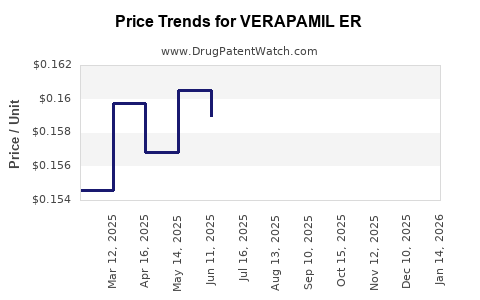
Average Pharmacy Cost for VERAPAMIL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VERAPAMIL ER 180 MG TABLET | 68462-0293-01 | 0.19318 | EACH | 2026-05-20 |
| VERAPAMIL ER 120 MG TABLET | 68462-0292-01 | 0.19266 | EACH | 2026-05-20 |
| VERAPAMIL ER 180 MG CAPSULE | 00378-6380-01 | 1.25516 | EACH | 2026-05-20 |
| VERAPAMIL ER 120 MG CAPSULE | 00378-6320-01 | 1.29595 | EACH | 2026-05-20 |
| VERAPAMIL ER 120 MG TABLET | 75834-0320-01 | 0.19266 | EACH | 2026-05-20 |
| VERAPAMIL ER 120 MG CAPSULE | 51079-0917-01 | 1.29595 | EACH | 2026-05-20 |
| VERAPAMIL ER 120 MG CAPSULE | 51079-0917-20 | 1.29595 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Verapamil ER Market Analysis and Price Projections
Verapamil ER, a calcium channel blocker used to treat hypertension, angina, and certain heart rhythm disorders, faces a dynamic market influenced by generic competition, evolving treatment guidelines, and patient adherence factors. Current market conditions indicate a stable demand driven by its established efficacy and cost-effectiveness. Price projections are tempered by ongoing generic erosion but supported by an aging global population and a persistent prevalence of cardiovascular diseases.
What is the current market size and growth trajectory for Verapamil ER?
The global market for Verapamil ER is a mature segment within the cardiovascular drug class. While precise, up-to-the-minute market size figures are proprietary and vary by data provider, industry estimates place the current global market value for Verapamil ER in the hundreds of millions of U.S. dollars annually. This segment has experienced low single-digit compound annual growth rates (CAGRs) over the past five years, largely driven by the increased prevalence of cardiovascular conditions and the drug's continued inclusion in treatment protocols for these ailments.
The primary driver of Verapamil ER's sustained market presence is its well-established safety and efficacy profile, particularly for managing chronic conditions like hypertension. Its affordability, especially in its generic forms, makes it a go-to treatment option, especially in developing economies and for cost-conscious healthcare systems.
However, the market growth is constrained by several factors:
- Generic Competition: Verapamil ER has long been off-patent, leading to intense competition among generic manufacturers. This has driven down prices significantly, limiting revenue growth for the overall market value.
- Competition from Newer Drug Classes: The development of newer antihypertensive and antiarrhythmic agents with potentially improved side-effect profiles or specific patient benefits has led to some substitution away from older drugs like Verapamil ER.
- Treating Specific Conditions: While Verapamil ER is versatile, its use is often narrowed down to specific patient profiles or conditions where it offers a distinct advantage over other available treatments.
Despite these constraints, the market is supported by:
- Aging Global Population: The increasing proportion of elderly individuals globally, a demographic highly susceptible to cardiovascular diseases, sustains a baseline demand for established treatments like Verapamil ER.
- Prevalence of Cardiovascular Diseases: Hypertension, angina, and supraventricular tachycardias remain significant public health challenges worldwide, ensuring a continuous need for effective and accessible therapeutic options.
- Established Treatment Guidelines: Verapamil ER is often recommended in established clinical guidelines for specific cardiovascular indications, reinforcing its place in therapeutic algorithms.
Market expansion, therefore, is more likely to be volume-driven rather than value-driven. Growth will be most pronounced in regions with expanding healthcare access and a rising burden of cardiovascular disease, where cost-effectiveness is a paramount consideration.
What is the competitive landscape for Verapamil ER?
The Verapamil ER competitive landscape is characterized by a high degree of genericization and numerous market participants. As a molecule with expired patent protection, the market is dominated by generic manufacturers producing bioequivalent versions of the drug.
Key aspects of the competitive landscape include:
- Generic Manufacturer Dominance: Major generic pharmaceutical companies, as well as smaller regional players, supply Verapamil ER. These companies compete primarily on price, manufacturing efficiency, and market access. Examples of companies that have historically produced or currently produce Verapamil ER include Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Aurobindo Pharma, and numerous others globally.
- Branded vs. Generic Pricing: While a branded version of Verapamil ER might have existed historically (e.g., Calan SR), the market now overwhelmingly consists of generic products. The price difference between branded and generic versions is substantial, with generics commanding the vast majority of the market volume.
- Therapeutic Equivalency: Regulatory bodies, such as the U.S. Food and Drug Administration (FDA), classify generic Verapamil ER products as therapeutically equivalent to their branded counterparts, allowing for substitution by pharmacists. This equivalence further intensifies price-based competition.
- Product Formulations: Verapamil ER is available in various extended-release formulations (e.g., tablets, capsules) and dosages (e.g., 120 mg, 180 mg, 240 mg). Manufacturers differentiate themselves by offering a range of these formulations and dosages to meet diverse prescribing needs and patient preferences.
- Supply Chain and Distribution: Competition also extends to supply chain efficiency and distribution networks. Companies with robust manufacturing capabilities and established distribution channels are better positioned to capture market share.
- Regulatory Compliance: Adherence to stringent regulatory standards for manufacturing (e.g., Current Good Manufacturing Practices - cGMP) is a baseline requirement. Companies that can maintain high compliance standards across multiple jurisdictions gain a competitive advantage.
- Emerging Market Penetration: Generic manufacturers are actively targeting emerging markets where Verapamil ER's affordability makes it an attractive option for large patient populations with limited healthcare budgets.
The absence of significant patent barriers means that new entrants can readily join the market, provided they meet regulatory requirements. This dynamic fosters continuous price pressure and necessitates operational efficiency from all stakeholders.
What are the key patents and intellectual property considerations for Verapamil ER?
Verapamil ER itself is a well-established drug with expired foundational patents. The original patents covering the composition of matter and methods of use for verapamil have long lapsed. For Verapamil ER, specific extended-release formulations might have had patents that also expired.
Key intellectual property considerations revolve around:
- Expired Composition of Matter Patents: The patent for verapamil as a chemical entity has expired, allowing for generic manufacturing.
- Expired Formulation Patents: Patents covering specific extended-release formulations and manufacturing processes for Verapamil ER have also largely expired or are nearing expiration. This is critical for generic manufacturers seeking to produce bioequivalent products.
- No Significant New IP: There are limited active patents that broadly protect Verapamil ER as a primary therapeutic agent. The focus of intellectual property in this space has shifted away from the core molecule and its basic extended-release mechanisms.
- Potential for Process Patents: While less common and often more challenging to enforce broadly, there could be niche patents related to novel manufacturing processes or specific polymorphic forms of verapamil that might offer minor protections. However, these are unlikely to create significant market barriers for established generic players.
- Exclusivity Periods: In some jurisdictions, generic manufacturers may benefit from short periods of market exclusivity after gaining regulatory approval, but this is not tied to new patent filings.
- Reference Product Exclusivity: For new generic entrants, the primary challenge is not intellectual property but rather navigating regulatory pathways and achieving bioequivalence to the reference listed drug.
In summary, for Verapamil ER, the landscape is characterized by the absence of significant patent protection for the drug itself or its primary extended-release formulations. This has led to the open generic market observed today. Companies interested in this space are not focused on novel IP for Verapamil ER but rather on efficient manufacturing and distribution of generic versions.
What are the projected price trends for Verapamil ER over the next five years?
Projected price trends for Verapamil ER over the next five years indicate continued price stability with gradual, incremental declines, primarily driven by generic competition and market dynamics. Significant price increases are highly unlikely.
Projected Price Trends (2024-2029):
- Current Average Selling Price (ASP): The ASP for generic Verapamil ER varies significantly by region, dosage, and distributor. However, the average wholesale price for a month's supply of standard dosages (e.g., 180 mg or 240 mg extended-release tablets) typically ranges from $10 to $50 USD, depending on market.
- Projected CAGR (Price): A negative CAGR of -1% to -3% is anticipated for the average selling price over the next five years.
- Key Drivers of Price Stability and Decline:
- Sustained Generic Competition: The ongoing presence of multiple generic manufacturers will continue to exert downward pressure on prices. Manufacturers will compete intensely on cost.
- Evolving Reimbursement Policies: Healthcare payers and formulary managers will continue to favor the most cost-effective treatment options, reinforcing the dominance of generic Verapamil ER and potentially leading to further price negotiations.
- Volume-Based Purchasing: Large healthcare systems and pharmacy benefit managers (PBMs) will leverage their purchasing power to negotiate lower prices from generic suppliers.
- Manufacturing Efficiencies: Generic manufacturers will continue to optimize production processes to reduce manufacturing costs, passing some of these savings to the market.
- Factors Preventing Steep Declines:
- Baseline Demand: The consistent demand for Verapamil ER due to its established therapeutic role will prevent prices from collapsing entirely.
- Manufacturing Costs: While efficiencies exist, the cost of active pharmaceutical ingredients (APIs), regulatory compliance, and quality control sets a floor for pricing.
- Supply Chain Stability: Major disruptions in the supply chain are unlikely for such a long-established drug, but localized shortages could temporarily impact regional pricing.
- Regional Variations: Prices will continue to vary significantly by country due to differences in regulatory environments, healthcare systems, generic market maturity, and local competition. Emerging markets may see slightly higher initial prices due to supply chain logistics but will also be susceptible to rapid price erosion as local competition emerges.
Overall, the Verapamil ER market is a mature, price-sensitive segment. Investors and R&D professionals should anticipate a market where profitability is driven by volume and operational efficiency rather than price appreciation.
What are the potential future market challenges and opportunities for Verapamil ER?
The future market for Verapamil ER is shaped by both persistent challenges and specific opportunities that will influence its continued relevance and commercial viability.
Market Challenges:
- Intensifying Generic Competition: The existing landscape of intense generic competition is expected to persist and potentially deepen. This could lead to further price erosion, squeezing profit margins for manufacturers.
- Therapeutic Displacement by Newer Agents: Ongoing research and development in cardiovascular medicine may yield newer drug classes or combination therapies that offer improved efficacy, safety profiles, or convenience for specific patient populations currently treated with Verapamil ER. For example, advancements in novel antihypertensive mechanisms or improved long-acting formulations of other drug classes could capture market share.
- Patient Adherence and Side Effects: While generally well-tolerated, Verapamil ER can cause side effects such as constipation, bradycardia, and dizziness. These can impact patient adherence, leading to treatment discontinuation and a shift to alternative therapies. Managing these side effects remains a challenge.
- Regulatory Scrutiny on Generics: Although well-established, generic drug manufacturers are subject to ongoing regulatory scrutiny regarding manufacturing quality and bioequivalence. Any adverse findings could impact market access and reputation.
- Market Saturation in Developed Economies: In highly developed markets with mature healthcare systems, the potential for significant new patient acquisition for Verapamil ER is limited. Growth will be primarily driven by patient turnover and shifts in prescribing patterns.
Market Opportunities:
- Increasing Prevalence of Chronic Cardiovascular Diseases: The global rise in hypertension, atrial fibrillation, and other cardiovascular conditions, particularly in aging populations and in emerging economies, ensures a sustained and growing demand for effective and affordable treatments like Verapamil ER.
- Cost-Effectiveness in Resource-Limited Settings: Verapamil ER's established efficacy and low cost make it an ideal therapeutic option for healthcare systems with budget constraints, particularly in low- and middle-income countries. Expansion into these markets represents a significant opportunity for generic manufacturers.
- Niche Indications and Off-Label Use: While its primary uses are well-defined, Verapamil ER may continue to find utility in specific niche cardiovascular indications or potentially in carefully monitored off-label applications where its pharmacological profile offers a unique benefit. Continued research or real-world evidence generation could solidify these roles.
- Optimized Generic Formulations and Manufacturing: Opportunities exist for generic manufacturers to differentiate through superior manufacturing processes that enhance product quality, stability, or reduce production costs, thereby strengthening their competitive position.
- Combination Therapies: While not a primary focus for Verapamil ER currently, the potential exists for its integration into fixed-dose combination therapies if clinical evidence supports synergistic benefits and improved patient compliance for specific cardiovascular conditions. This would require new development and regulatory pathways.
- Global Health Initiatives: As global health organizations focus on managing the burden of non-communicable diseases, established, affordable drugs like Verapamil ER are crucial components of public health programs.
The future of Verapamil ER lies in its ability to maintain its position as a cost-effective, reliable treatment option in the face of evolving therapeutic landscapes and intense generic competition, particularly by leveraging its affordability in growing global markets.
Key Takeaways
- The Verapamil ER global market is valued in the hundreds of millions of U.S. dollars, experiencing low single-digit CAGR driven by established efficacy and cost.
- Intense generic competition dominates the market, with numerous manufacturers competing primarily on price.
- Verapamil ER has no significant active patents protecting the drug or its extended-release formulations, enabling widespread generic availability.
- Projected price trends indicate continued stability with slight declines (-1% to -3% CAGR) over the next five years due to ongoing generic erosion.
- Future market challenges include escalating generic competition and displacement by newer therapies, while opportunities lie in the rising prevalence of cardiovascular diseases, cost-effectiveness in emerging markets, and niche indications.
Frequently Asked Questions
-
What is the primary mechanism of action for Verapamil ER? Verapamil ER is a non-dihydropyridine calcium channel blocker that inhibits the influx of calcium ions into cardiac and vascular smooth muscle cells. This leads to vasodilation, a decrease in heart rate, and reduced myocardial contractility, which collectively lower blood pressure and reduce the heart's workload.
-
Which specific cardiovascular conditions does Verapamil ER treat? Verapamil ER is indicated for the treatment of hypertension, chronic stable angina, and certain supraventricular arrhythmias, such as paroxysmal supraventricular tachycardia.
-
Are there any significant side effects associated with Verapamil ER use? Common side effects include constipation, dizziness, headache, nausea, and bradycardia. Less common but more serious side effects can include hypotension, heart failure, and liver enzyme elevations.
-
How does Verapamil ER compare to other classes of antihypertensive drugs? Verapamil ER belongs to the calcium channel blocker class. It differs from other classes like ACE inhibitors, ARBs, and beta-blockers in its specific mechanism of action and side effect profile. While effective for hypertension, it is often chosen for patients with specific co-existing conditions, such as arrhythmias, or when other drug classes are not tolerated or effective.
-
What are the typical dosage forms and strengths available for Verapamil ER? Verapamil ER is commonly available in extended-release tablet and capsule formulations. Typical strengths include 120 mg, 180 mg, and 240 mg, with dosages adjusted based on patient response and tolerability under medical supervision.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Website] [2] International Society of Pharmaceutical Engineers. (n.d.). Good Manufacturing Practices (GMP). Retrieved from [ISPE Website] [3] World Health Organization. (n.d.). Cardiovascular Diseases. Retrieved from [WHO Website] [4] Various Pharmaceutical Market Research Reports (proprietary data not publicly available). [5] Clinical Pharmacology Database (proprietary access required).
More… ↓
