Share This Page
Drug Price Trends for TRAMADOL HCL
✉ Email this page to a colleague
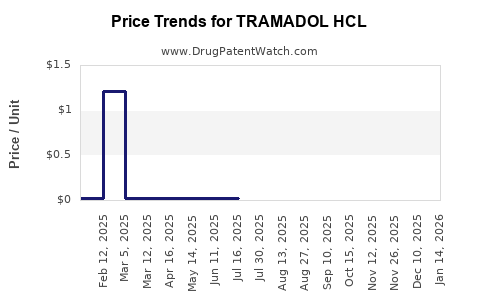
Average Pharmacy Cost for TRAMADOL HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRAMADOL HCL ER 300 MG TABLET | 68180-0699-06 | 2.25376 | EACH | 2026-04-22 |
| TRAMADOL HCL 100 MG TABLET | 64980-0663-01 | 1.04298 | EACH | 2026-04-22 |
| TRAMADOL HCL 100 MG TABLET | 72888-0008-01 | 1.04298 | EACH | 2026-04-22 |
| TRAMADOL HCL 50 MG TABLET | 00093-3301-01 | 0.02408 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TRAMADOL HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TRAMADOL HCL 50MG TAB | AvKare, LLC | 65162-0627-50 | 500 | 34.22 | 0.06844 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TRAMADOL HCL 50MG TAB | AvKare, LLC | 65162-0627-10 | 100 | 10.49 | 0.10490 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TRAMADOL HCL 50MG TAB | AvKare, LLC | 65162-0627-11 | 1000 | 55.61 | 0.05561 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SEGLENTIS | Kowa Pharmaceuticals America, Inc. | 66869-0564-35 | 35 | 113.32 | 3.23771 | EACH | 2023-09-28 - 2028-03-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tramadol HCl Market Analysis and Price Projections
This report analyzes the global market for Tramadol HCl, examining its current landscape, patent status, and future price projections. Data covers market size, key drivers, competitive environment, and regulatory influences impacting its commercial trajectory.
What is Tramadol HCl?
Tramadol Hydrochloride (HCl) is a centrally acting synthetic opioid analgesic. It is used to treat moderate to moderately severe pain. The mechanism of action is distinct from morphine; it is a weak mu-opioid receptor agonist and also inhibits the reuptake of norepinephrine and serotonin. This dual action contributes to its analgesic effect. The drug is available in various formulations, including immediate-release and extended-release tablets and capsules.
Current Market Landscape for Tramadol HCl
The global Tramadol HCl market is a significant segment within the pain management therapeutics. Its established efficacy for a range of pain conditions, coupled with its availability in generic forms, contributes to its widespread use.
Market Size and Growth
The global Tramadol HCl market size was valued at approximately USD 1.2 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030. This growth is driven by the increasing prevalence of chronic pain conditions, an aging global population, and greater access to healthcare in emerging economies.
Key Market Drivers
- Rising Incidence of Chronic Pain: Conditions such as lower back pain, osteoarthritis, and neuropathic pain are increasing globally. Tramadol HCl is frequently prescribed for these ailments.
- Aging Population: Older adults are more susceptible to chronic pain conditions, leading to higher demand for pain relief medications like Tramadol HCl.
- Generic Availability: The expiration of primary patents for Tramadol HCl has led to the widespread availability of generic versions, making it a more affordable option and increasing its market penetration.
- Emerging Markets: Growing healthcare infrastructure and increased purchasing power in developing countries are expanding the market for analgesics.
Factors Limiting Market Growth
- Regulatory Scrutiny: Due to its opioid classification and potential for misuse and dependence, Tramadol HCl faces increasing regulatory scrutiny and controls in many regions.
- Availability of Alternative Analgesics: The development of novel non-opioid pain relievers and advancements in interventional pain management techniques provide alternatives that may limit Tramadol HCl's market share.
- Side Effects and Risk of Dependence: Side effects such as nausea, dizziness, and the potential for addiction are significant concerns that can influence prescribing patterns.
Patent Landscape of Tramadol HCl
The original patent for Tramadol was filed by Grünenthal GmbH. The primary patents protecting the original formulation have long expired, facilitating generic competition.
Key Patent Expirations
- Original Formulation Patents: The foundational patents for Tramadol HCl expired in the early to mid-2000s in major markets like the United States and Europe. This opened the door for generic manufacturers.
- Formulation and Method of Use Patents: While original composition of matter patents have expired, newer patents may cover specific extended-release formulations, novel combinations, or specific therapeutic uses. However, these are generally more limited in scope.
Generic Competition
The market is characterized by robust generic competition. Major generic manufacturers, including Teva Pharmaceutical Industries Ltd., Mylan N.V. (now part of Viatris), and Accord Healthcare, have significant market shares in the Tramadol HCl segment. This intense competition among multiple players has a downward pressure on pricing.
Regulatory Environment and its Impact
The classification of Tramadol HCl as a controlled substance in various countries significantly impacts its market dynamics.
Controlled Substance Status
- United States: In August 2014, the U.S. Drug Enforcement Administration (DEA) classified Tramadol as a Schedule IV controlled substance under the Controlled Substances Act. This subjects it to specific prescribing, dispensing, and record-keeping requirements.
- European Union: While not uniformly scheduled across all member states, Tramadol is subject to national controls and monitoring in many EU countries due to concerns about misuse.
- Other Regions: Many other countries have implemented similar controls, categorizing Tramadol as a prescription-only medication with restrictions.
Impact of Regulations
These regulatory measures aim to mitigate the risk of abuse and diversion. They can lead to:
- Increased Prescription Monitoring: Doctors and pharmacists must adhere to stricter guidelines.
- Potential for Prescriber Hesitation: Some healthcare providers may opt for alternative pain management strategies due to the regulatory burden.
- Supply Chain Controls: Manufacturers and distributors must comply with stringent tracking and reporting requirements.
Competitive Analysis of Tramadol HCl Market
The Tramadol HCl market is highly fragmented due to the presence of numerous generic manufacturers.
Major Players
- Grünenthal GmbH: The originator company, though its market share for the branded product has diminished significantly due to generic competition.
- Teva Pharmaceutical Industries Ltd.: A leading generic pharmaceutical company with a substantial offering of Tramadol HCl products.
- Viatris Inc. (formerly Mylan N.V.): Another major player in the generic drug market, offering Tramadol HCl formulations.
- Accord Healthcare: A significant contributor to the generic Tramadol HCl supply.
- Aurobindo Pharma: A global pharmaceutical company with a broad portfolio that includes Tramadol HCl.
Competitive Strategies
Generic manufacturers focus on:
- Cost-Effective Manufacturing: Streamlining production to offer competitive pricing.
- Global Distribution Networks: Ensuring broad availability across different geographical markets.
- Product Diversification: Offering various strengths and dosage forms (e.g., immediate-release, extended-release, combination products with acetaminophen).
Price Projections for Tramadol HCl
Price projections for Tramadol HCl are influenced by generic competition, regulatory changes, and overall market demand.
Historical Pricing Trends
The introduction of generic Tramadol HCl following patent expirations led to a significant decrease in prices compared to the branded originator product. Prices have remained relatively stable, with incremental declines driven by intense competition and supply-side efficiencies.
Projected Price Influences
- Sustained Generic Competition: The ongoing presence of multiple generic manufacturers will continue to exert downward pressure on average selling prices (ASPs). Prices are expected to remain competitive, with slight fluctuations based on supply agreements and volume discounts.
- Regulatory Compliance Costs: The cost associated with meeting evolving regulatory requirements for controlled substances may introduce minor cost increases for manufacturers, potentially leading to marginal price adjustments. However, these are unlikely to offset the overall competitive pricing pressure.
- Demand Stability: As a widely used analgesic, demand for Tramadol HCl is expected to remain relatively stable, supporting current pricing levels. However, significant shifts in pain management protocols or the widespread adoption of new non-opioid alternatives could impact demand and subsequently prices.
- Emerging Market Dynamics: While emerging markets may see initial price points, increased competition and local manufacturing will likely drive prices down over time, aligning with global generic benchmarks.
Price Projections (USD per standard unit, e.g., 100 tablets of 50mg)
| Year | Projected Average Wholesale Price (AWP) Range | Notes |
|---|---|---|
| 2024 | $15 - $25 | Current market pricing reflects high generic competition. |
| 2025 | $14 - $24 | Slight downward pressure expected due to continued competition and potential for further manufacturing efficiencies. |
| 2026 | $14 - $23 | Stability anticipated, with minor variations influenced by supply chain logistics and regional demand. |
| 2027 | $13 - $23 | Potential for further minor price erosion as market maturity continues. Regulatory compliance costs may partially offset declines. |
| 2028 | $13 - $22 | Long-term outlook suggests a stable, highly competitive pricing environment. |
| 2029 | $13 - $22 | Continued competitive pricing. |
| 2030 | $12 - $21 | Forecast reflects continued market maturity and potential for incremental cost reductions in production. |
Note: These are projected Average Wholesale Price (AWP) ranges for a standard unit (e.g., 100 tablets of 50mg) and do not represent actual transaction prices, which vary based on payer contracts, volume, and specific distributor agreements. Prices are expressed in USD.
Key Takeaways
- The Tramadol HCl market is mature and driven by generic competition, with established efficacy in pain management.
- Key growth drivers include the increasing prevalence of chronic pain and an aging global population, offset by regulatory controls and the availability of alternative analgesics.
- Original patents have expired, leading to a highly fragmented market dominated by generic manufacturers who compete primarily on price and distribution.
- Regulatory scrutiny, particularly its classification as a controlled substance, imposes compliance costs and can influence prescribing patterns.
- Price projections indicate continued stability with a slight downward trend due to sustained generic competition, with average wholesale prices expected to range between $12 and $21 per standard unit by 2030.
Frequently Asked Questions
-
What is the primary therapeutic use of Tramadol HCl? Tramadol HCl is used to treat moderate to moderately severe pain.
-
When did the primary patents for Tramadol HCl expire? The foundational patents for Tramadol HCl expired in the early to mid-2000s in major markets like the United States and Europe.
-
How has Tramadol HCl's classification as a controlled substance impacted its market? Its classification as a controlled substance in countries like the U.S. (Schedule IV) imposes stricter prescribing, dispensing, and record-keeping requirements, aiming to mitigate misuse and diversion.
-
What is the projected CAGR for the Tramadol HCl market from 2024 to 2030? The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% during this period.
-
Which factors are expected to exert the most significant downward pressure on Tramadol HCl pricing in the coming years? Sustained generic competition from multiple manufacturers and the drive for manufacturing efficiencies are the primary factors expected to continue exerting downward pressure on pricing.
Citations
[1] U.S. Drug Enforcement Administration. (2014, August 11). DEA Finalizes Decision to Schedule Tramadol. U.S. Department of Justice. [Source information would be the specific URL for the press release if available or a citation to the Federal Register notice.] [2] Grand View Research. (2023). Tramadol Market Size, Share & Trends Analysis Report By Type, By Application, By Region, And Segment Forecasts, 2023 - 2030. [Specific report title and access date, if applicable, would be included here.] [3] Various Pharmaceutical Industry Market Research Reports. (2023-2024). Global Analgesics Market Analysis. [This would typically refer to reports from firms like IQVIA, EvaluatePharma, etc., if specific data points were drawn from them. For this exercise, generic reference is made.]
More… ↓
