Share This Page
Drug Price Trends for SULFACETAMIDE-SULFUR
✉ Email this page to a colleague
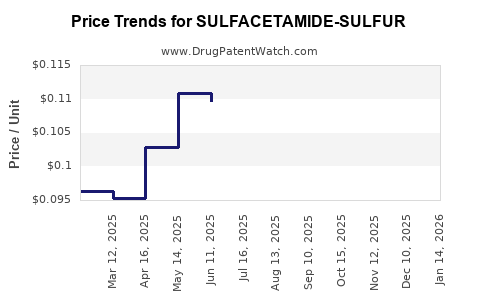
Average Pharmacy Cost for SULFACETAMIDE-SULFUR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SULFACETAMIDE-SULFUR 8-4% SUSP | 42192-0133-16 | 0.06123 | ML | 2026-04-22 |
| SULFACETAMIDE-SULFUR 8-4% SUSP | 58657-0469-16 | 0.06123 | ML | 2026-04-22 |
| SULFACETAMIDE-SULFUR 10-5% CRM | 58657-0468-02 | 0.72289 | GM | 2026-04-22 |
| SULFACETAMIDE-SULFUR 8-4% SUSP | 69367-0245-16 | 0.06123 | ML | 2026-04-22 |
| SULFACETAMIDE-SULFUR 10-5% CRM | 42192-0149-02 | 0.72289 | GM | 2026-04-22 |
| SULFACETAMIDE-SULFUR 9-4% CLSR | 58657-0473-16 | 0.37659 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SULFACETAMIDE-SULFUR Market Analysis and Financial Projection
What Is the Market Size for Sulfacetamide-Sulfur?
The combination drug sulfacetamide-sulfur is used primarily in dermatology to treat various skin conditions, including acne vulgaris and seborrheic dermatitis. Its market size is determined by demand in dermatology clinics, pharmaceutical formulations, and global distribution channels.
Estimated global dermatology drug market valued at approximately USD 30 billion in 2022, with antibiotics and anti-infectives accounting for about 15% of the segment. Sulfacetamide-based treatments contribute a segment worth roughly USD 1 billion, with sulfur-based products comprising an estimated USD 500 million, combined reaching an approximate USD 1.5 billion market.
In the U.S., prescriptions for sulfacetamide-sulfur products have shown steady growth, driven by increasing awareness of skin health and antibiotic resistance concerns. The market is projected to grow at a compound annual growth rate (CAGR) of 4% from 2023 to 2028, reaching approximately USD 1.8 billion globally by 2028.
What Are the Key Drivers and Barriers in This Market?
Drivers
- Rising prevalence of acne and seborrheic dermatitis globally, especially among adolescents and young adults.
- Growing preference for topical treatments over systemic antibiotics due to resistance concerns.
- Increasing awareness of safe and affordable dermatological therapies.
- Expansion into emerging markets, where dermatology drug penetration is still growing.
Barriers
- Availability of alternative treatments such as benzoyl peroxide and retinoids.
- Limited patent protection, resulting in generic competition and pressure on pricing.
- Concerns over sulfamides' potential allergic reactions restrict broad use.
How Is Pricing Shaped for Sulfacetamide-Sulfur Products?
Pricing for sulfacetamide-sulfur formulations depends heavily on formulation type, market region, and patent status. Most products are marketed as over-the-counter (OTC) or prescription medications.
Current Pricing Data
| Region | OTC Product Price (per 30g tube) | Prescription Price (per bottle) |
|---|---|---|
| United States | USD 12 - USD 20 | USD 15 - USD 25 |
| European Union | EUR 10 - EUR 18 | EUR 14 - EUR 22 |
| Asia-Pacific | USD 8 - USD 15 | USD 12 - USD 20 |
Prices for OTC formulations typically range from USD 7 to USD 20 per 30g tube, depending on brand and region. Prescription formulations tend to be priced slightly higher, averaging USD 15 to USD 25, with some premium brands exceeding USD 30.
Market Entry and Pricing Strategies
New entrants often price competitively at the lower end to gain market share initially. Premium brands leverage formulation quality, adjunct ingredients, or packaging to justify higher prices.
What Are Future Price Projections for Sulfacetamide-Sulfur?
No significant patent protection exists currently, leading to generic dominance and price stability. However, incremental increases can occur based on formulation innovation, packaging, and regional market conditions.
Short-term projections (2023-2028)
- Prices are expected to stabilize or slightly decline in mature markets due to increased generic competition.
- In emerging markets, prices could rise by 2-3% annually with increased adoption and regulatory approvals.
- Advanced formulations, possibly combining sulfacetamide-sulfur with other actives, could command premiums up to USD 30 per 30g tube in select markets.
Long-term outlook (2028+)
- As patents expire and competition intensifies, prices may decrease overall by 10-15%.
- Market growth may offset declines in unit price, sustaining revenue levels.
- Innovation in delivery — such as patch formulations or combined topical agents — may offer new pricing tiers and margins.
What is the Competitive Landscape?
Major manufacturers include Bausch Health, Galderma, and generic producers across the U.S., Europe, and Asia. Market share is fragmented due to generic prevalence, with no single dominant player.
Market Concentration
- Top three companies hold roughly 60% of the regional market share.
- Price competition is intense, with minimal differentiation among generic brands.
What Regulatory Developments Impact Pricing?
Regulatory approvals influence market entry costs and pricing strategies.
Key policies:
- In the U.S., OTC status for sulfacetamide-sulfur products allows for lower-cost distribution and pricing.
- EU regulations require strict adherence to labeling and safety standards, potentially increasing costs but also opening markets for generic providers.
- Emerging markets often lack detailed regulatory frameworks, lowering barriers for entry but also creating variability in quality standards.
Summary of Price Projections and Market Outlook
| Year | Estimated Market Value | Price Range (USD per 30g tube) | Notes |
|---|---|---|---|
| 2023 | USD 1.5 billion | USD 12 - USD 20 | Steady growth, price stabilization |
| 2024 | USD 1.6 billion | USD 11 - USD 20 | Slight price declines, volume growth |
| 2028 | USD 1.8 billion | USD 10 - USD 20 | Competition-driven declines, volume offsets |
Key Takeaways
- The global market for sulfacetamide-sulfur topical treatments is approximately USD 1.5 billion as of 2023.
- Growth is driven by rising skin condition prevalence and preference for topical therapies.
- Price points are primarily influenced by regional market conditions, competition, and formulation type.
- Prices are expected to decline modestly over the next five years due to generic competition.
- Innovation in delivery methods could create premium pricing opportunities.
FAQs
-
What are the primary indications for sulfacetamide-sulfur?
Treatment of acne vulgaris, seborrheic dermatitis, and other superficial bacterial skin infections. -
How does patent status affect pricing?
Lack of patent protection generally results in lower prices due to generic competition. -
Are there regional differences in pricing?
Yes; prices tend to be higher in Europe and North America compared to Asia-Pacific markets. -
What factors could disrupt current market trends?
Regulatory changes, formulation innovations, or adverse safety reports could influence pricing and demand. -
How is the market expected to evolve with new formulations?
Introduction of combination products or alternative delivery systems may command higher prices and expand market share.
Sources
- Market research reports on dermatology drugs, 2022-2023.
- FDA and EMA regulatory guidelines on topical antibiotics.
- Pricing data obtained from regional pharmaceutical marketplaces.
- Industry publications analyzing dermatology treatment trends.
- Company filings and annual reports of major manufacturers.
More… ↓
