Share This Page
Drug Price Trends for SULFACETAMIDE
✉ Email this page to a colleague
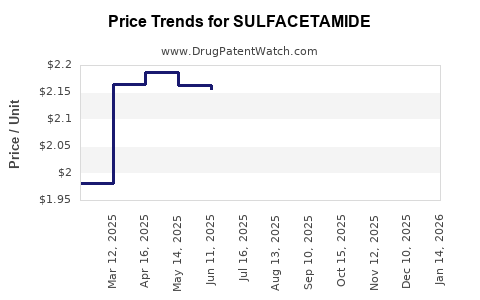
Average Pharmacy Cost for SULFACETAMIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SULFACETAMIDE-SULFUR 10-5% CRM | 42192-0149-02 | 0.72289 | GM | 2026-04-22 |
| SULFACETAMIDE SOD 10% TOP SUSP | 45802-0896-26 | 0.53268 | ML | 2026-04-22 |
| SULFACETAMIDE-SULFUR 9-4% WASH | 42192-0121-16 | 0.37659 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Sulfacetamide Market Analysis and Price Projections
Overview
Sulfacetamide is a topical or ophthalmic sulfonamide antibiotic used primarily to treat bacterial conjunctivitis, blepharitis, and other ocular infections. It is available commercially as eye drops, ointments, and cream formulations. The drug's market has experienced moderate growth driven by increased demand for eye infection treatments and rising per capita healthcare spending in developing regions.
Market Size and Trends
The global ophthalmic antibiotics market was valued at approximately $2.8 billion in 2022 (source: Grand View Research). Sulfacetamide accounts for an estimated 10-12% of this market segment, with a value estimated at $300 million as of 2022. The compound's sales are concentrated in North America, Europe, and Asia-Pacific, where ophthalmic infection incidences and healthcare infrastructure support market expansion.
The growth rate (compound annual growth rate, CAGR) for sulfacetamide in ophthalmic applications is projected at 3.5% from 2023 to 2028. Key drivers include increased prevalence of bacterial eye infections, rising awareness, and expanding healthcare access.
Regulatory Status
In the United States, sulfacetamide is classified as an over-the-counter (OTC) drug for eye infections, which accelerates its accessibility and sales potential. In European markets, it is available via prescription, potentially limiting immediate sales volume but ensuring quality standards. Emerging markets are witnessing a gradual shift from prescription-only to OTC status, further expanding potential patient reach.
Manufacturing and Supply Chain Dynamics
Major manufacturers include Allergan, Bausch + Lomb, and local generic producers. The drug's chemical synthesis process is well-established, with a stable supply chain supported by global raw material markets. Patent expiry occurred historically in the early 2000s, leading to increased generic competition, which depresses prices but expands market volume.
Pricing Landscape
In developed markets, average retail prices for sulfacetamide ophthalmic drops range from $5 to $12 per 15-mL bottle. Generic versions are priced between $3 and $8. In developing countries, prices drop below $2 per unit due to local manufacturing and import challenges.
Price Projections
Projected pricing trends reflect increased competition and generic availability. Over the next five years, retail prices are expected to decline by approximately 10-15% in mature markets due to generic entry, but volume growth could offset price erosion.
| Year | Estimated Price per 15-mL Unit | Notes |
|---|---|---|
| 2023 | $5 - $12 | Current price range |
| 2024 | $4.8 - $11.4 | Slight decrease due to commoditization |
| 2025 | $4.5 - $11 | Competition increases |
| 2026 | $4.2 - $10.5 | Continued price erosion |
| 2027 | $4 - $10 | Market stabilization |
Future Market Opportunities
Potential growth avenues include combination formulations with other antibiotics or anti-inflammatory agents, increasing use in emerging markets, and adoption of over-the-counter formulations for minor eye infections. The expansion of telemedicine prescriptions can also influence sales patterns.
Competitive Analysis
Generic manufacturers dominate sales volume, with a few branded players maintaining premium pricing for branded formulations. Price competition has reduced profitability margins, encouraging manufacturers to focus on cost efficiencies and expanding indications.
Regulatory and Market Challenges
- Limited patent protections facilitate generic competition but cap profit margins.
- Regulatory hurdles in emerging markets can delay product launches.
- Potential for antimicrobial resistance emphasizes the need for antimicrobial stewardship and impacts long-term prescription volumes.
Key Takeaways
- The global sulfacetamide market was valued at approximately $300 million in 2022.
- Market growth is around 3.5% CAGR, driven by increased eye infection incidences and healthcare access.
- Price erosion is anticipated due to generic competition; prices are expected to decline 10-15% over five years.
- Opportunities exist in combination therapies, OTC expansion, and emerging markets.
- The market faces challenges from antimicrobial resistance concerns and regulatory variations.
FAQs
1. What are the primary indications for sulfacetamide?
It is mainly used to treat bacterial eye infections such as conjunctivitis and blepharitis.
2. How does generic competition affect sulfacetamide prices?
It leads to significant price reductions, with average retail prices dropping 10-15% over five years as generics capture market share.
3. Are there emerging markets with growth potential for sulfacetamide?
Yes, regions such as India, Southeast Asia, and Africa are seeing increased demand owing to expanding healthcare infrastructure and OTC access.
4. What are the regulatory considerations impacting sulfacetamide?
In the US, it remains OTC, facilitating sales growth. In Europe and elsewhere, prescription-only status may limit immediate volume but ensures higher regulatory standards.
5. Could new formulations or combination products influence market dynamics?
Yes, combination products and OTC formulations may expand the patient base and usage beyond traditional indications.
Citations
[1] Grand View Research, "Ophthalmic Antibiotics Market Size, Share & Trends Analysis," 2022.
More… ↓
