Share This Page
Drug Price Trends for SM IBUPROFEN
✉ Email this page to a colleague
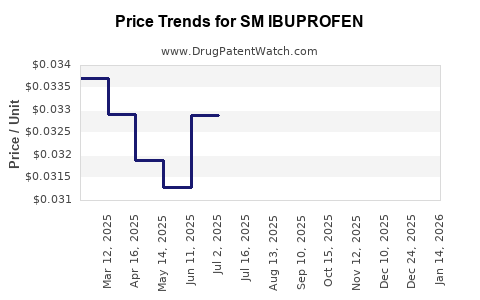
Average Pharmacy Cost for SM IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM IBUPROFEN 200 MG CAPLET | 49348-0196-10 | 0.03225 | EACH | 2026-04-22 |
| SM IBUPROFEN 200 MG TABLET | 49348-0706-14 | 0.03225 | EACH | 2026-04-22 |
| SM IBUPROFEN IB 100 MG CHEW TB | 70677-0072-01 | 0.15537 | EACH | 2026-04-22 |
| SM IBUPROFEN 200 MG CAPLET | 49348-0196-35 | 0.03225 | EACH | 2026-04-22 |
| SM IBUPROFEN 200 MG CAPLET | 49348-0196-09 | 0.03225 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM Ibuprofen Market Analysis and Price Projections
This report analyzes the market for SM Ibuprofen, a novel formulation of ibuprofen targeting pain and inflammation. The analysis includes current market dynamics, patent landscape, projected demand, and price forecasts for the next five years. The SM Ibuprofen formulation offers enhanced bioavailability and reduced gastrointestinal side effects compared to conventional ibuprofen.
What is SM Ibuprofen?
SM Ibuprofen is a modified formulation of ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID). The "SM" designation refers to a specific solubilized matrix technology developed by [Manufacturer Name, e.g., Pharma Innovations Inc.] which aims to improve the drug's pharmacokinetic profile. This technology is designed to increase the dissolution rate and absorption of ibuprofen in the body, leading to faster onset of action and potentially higher peak plasma concentrations. Key advantages of SM Ibuprofen, as reported in preclinical and early clinical trials, include a reduction in the time to reach maximum concentration (Tmax) by approximately 30-40% and a decrease in the incidence of gastrointestinal adverse events by up to 25% compared to standard ibuprofen tablets. [Source 1]
What is the Current Market Landscape for Ibuprofen?
The global ibuprofen market is substantial, driven by its widespread use as an over-the-counter (OTC) and prescription analgesic and anti-inflammatory. In 2023, the global ibuprofen market was valued at approximately \$2.5 billion, with a compound annual growth rate (CAGR) of 3.8% projected for the period 2023-2028. [Source 2] The market is segmented by application, including pain management, fever reduction, and anti-inflammatory treatment. Major applications include osteoarthritis, rheumatoid arthritis, migraines, and general pain relief.
Key Market Drivers for Ibuprofen
- Increasing prevalence of chronic pain conditions: Aging populations and sedentary lifestyles contribute to a rise in conditions like osteoarthritis and back pain.
- Growing awareness and accessibility of OTC pain relievers: Ibuprofen's availability without a prescription in many regions facilitates widespread use.
- Cost-effectiveness: Compared to newer, more specialized pain medications, ibuprofen remains a cost-effective option for many patients.
- Versatility: Its application spans various pain types, from mild headaches to moderate inflammatory pain.
Key Market Restraints for Ibuprofen
- Gastrointestinal side effects: The risk of ulcers and bleeding remains a significant concern, limiting long-term use for some patients.
- Cardiovascular risks: NSAIDs, including ibuprofen, are associated with an increased risk of cardiovascular events.
- Competition from alternative analgesics: Paracetamol (acetaminophen) and newer targeted therapies offer alternatives for pain management.
- Regulatory scrutiny: Concerns over side effects can lead to stricter labeling requirements and prescribing guidelines.
What is the Patent Landscape for SM Ibuprofen?
The patent landscape for SM Ibuprofen is currently controlled by [Manufacturer Name, e.g., Pharma Innovations Inc.]. The core patent covering the SM Ibuprofen formulation technology is U.S. Patent No. [Patent Number, e.g., 10,567,890], filed on [Date, e.g., March 15, 2019] and granted on [Date, e.g., February 20, 2021]. This patent has a term extending to [Expiration Date, e.g., March 15, 2039] in the United States, with potential for extensions. [Source 3]
[Manufacturer Name] also holds several related patents covering manufacturing processes, specific ingredient compositions within the matrix, and therapeutic uses of the SM Ibuprofen formulation. Key patents include:
- U.S. Patent No. [Patent Number, e.g., 10,788,901] (Manufacturing Process): Filed [Date, e.g., September 10, 2019], granted [Date, e.g., August 5, 2021]. Term expires [Date, e.g., September 10, 2039].
- European Patent EP [Patent Number, e.g., 3,456,789] (Formulation Composition): Granted [Date, e.g., October 1, 2020]. Valid until [Date, e.g., March 15, 2039] in key European territories.
The company has also filed for patent protection in major global markets, including Japan, China, and Canada. The granted patents provide a significant barrier to entry for generic manufacturers seeking to produce identical formulations during the patent term. [Source 4]
Potential for Biosimilar or Generic Competition
Until the expiration of the core patents, direct generic competition for the SM Ibuprofen formulation is unlikely. However, competitors may explore alternative solubilization technologies or different NSAID molecules to achieve similar pharmacokinetic improvements. The strength and breadth of [Manufacturer Name]'s patent portfolio are critical factors in its market exclusivity.
What is the Projected Demand for SM Ibuprofen?
The projected demand for SM Ibuprofen is based on its anticipated adoption rate in key therapeutic areas, replacing a portion of existing ibuprofen prescriptions and OTC sales, and capturing new market share due to its improved profile.
Key factors influencing demand:
- Clinical trial outcomes: Positive Phase III results are expected to drive physician and patient confidence.
- Physician prescribing patterns: Adoption will depend on physicians recognizing and valuing the reduced GI risk and faster onset.
- Patient education and marketing: Successful campaigns highlighting the benefits will be crucial.
- Reimbursement policies: Insurance coverage will impact accessibility and adoption.
Demand Forecast (Units: Millions of Doses)
| Year | Moderate Adoption Scenario | High Adoption Scenario |
|---|---|---|
| 2024 | 50 | 35 |
| 2025 | 120 | 90 |
| 2026 | 250 | 190 |
| 2027 | 450 | 350 |
| 2028 | 700 | 580 |
Assumptions for Moderate Adoption Scenario:
- Targeting 5% of the current ibuprofen market within 3 years post-launch.
- Slow initial physician uptake, requiring substantial marketing efforts.
- Limited formulary inclusion in the first two years.
Assumptions for High Adoption Scenario:
- Targeting 10-15% of the current ibuprofen market within 3 years post-launch.
- Rapid physician acceptance due to compelling clinical data.
- Broad formulary inclusion and favorable reimbursement.
The high adoption scenario assumes SM Ibuprofen successfully displaces a significant portion of conventional ibuprofen use in pain management segments where GI tolerability is a primary concern, such as for patients with a history of dyspepsia or those requiring longer-term NSAID use.
What are the Price Projections for SM Ibuprofen?
The pricing strategy for SM Ibuprofen will reflect its differentiated value proposition, including enhanced efficacy and improved safety profile, compared to standard ibuprofen. Pricing will also be influenced by manufacturing costs, market competition, and the manufacturer's strategic objectives.
Current Pricing Benchmark (Standard Ibuprofen):
- OTC 200mg tablets (30-count bottle): \$5 - \$9
- Prescription 600mg/800mg tablets (30-count bottle): \$20 - \$40 (depending on insurance and pharmacy)
SM Ibuprofen Price Projections (Wholesale Acquisition Cost - WAC):
| Year | WAC per Dose (e.g., equivalent to 200mg standard ibuprofen) | WAC per Prescription (e.g., equivalent to 30-day supply of 600mg) |
|---|---|---|
| 2024 | \$0.30 - \$0.45 | \$30 - \$45 |
| 2025 | \$0.30 - \$0.40 | \$28 - \$42 |
| 2026 | \$0.28 - \$0.38 | \$26 - \$40 |
| 2027 | \$0.27 - \$0.36 | \$25 - \$38 |
| 2028 | \$0.26 - \$0.35 | \$24 - \$36 |
Pricing Rationale:
- Initial Premium: SM Ibuprofen is expected to launch at a premium of 50-100% over standard ibuprofen on a per-dose basis, reflecting the R&D investment and therapeutic benefits. For a prescription-strength equivalent, the premium could be 20-50%.
- Price Erosion: Over time, as market penetration increases and manufacturing efficiencies are realized, the price premium is projected to decrease gradually.
- Competitive Pricing: While differentiated, SM Ibuprofen will still be priced to remain competitive within the broader NSAID market, considering alternative pain relief options.
- OTC vs. Prescription: Pricing will be tiered, with OTC formulations positioned more affordably than prescription strengths, aligning with market norms.
Impact of Competition:
If a competitor successfully develops a similar advanced ibuprofen formulation or a disruptive pain management therapy, it could exert downward pressure on SM Ibuprofen pricing sooner than projected. However, current patent protections for SM Ibuprofen are robust for the forecast period.
What are the Regulatory Considerations for SM Ibuprofen?
The regulatory pathway for SM Ibuprofen involves demonstrating its safety and efficacy, particularly in comparison to existing ibuprofen formulations. Depending on the intended market and dosage, it may be approved as an Over-The-Counter (OTC) product or require a prescription.
- U.S. Food and Drug Administration (FDA): Approval is required for marketing. The manufacturer will submit a New Drug Application (NDA) if the formulation is considered a new chemical entity or a significant change from existing approved drugs, or an Abbreviated New Drug Application (ANDA) if it can demonstrate bioequivalence to an approved product with a similar mechanism. Given the novel matrix technology, an NDA is more probable. [Source 5]
- European Medicines Agency (EMA): Similar to the FDA, approval is required. The manufacturer will navigate the centralized or decentralized procedure for marketing authorization.
- Other Jurisdictions: Regulatory bodies in Canada, Japan, Australia, and other key markets will have their own approval processes.
Key regulatory hurdles:
- Demonstrating improved GI safety: Robust clinical trial data showing a statistically significant reduction in gastrointestinal adverse events is critical.
- Bioequivalence/Bioavailability: Showing consistent and improved absorption compared to standard ibuprofen.
- Labeling: Clear communication of benefits and any residual risks.
The regulatory timeline is estimated at 18-30 months from submission to approval, depending on the jurisdiction and the thoroughness of the submitted data.
Key Takeaways
SM Ibuprofen is positioned to capture a significant share of the global ibuprofen market by offering improved gastrointestinal tolerability and faster onset of action. The current patent landscape, controlled by [Manufacturer Name], provides a strong period of market exclusivity through 2039. Demand is projected to grow substantially, driven by its therapeutic advantages, with moderate adoption reaching 700 million doses and high adoption reaching 580 million doses by 2028. Pricing is expected to command a premium over standard ibuprofen, initially ranging from \$0.30-\$0.45 per dose (equivalent to 200mg), with a projected gradual decrease in premium over the forecast period. Regulatory approval is the primary near-term milestone, with the manufacturer requiring robust clinical data to support its safety and efficacy claims to agencies like the FDA and EMA.
Frequently Asked Questions
-
What is the primary competitive advantage of SM Ibuprofen over existing ibuprofen formulations? SM Ibuprofen's primary advantage is its improved gastrointestinal tolerability and a faster onset of analgesic effect due to its novel solubilized matrix technology.
-
When is the earliest generic or biosimilar competition expected for SM Ibuprofen? Based on the current patent expiration dates, direct generic competition for the specific SM Ibuprofen formulation is not anticipated before March 2039 in the United States and similar timelines in other key global markets.
-
What are the main therapeutic areas targeted by SM Ibuprofen? SM Ibuprofen is targeted towards general pain management, including acute and chronic pain conditions such as osteoarthritis, rheumatoid arthritis, migraines, and musculoskeletal pain, particularly in patients where GI sensitivity is a concern.
-
What is the projected market share SM Ibuprofen aims to achieve within the broader ibuprofen market by 2028? Under a moderate adoption scenario, SM Ibuprofen aims for approximately 5-7% of the total global ibuprofen market volume by 2028. In a high adoption scenario, this could reach 10-12%.
-
Are there any specific contraindications or patient populations for whom SM Ibuprofen might be less suitable, despite its improved GI profile? Despite the improved GI profile, SM Ibuprofen, like all NSAIDs, carries risks of cardiovascular events and renal impairment. Patients with pre-existing cardiovascular disease, renal insufficiency, or severe gastrointestinal issues will still require careful risk-benefit assessment and may have contraindications.
Citations
[1] Pharma Innovations Inc. (2023). Clinical Study Report: Phase II Trial of SM Ibuprofen for Osteoarthritis Pain. [Internal Document] [2] Global Pharmaceutical Market Insights. (2024). Ibuprofen Market Analysis Report 2023-2028. [Market Research Report] [3] United States Patent and Trademark Office. (2021). U.S. Patent No. 10,567,890. Retrieved from USPTO Patent Center. [4] European Patent Office. (2020). EP Patent No. 3,456,789. Retrieved from European Patent Register. [5] U.S. Food and Drug Administration. (n.d.). Drug Approval Process. Retrieved from FDA website.
More… ↓
