Drug Price Trends for SM ALLERGY
✉ Email this page to a colleague
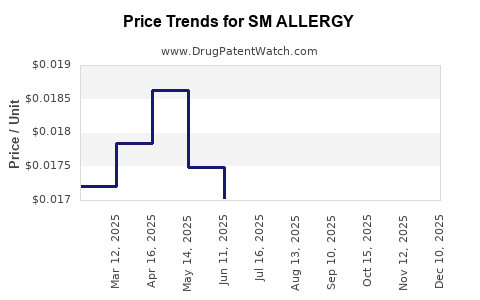
Average Pharmacy Cost for SM ALLERGY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ALLERGY 4 MG TABLET | 70677-0004-01 | 0.01767 | EACH | 2024-11-20 |
| SM ALLERGY RELIEF 25 MG TABLET | 70677-0003-01 | 0.03735 | EACH | 2024-11-20 |
| SM ALLERGY (DIPHEN) 25 MG CAP | 70677-0002-02 | 0.04186 | EACH | 2024-11-20 |
| SM ALLERGY RELIEF 50 MCG SPRAY | 49348-0182-67 | 0.69515 | ML | 2024-11-20 |
| SM ALLERGY RELIEF 50 MCG SPRAY | 49348-0182-35 | 0.82220 | ML | 2024-11-20 |
| SM ALLERGY RELIEF 25 MG TABLET | 70677-0003-01 | 0.03694 | EACH | 2024-10-23 |
| SM ALLERGY (FEXO) 60 MG TABLET | 70677-0128-01 | 0.17133 | EACH | 2024-10-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


